Learning Outcomes
- After this course, participants will be able to discuss the basic principles of evidence-based practice and how they apply to clinical decision-making in the hearing aid selection process.
- After this course, participants will be able to explain outcomes of lab-based studies of Signia’s IX signal processing and how to apply the findings of those studies to clinical decision-making in the hearing aid selection process.
- After this course, participants will be able to describe outcomes of real-world studies of Signia’s IX signal processing and how to apply the findings of those studies to clinical decision-making in the hearing aid selection process.
In an era where evidence-based decision-making is essential to successful hearing aid fitting in the office or the clinic, audiologists need more than puffy marketing claims—they need proof. The objective of this article is twofold. First, it outlines a framework for how Signia conducts its product research and how its approach fits into an evidence-based practice paradigm. Second, the article presents a concise summary of benchtop studies, lab-based measures, and real-world data evaluating the efficacy and effectiveness of Signia’s Integrated Xperience (IX) hearing aid platform and how these studies fit into this framework.
Regardless of your preferred manufacturing partner, in this article, we propose three questions that we believe audiologists should ask hearing aid manufacturer representatives when they visit your clinic, or when you attend one of their seminars. Their answers to these three questions will help audiologists evaluate the quality of their product research and the claims that stem from it.
Lastly, whether you consistently recommend Signia hearing aids or have never fitted them, this article attempts to equip audiologists with the scientific rigor needed to make confident, informed hearing aid selection and fitting decisions that best-align with the principles of evidence-based practice.
The past 30 years: Rapid innovations and a blizzard of research activity.
When a hearing aid manufacturer brings a new product to market, the FDA requires them to use data collected from thoughtful, well-constructed studies to substantiate any claim used in their promotional materials. This requirement ensures that all product claims, as well as all promotional materials originating from these claims, are supported by evidence that has been collected and analyzed in a scientifically valid manner. Given that every new product launch has many claims associated with it, it’s easy for audiologists to be overwhelmed and confused by the abundance of these claims. For example, a manufacturer might claim their newest noise reduction algorithm yields a significant improvement compared to a competitor’s device. A few months later, a different manufacturer might make a similar claim, publishing research that asserts their newest noise reduction feature significantly improves the wearer’s SNR. Which claim is correct? Can they both be correct? The correct answer, as this article highlights, depends on the study design, how the data is analyzed, and finally, how the findings of each study are interpreted and communicated by the consumer of the research, the audiologist.
To appreciate both the volume and type of manufacturer-sponsored product research published today, it helps to provide some historical perspective. In the early 1990s, a well-known hearing aid brand faced significant public scrutiny for claims, often voiced on national TV commercials, that its “noise-cancelling” hearing aid could suppress unwanted background noise to make speech clear in noisy environments. These marketing claims triggered a shift in how the FDA regulated the industry, leading to stricter substantiation requirements for performance claims. The name David Kessler, head of the FDA at the time, still strikes fear in the heart of industry veterans. Under his direction, in 1994, the FDA warned manufacturers that claims regarding the elimination of background noise or improved speech intelligibility in noisy settings (e.g., restaurants or theaters) must be supported by rigorous clinical data. This led to the Hearing Instrument Association forming its own internal committee to review product research and a tightening of how manufacturers conduct the research they use to make product claims.
A few short years after the implementation of these stricter product claim guidelines, hearing aids entered the digital era. The digital era ushered in an avalanche of rapid innovation and product launches. Rather than a single product launch every two to three years, manufacturers were launching new models (and new features) two to three times per year. With each launch, of course, came research to substantiate the product claims that were used to market every new launch.
How to make sense of new product research?
It is estimated that each of the so-called "Big Five" global hearing aid manufacturers releases 5 to 10 technical white papers or publishes journal articles annually to coincide with each new product launch. We can all agree that supporting clinical data is a good thing, but it is difficult for audiologists to make sense of dueling claims from hearing aid manufacturers. Given the abundance of these publications created by hearing aid manufacturers, a legitimate question for audiologists is this: Can this research be trusted? To help answer this question, we turn to the topic of evidence-based practice, which is considered the gold standard for clinical decision-making.
