Hearing instrument verification remains a fundamental pillar of audiological best practice, ensuring that prescriptive targets are met and patient outcomes are optimized. This course provides an in-depth review of the latest hardware and software enhancements within Audioscan fitting systems. Throughout this month, the industry is showcasing groundbreaking advancements, and this course focuses on the specific practical applications of these innovations. Key topics include the evolution of ProbeGUIDE+ for pediatric populations, the implementation of new calibrated speech stimuli, advancements in bone conduction device verification through DSL-BCD 2.0 and VerifitLINK, and a preview of upcoming innovations like the RBM microphone and NAL-NL3.
Learning Outcomes
After this course, participants will be able to:
- Discuss hearing instrument verification best practices
- Describe and explain software-assisted probe tube placement
- Describe and explain bone-conduction device verification
Latest Enhancements in Audioscan Software
The field of real-ear measurement (REM) is constantly evolving to improve both clinical efficiency and the accuracy of probe tube placement. Audioscan has introduced several software updates aimed at streamlining these essential procedures, particularly focusing on the challenges of pediatric fittings and patient engagement.
ProbeGUIDE+
ProbeGUIDE was first introduced in 2019 to provide real-time software-assistance for accurately placing the probe tube at the clinically recommended 5 millimeters from the eardrum. The primary goal of this tool is to enhance clinician confidence and streamline a procedure that many clinicians find technically demanding. With the launch of ProbeGUIDE+, this functionality has been significantly expanded to include specialized support for pediatric patients, covering an age range from 1 to 18 years.
The software utilizes a sophisticated acoustic model developed through machine learning. For adult patients (≥ 18 years), the system uses a model that has been trained and optimized on a vast dataset of adult ear canals. By contrast, the new "Child" model is optimized specifically for the unique acoustics of pediatric ears. This pediatric model has been validated through independent studies to ensure effective placement for children as young as one year old.
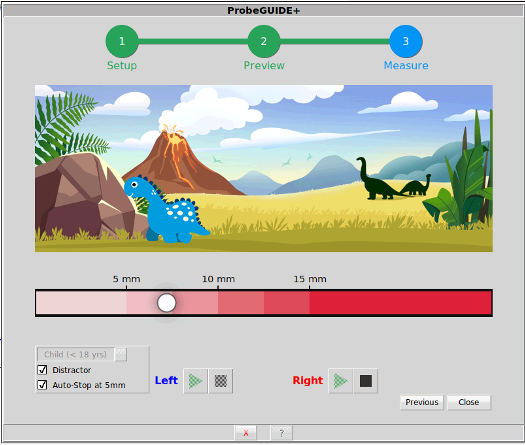
Figure 1. The ProbeGUIDE+ interface allows clinicians to toggle between age-specific acoustic models, ensuring accurate depth prediction across a wide demographic.
To improve the clinical experience with younger patients, ProbeGUIDE+ includes a "dinosaur distractor." When enabled, a dinosaur icon tracks a white indicator ball representing the probe tube's real-time position in the ear canal. The goal is to keep young patients calm and focused during insertion. Feedback from the field indicates that many clinicians also use this feature with adult patients who appreciate the visual engagement. Furthermore, the "Auto-Stop" feature frees the clinician's hands: once the probe tube reaches a 5mm distance from the eardrum target, a green checkmark is displayed and, if the distractor is enabled, the dinosaur receives a celebratory party hat. The measurement then stops automatically after a brief delay. Continuing to perform otoscopy and using the probe tube’s black marker ring to confirm placement is recommended and aligns with clinical best practices.
New Calibrated Speech Passages
Audioscan has expanded its library of calibrated speech stimuli to offer more culturally relevant and engaging options. Selecting a patient's native language or a topically relevant passage can significantly improve patient comfort during the verification process.
Speech-ped: A new narrative speech passage about dinosaurs specifically designed to be used in conjunction with ProbeGUIDE+. This creates a thematically consistent experience for pediatric patients.
Updated Care Passages: The standard English male passage - formerly focused on "eyes and ears" has been replaced with a male-voiced “carrot” passage. A female “carrot” passage is also available.
Linguistic Expansion: New translations of the “carrot” speech passage include Canadian French, French, and Arabic. Arabic represents a new language addition for the system's calibrated stimuli.
Crucially, the speech spectrum for every one of these signals is equivalent to the International Speech Test Signal (ISTS). This ensures that targets and audibility calculations remain consistent regardless of which passage is selected. Clinicians can manage these stimuli by navigating to Settings, selecting Speechmap, and accessing the "Stimuli" menu under calibrated targets.
Sensory Loss Simulator Stimuli
The Sensory Loss Simulator (SLS) serves as a critical counseling bridge, allowing family members to experience sound as the patient hears it via the loudspeaker or the monitor headphones. By demonstrating the tangible impact of hearing loss, clinicians can deepen the family's understanding of the patient's daily auditory struggles.
Recent software updates have integrated all calibrated speech passages from Speechmap into the SLS. This allows for a more personalized counseling experience. For example, a clinician can use the dinosaur narrative to demonstrate a child’s hearing loss to their parents or use a native-language passage for the family of an adult patient. The tool is accessed through the counseling buttons on either the left or right side of the Speechmap screen.