A revisit of evidence-based practice in audiology
Many audiologists have been trained in principles associated with evidence-based decision making. It is helpful to think of evidence-based practice (EBP) as a triad used to answer questions associated with patient care by integrating the following: 1) the interpretation of the best available evidence, 2) clinical expertise and experience, and 3) patient preferences. It is a concept taught in many realms of healthcare (Sackett et al, 1996), including audiology. The use of EBP in audiology was put on the map in 2005 when the Journal of the American Academy of Audiology (JAAA) devoted an entire issue to it. In that special issue, Cox (2005) outlined a detailed 5-step approach to how EBP can be implemented by audiologists. The implementation of EBP in this five-step process includes:
- Generate a focused, answerable clinical question.
- Find the best available evidence,
- Evaluate the validity, importance, and relevance of the evidence,
- Generate a recommendation by combining evidence with clinical expertise and patient preferences
- Evaluate the results of the decision or recommendation and seek ways to improve.
Within these five steps, clinicians are taught to carefully read studies and integrate them into a hierarchy of evidence. This hierarchy is illustrated in Table1. Essentially, the higher the level of evidence (Level 1 is the highest), the stronger or more convincing the evidence. Although it might be tempting, the use of EBP is not about automatically choosing the highest-level research (e.g., randomized controlled trials or systematic reviews) and applying it uniformly. After all, many of these higher levels (Level 1 or 2) of evidence are in short supply when trying to make patient care decisions in the clinic. Instead, EBP requires integrating the three elements of the triad: the best available research evidence, clinical expertise, and patient preferences. High-quality evidence may inform decisions, but it does not replace professional judgment or individual patient preferences.
| Level | Type of Evidence |
|---|---|
1 | Systematic reviews and meta-analyses of high-quality studies |
2 | Randomized controlled trials |
3 | Nonrandomized intervention studies |
4 | Nonintervention studies, cross-sectional surveys |
5 | Case reports |
6 | Expert opinion |
Table 1. The hierarchy of evidence used by audiologists trained in EBP. From Cox, 2005.
Since that 2005 JAAA publication, EBP and the ability to read, interpret, and communicate research have become a key part of decision-making for audiologists. Although few AuD programs offer an entire course on EBP, the American Speech-Language-Hearing Association (ASHA) promotes EBP and expects all accredited programs to incorporate it into their curriculum, often through clinical experiences, capstone projects, and embedded learning within various courses. Therefore, it is expected that US-trained audiologists will follow EBP principles.
The use of EBP principles, moreover, is designed to replace what audiologists might read on the Audiology Happy Hour Facebook site with information from vetted sources. Clinicians steeped in EBP thinking often rely on review articles and sessions at annual meetings to stay current on the latest research findings that can be applied to their decision-making process. (It’s not surprising, for example, that the session, Hearing Aid Research Year in Review, conducted annually at the American Academy of Audiology meeting, is standing room only). As you might expect, recent reports suggest healthcare providers who rely on EBP decision-making, rather than using crowdsourced opinions on social media, deliver better patient outcomes (Lehane et al 2019, Connor et al 2023).
For EBP to be an effective decision-making technique, it requires an abundance of high-quality research, preferably from independent, unbiased sources. A major challenge associated with EBP, especially as it relates to hearing aid selection and fitting, is this: Product launches are fast-paced, cyclical, and commercial-oriented, while independent research is slow, methodical, and academic. In any given year, there is a scarcity of hearing aid studies that coincide with the new features found in a current product launch. Industry research fills this void in the evidence ecosystem. This makes it particularly important that hearing aid manufacturers maintain consistent standards in study design, collaborate with independent researchers, and, when feasible, publish in respected trade journals and peer-reviewed publications.
How does product research fit within an EBP paradigm?
All major hearing aid manufacturers have their own research division, and the data for many of the white papers are generated internally. However, it has become common over the past 20 years or so, for manufacturers to partner with established University hearing aid research sites. When manufacturer-sponsored research is conducted by external researchers, it can be a valuable part of an EBP paradigm. There are both potential strengths (areas that are done well) and limitations (areas that may carry a bias) associated with this product research approach. These biases and limitations do not invalidate evidence from manufacturer-sponsored research, but it changes how this research should be interpreted
Product research occupies a nuanced but legitimate place within evidence-based practice (EBP) for audiology. Because they must align with FDA product claim guidelines and they have ample financial resources, manufacturers are often best positioned to conduct a range of benchtop, lab-based, and real-world studies that would be difficult to fund independently and publish expeditiously. Manufacturer-sponsored studies can provide clinically relevant insights into signal processing, fitting strategies, and outcome trends. However, their inherent biases introduce predictable limitations, including selective (“cherry-picked”) outcomes, optimized conditions, and limited generalizability.
Within an EBP paradigm, however, manufacturer-sponsored research should neither be dismissed nor accepted uncritically. Instead, it should be interpreted as one component of the broader evidence base—most useful when findings converge with independent studies, meta-analyses, and the clinician’s own experiences. The key to integrating manufacturer-sponsored research in EBP decision making is this: Understanding the strengths and limitations of study design, and the generalizability of results to individual patients, is essential to how products areselected and fitted by audiologists. Manufacturer-sponsored research informs practice when integrated thoughtfully, transparently, and in balance with clinician expertise and patient preferences.
Signia’s Approach to Product Research
Signia has a long and rich legacy of conducting product research that is both efficacious and effective. Recall that efficacy is the capability of a new feature in ideal conditions to yield improved wearer benefit, whereas effectiveness is the ability of the new feature to deliver wearer benefit in everyday listening situations. Whenever Signia launches a new product, they ensure its efficacy and effectiveness by conducting a series of rigorous benchtop, laboratory, and real-world studies of the new product. Rather than rely on a single study, the main objective of Signia’s research is to demonstrate a pattern of results that reflects a consistent outcome. Additionally, Signia publishes its studies in a variety of places for the following reasons: White papers, trade journal articles, and peer-reviewed articles each contribute a different perspective on product efficacy and effectiveness, and together they provide a well-rounded evidence base. The outlet (where the study is published) doesn’t determine the quality of the study.
White papers typically focus on explaining a product’s design and performance claims. They are useful for understanding the product rationale and early results so that they correspond with the timing of a product launch. Signia uses white papers to distribute scientific information so that it coincides with a product launch or addresses a common question from the field. Signia white papers rely on study designs that reflect real-world listening situations. The study designs used in Signia white papers often serve as the foundation for further studies that are eventually published in trade or refereed journals. Importantly, white papers sometimes present results from peer-reviewed research in a more popular, digestible (i.e., easier to access) format that is appealing to busy clinicians.
Trade journal articles translate technical information into practical insights for industry professionals, often highlighting laboratory studies, clinical trials, and comparative performance. While less rigorous than academic, peer-reviewed research, studies that are published in trade journals help audiologists assess how new hearing aid technology or a specific feature might perform in practice. In contrast, peer-reviewed articles offer a higher level of scientific rigor and reflect the work of WSAudiology’s own research lab (ORCA) or independent researchers who collaborate with Signia.
Studies published in refereed journals establish scientific credibility, while trade journal articles demonstrate practical effectiveness, and white papers provide context and technical detail. Together, publishing research in all three outlets creates a comprehensive picture of both how well hearing aid technology works and what benefits can be expected in everyday listening situations.
Besides where Signia publishes its studies, it is important to review how Signia conducts its research. There are basically three different types of studies that reflect the efficacy and effectiveness of our product research. Let’s examine each of them.