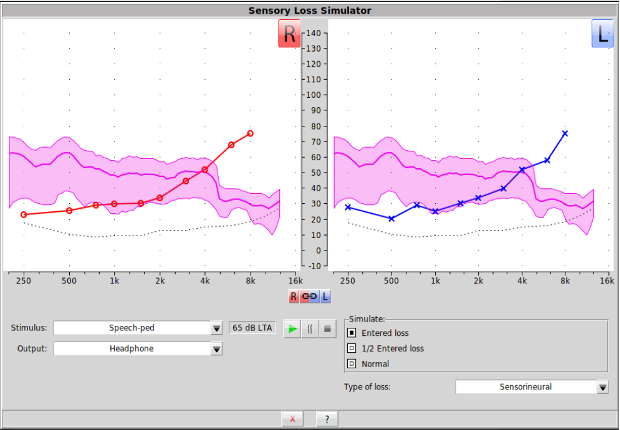
Figure 2. The SLS helps families understand the impact of sensory loss by simulating filtered sound based on the patient's audiogram, now using a wider range of speech stimuli.
Instructional Content Enhancements
Audioscan has updated its instructional help content to support common clinical workflows. These updates are found within the system’s help icon and provide step-by-step guidance for several quality control and specialty verification tasks:
ANSI/IEC Testing: Enhanced guidance for interpreting electroacoustic quality control tests. The user guide now provides specific tolerances to help clinicians determine if a hearing aid meets its factory data sheet specifications.
Tinnitus Masker Verification: New instructions describe using the "Speech-live" stimulus to measure the output of tinnitus maskers, allowing for precise clinical adjustments.
Hearing Protection Verification: This procedure utilizes insertion gain to evaluate the effectiveness of hearing protection. By comparing the real-ear unaided response (REUR) to the response with the hearing protection in the ear, clinicians can measure and evaluate attenuation.
Audioscan Noah Module: PC Integration
The Audioscan Noah module provides essential flexibility for PC-based workflows. It is now available in two distinct installation modes to accommodate different clinical environments:
Noah Installation (Standard): This mode is ideal for clinicians utilizing a Noah 4 database. it optimizes the workflow by facilitating the seamless transfer of audiometric data, RECDs, and comparison curves between the PC and the Audioscan hardware.
Standalone Installation: This new option is designed for clinicians who do not use a Noah-based database. It functions as a standalone desktop application on the PC. While it cannot access a Noah database, it retains powerful features such as the Remote Console (for PC-based control of the hardware), the ability to save session XML files directly to PC folders (using the PC as a local database), report printing, and software update management.
Enhancements in Bone Conduction Verification
Bone conduction device (BCD) verification is a specialized area of practice that has seen significant advancement. The most common application for verification is Speechmap, which enables the evaluation and fine-tuning of aided output relative to prescriptive targets and the patient's dynamic range.
Verifit Skull Simulator
The Verifit Skull Simulator has been an essential accessory for clinicians using the Verifit 2 to measure and adjust bone conduction hearing devices since it’s release in 2018. It is specifically designed for percutaneous devices that connect to an abutment (e.g., Cochlear Baha, Oticon Medical Ponto). In the test box, this simulator replaces the standard 2cc coupler to measure force level output (dB FL) rather than sound pressure level (SPL).
Beyond standard Speechmap measurements, the Skull Simulator enables clinicians to perform quality control via ANSI and IEC tests, verify remote microphones or FM systems, and test adaptive features such as noise reduction and directionality. This ensures that bone‑conduction devices are verified using the same best‑practice clinical protocols as air‑conduction hearing aids.
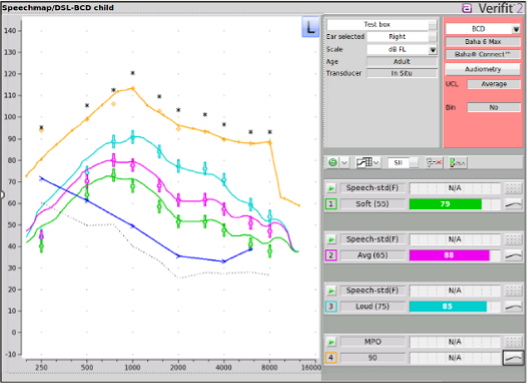
Figure 3. BCD verification measures vibration in force levels (dB FL) and visualizes the output relative to the patient's bone conduction thresholds, prescriptive targets at various input levels, and the device-specific MFO.
DSL-BCD 2.0 and VerifitLINK
In 2024, the integration of DSL-BCD 2.0 provided a breakthrough in workflow flexibility. Clinicians can now use standard bone oscillator audiometric thresholds—including ABR bone oscillator data—in place of in situ thresholds measured through the bone conduction device in the fitting software. The use of bone oscillator thresholds is especially helpful when thresholds through the bone conduction device cannot be obtained for young patients. This capability is enabled by the application of age-based skin‑transmission‑loss corrections that have been validated for both adults and children.