- Benchtop studies are a controlled evaluation of hearing aids technical performance, conducted without human listeners. The hearing aid is tested using standardized systems such as couplers or the KEMAR, to replicate certain aspects of the human ear and the listening environment. In a benchtop study, researchers measure objective electroacoustic parameters, such as noise reduction under precisely defined conditions. Because variables like input signal, acoustic environment, and device settings are tightly controlled, benchtop studies allow for repeatable, apples-to-apples comparisons between devices or algorithms. They are especially valuable for verifying engineering claims, regulatory compliance, and baseline performance limits. However, benchtop studies do not capture wearer behavior, individual hearing differences, or subjective listening experience. Consequently, a benchtop study provides a high degree of technical efficacy, but its results don’t necessarily tell us about the real-world effectiveness of a feature. However, if done properly (e.g., simulating a relevant listening situation), benchtop study results may suggest the likely outcome of a study that uses human participants.
- Laboratory studies offer more complexity while still maintaining tight control. Usually conducted in soundproof booths, lab studies often involve hearing aid wearers conducting some type of listening task under tightly controlled conditions. Of course, these conditions must reflect some attribute of real-world listening in order be valid. For example, the design of a lab-based study of conversational performance should replicate typical conversational situations with diffuse noise and multiple talkers. While laboratory studies are more realistic than benchtop experiments, they can still differ from everyday conditions.
Real-world studies are conducted in everyday listening situations, such as a restaurant, or in some cases, the wide range of listening situations experienced by the study participant. Researchers observe or measure outcomes under everyday listening conditions, using a validated self-report of satisfaction, benefit, or preference. Real-world studies provide high ecological validity and show how a hearing aid actually performs in practice, but they are vulnerable to several confounding factors such as wear-time, wearer expectations, auditory ecology, and other variables. Because of these factors, many manufacturers rarely, if ever, conduct and publish real-world studies.
With respect to where studies are published and the three different study types, the bottom line is this: Rather than point to one study that reflects a single product claim, Signia believes it is important to examine the entire body of research that supports each new product or feature. When findings from multiple studies align and real-world outcomes support lab and benchtop data, clinicians can have confidence in the efficacy and effectiveness of the product.
Signia’s Current Signature Feature: RealTime Conversation Enhancement (RTCE)
In this final section, we examine Signia’s most current platform, Integrated Xperience (IX) with Real Time Conversation Enhancement. We will review the studies with this product and how the findings of these studies can be integrated into EBP.
In 2023, Signia introduced its Integrated Xperience (IX) platform. This technology uses adaptive beamforming technology to provide the wearer with signal-to-noise (SNR) improvements that result in several potential benefits. The foundation of IX is a proprietary signal processing algorithm known as Real Time Conversation Enhancement (RTCE). As illustrated in Figure 1, RTCE works by analyzing the incoming sound stream to detect different talkers and determine talker locations and turn-taking dynamics. The system then selectively enhances different talkers by deploying appropriate combinations of front-facing monaural and binaural beams. RTCE was designed to improve wearer performance in group conversations in noise.
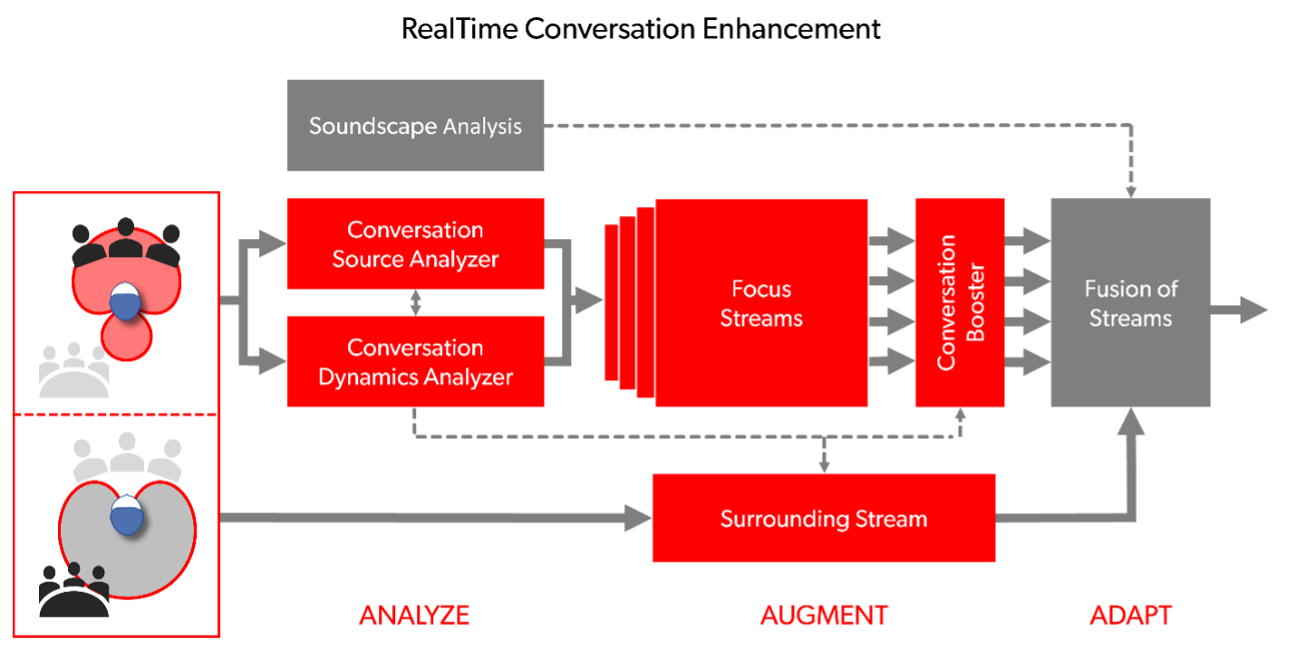
Fig. 1. A schematic of RTCE
The RTCE system builds upon Signia's existing split-processing technology (Jensen et al., 2021), which allows speech signals to be processed using a separate signal processing pathway (less compression, etc.) from that used to process the surrounding scene (more compression). In this way, RTCE allows split processing to be more selective of which regions in front of the listener should be assigned to the hearing aid's "speech" pathway versus its "surroundings" pathway. See Jensen et al (2023) for a detailed description of IX and RTCE.
A brief word on performance vs. benefit
We believe there are three questions that audiologists should ask their manufacturer partners about their research. However, before we get to those questions, it’s important to be grounded in the difference between two separate yet connected concepts: performance and benefit. Performance describes how well a hearing aid feature functions under ideal conditions. Performance is often measured objectively with validated lab tests. In contrast, benefit is related to the individuals’ own everyday experience wearing the hearing aids. Because good lab performance doesn’t always equate to benefit in real-life listening, manufacturers need to generate both types of research. Audiologists should rely on both types of evidence when fitting and recommending devices.
Three questions every clinician should ask manufacturers about their new products
Audiologists who rely on EBP must be able to trust the quality of research from hearing aid manufacturers. To accomplish this, a framework is needed to help them better understand how product research fits into the larger paradigm of EBP. To support this framework – the same framework, coincidentally, used by Signia when they design studies to support a new feature -- we think there are three questions audiologists should ask manufacturers about their product research. The three questions are summarized in the call-out box below.
Question 1: The “Technical” Question: How does the new feature work compared to an existing feature or to a comparable feature from a competitor? Question 2: The “Performance” Question: What are the performance improvements under ideal, controlled conditions compared to an existing feature or comparable feature from a competitor? Question 3: The “Benefit” Question: What are the expected incremental patient benefits in everyday, real-world listening conditions with this feature compared to an existing feature or comparable feature from a competitor? |
Now that readers are familiar with each question, let’s go into detail on how Signia attempts to answer them.