Furthermore, VerifitLINK now supports automated fit-to-target capabilities for the Baha 7. This automation simplifies the workflow by allowing the fitting software to communicate directly with the Verifit 2. The system runs a measurement, determines the distance from the target, and the fitting software makes the necessary adjustments automatically. Typically, a match to target from 250 Hz to 8 kHz can be achieved in approximately two minutes. This feature improves clinical efficiency and ensures that patients receive the intended prescriptive benefit from their first visit.
Future Innovations: RBM and NAL-NL3
Looking ahead to late 2026, Audioscan is developing new solutions for measuring a wide range of bone conduction devices, including transcutaneous bone conduction products.
RBM Microphone
The Real Bone Measurement (RBM) microphone is an upcoming accessory for the Verifit 2, designed to measure bone conduction device vibration on the patient’s head. This industry-first tool is similar in concept to real-ear measurement.

Figure 4. The RBM microphone expands verification to transcutaneous systems, allowing for on-patient vibration measurement.
Evidence supporting this concept has been explored for several years, including recent research from Bill Hodgetts and colleagues at the University of Edmonton and Bo Hakansson and colleagues at Chalmers University in Sweden. Their findings show strong agreement with skull simulator audibility estimates and confirm that the RBM microphone can inform fitting adjustments to improve real-world audibility.
NAL-NL3
The integration of NAL-NL3 in an upcoming 2026 version of our product software will provide clinicians with the latest prescriptive rationale. Per the NAL development team, NL3 optimizes targets based on a decade of machine learning and clinical data gathered since the release of NL2. It will include specialized modules for specific populations and listening environments:
Comfort in Noise Module: Prioritizes listening in noise by reducing loudness while maintaining speech intelligibility.
Minimal Hearing Loss Module: Provides prescriptive targets for individuals with normal to mild hearing loss who still struggle with speech understanding.
Summary
The latest enhancements from Audioscan represent a significant step forward in verification technology, merging clinical precision with increased workflow efficiency. By expanding ProbeGUIDE+ to pediatric populations and diversifying calibrated stimuli to include narrative and new multi-lingual options, clinicians can provide a more tailored and engaging experience for a wider range of patients. Furthermore, the evolution of PC integration through standalone Noah modules and the automation provided by VerifitLINK for bone conduction devices underscores a commitment to reducing procedural time without sacrificing the accuracy that evidence-based practice demands.
Looking toward the future, the introduction of the RBM microphone and the NAL-NL3 prescriptive rationale in late 2026 will address long-standing challenges in the industry. The ability to verify transcutaneous bone conduction systems directly on the patient’s head and the utilization of NAL-NL3’s specialized modules for noise comfort and minimal hearing loss will empower hearing care professionals to serve previously underserved populations more effectively. As these technologies continue to evolve, staying current with these hardware and software advancements ensures that clinicians remain at the forefront of providing optimal audiological care.
References
Denys, S., Latzel, M., Francart, T., & Wouters, J. (2019). A preliminary investigation into hearing aid fitting based on automated real-ear measurements integrated in the fitting software: Test–retest reliability, matching accuracy and perceptual outcomes. International Journal of Audiology, 58(3), 132–140.
Folkeard, P., Pumford, J., Abbasalipour, P., Willis, N., & Scollie, S. (2018). A comparison of automated real-ear and traditional hearing aid fitting methods. Hearing Review, 25(11), 28–32.
McInerney, C. (2026, February 24). Exploring the latest verification enhancements in Audioscan hearing instrument fitting systems (Course No. 41566) [Video webinar]. AudiologyOnline. https://www.audiologyonline.com/audiology-ceus/course/exploring-latest-verification-enhancements-in-41566
Mueller, H. G., & Ricketts, T. A. (2018). 20Q: Hearing aid verification – Will autoREMfit move the sticks? AudiologyOnline. https://www.audiologyonline.com/articles/20q-hearing-aid-verification-will-23532
Persson, A. C., Håkansson, B., Caveramma Mechanda, M., Hodgetts, W. E., Fredén Jansson, K. J., Eeg-Olofsson, M., & Reinfeldt, S. (2023). A novel method for objective in-situ measurement of audibility in bone conduction hearing devices – A pilot study using a skin drive BCD. International Journal of Audiology, 62(4), 357–361. https://doi.org/10.1080/14992027.2022.2041739
Persson, A. C., Håkansson, B., Fredén Jansson, K. J., Reinfeldt, S., & Eeg-Olofsson, M. (2024). Objective verification of audibility in bone conduction devices. International Journal of Audiology, 64(3), 217–223. https://doi.org/10.1080/14992027.2024.2335511
Pumford, J. (2026, February 5). Modern approaches to bone conduction device verification: Current methods, enhancements, and future directions (Course 41588) [Video webinar]. AudiologyOnline. https://www.audiologyonline.com/audiology-ceus/course/modern-approaches-to-bone-conduction-41588
Pumford, J., & Mueller, H. G. (2020). Using autoREMfit for hearing aid fitting and verification: Evidence of accuracy and reliability. Hearing Review, 27(8), 24–27.