Question 1: The “Technical” Question: How does the new feature work compared to an existing feature or to a comparable feature from a competitor?
To answer this question, manufacturers rely on benchtop studies. Benchtop studies are used to verify that a hearing aid meets specific technical and engineering standards, or put simply, how a new feature works relative to a comparable one. As previously mentioned, benchtop studies are also used to demonstrate how a new feature might outperform existing features from competitors. The first study, summarized next, is an example of a benchtop study used for this purpose.
Benchtop study:
Jensen, N. S., Wilson, C., Kamkar Parsi, H., & Taylor, B. (2023). Improving the signal-to-noise ratio in group conversations with Signia Integrated Xperience and RealTime Conversation Enhancement (Signia White Paper). Signia. Retrieved from www.signia-library.com.
This benchtop study evaluated how well IX improves the signal-to-noise ratio (SNR) in simulated noisy group conversations compared with premium hearing aids from other manufacturers. Signia IX was compared to four premium receiver-in-canal (RIC) competitor devices using the same fitting and test conditions.
Study design: The researchers used a Hagerman phase-inversion technique, a well-accepted method for objectively measuring output SNR at the hearing aid output when both speech and noise are present simultaneously. This involves repeated recordings with phase-inverted noise to isolate and estimate the processed speech and noise signals. A sound-treated room setup simulated a group conversation: two talkers took turns speaking while cafeteria noise played from other directions. Recordings were made on a KEMAR wearing programmable hearing aids. Figure 2 shows the setup for this study.
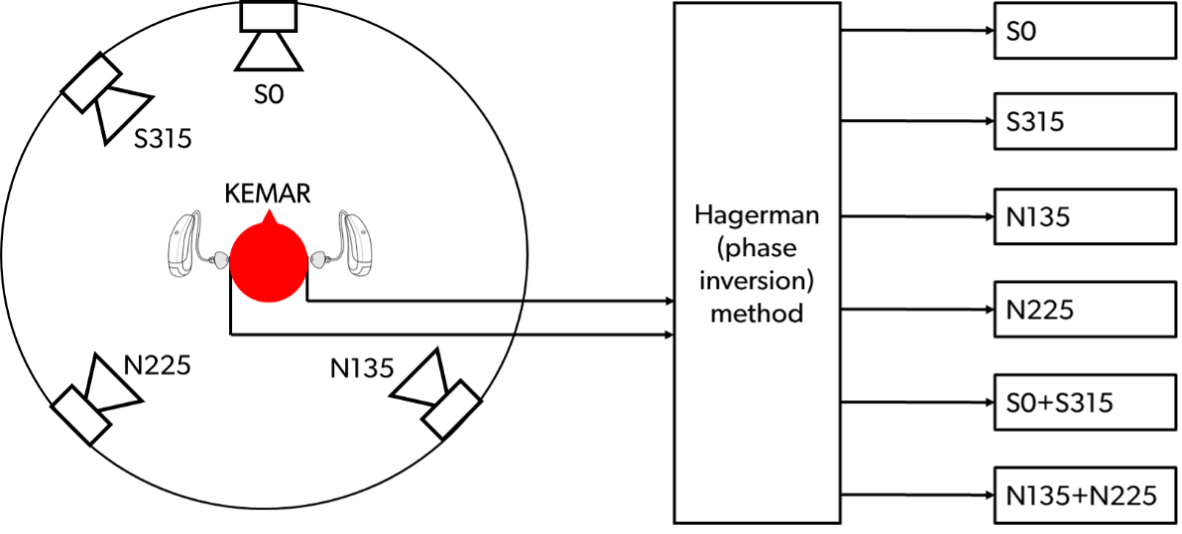
Figure 2. Benchtop study setup designed to simulate back and forth conversation.
Results: The overall mean output SNR for Signia IX was 11.8 dB, compared with around 7.7 dB for the best competitor—a 4.1 dB SNR advantage. Speech enhancement advantages were consistent for both frontal and lateral talker positions, with Signia IX outperforming competitors by about 3.6–3.8 dB in those specific directions. In this specific set-up, IX delivered significantly better SNR in the simulated group conversation than all competitor devices tested. In a follow-up study (Jensen et al 2024b), in which RTCE was compared to a competitor’s DNN-based noise reduction algorithm, RTCE still maintained a 3.2 dB advantage.
How do these findings relate to clinical practice? Using an industry-accepted objective test, IX improves SNR compared to other similar features from competitors. The technical SNR improvements found in this benchtop study are consistent with subsequent research showing measurable improvements in actual speech understanding for people with hearing loss when using Signia IX. These benchtop study results lay the foundation for possible improvements found in laboratory and real-world listening conditions with human study participants.
Question 2: The “Performance” Question: What are the performance improvements under ideal, controlled conditions compared to an existing feature or comparable feature from a competitor?
To answer this question, manufacturers rely on laboratory studies. As we discussed earlier, laboratory studies use highly controlled listening conditions and standardized technologies that bridge the gap between technical benchtop data and real-world performance. The design of lab-based studies should reflect a common wearer challenge associated with hearing aid use, such as speech intelligibility in noise and listening effort. According to MarkeTrak data, these are two of the most common challenges for hearing aid wearers. Signia’s approach to lab-based product research reflects this. Next, we summarize two tracks of lab-based research: Track 1 is speech intelligibility in noise, and Track 2 is listening effort.
Track 1: Speech intelligibility in noise
Jensen, N.S., Samra, B., Kamkar Parsi, H., Bilert, S., & Taylor, B. (2023). Power the conversation with Signia Integrated Xperience and RealTime Conversation Enhancement. Signia White Paper. Retrieved from www.signia-library.com
Study design: The study evaluated whether RealTime Conversation Enhancement (RTCE) in Signia’s Integrated Xperience (IX) hearing aids improves speech understanding in a simulated conversational situation. Study participants consisted of 20 experienced hearing aid wearers with sloping sensorineural hearing loss. They were fitted with Signia Pure Charge&Go T IX hearing aids. Two programs were used for comparison:
- RTCE ON: all IX features, including RealTime Conversation Enhancement activated.
- RTCE OFF: RTCE disabled, but other processing (e.g., split processing) remained active.
A modified version of the Oldenburg Speech in Noise Test (OLSA) (a/k/a Matrix Test) was used to simulate a group conversation with target speech alternating from two frontal locations and noise from behind, as illustrated in Figure 3. During the testing, participants repeated the speech utterance with RTCE either turned on or off. The order was counterbalanced and they were blinded to which setting was active.
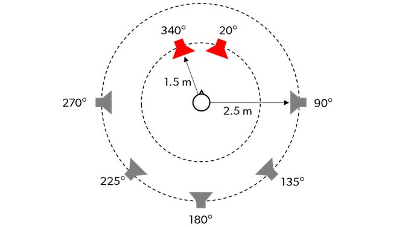
Figure 3. The speaker arrangement for the first lab-based study cited above. The red speakers denote the alternating speech that simulated a group conversation, and the gray speakers denote the background noise.
Results: Results indicated that 95% of participants had improved speech understanding with RTCE activated. The mean Speech Reception Threshold (SRT) benefit was 1.1 dB, statistically significant and translated to about 20% better speech understanding in this simulated conversational scenario. RTCE produced a consistent improvement in speech-in-noise performance across most participants. While the average SRT benefit (~1.1 dB) is modest y, such gains may contribute meaningfully when hearing aids are properly fitted (i.e., prescription gain targets matched) and when worn consistently.
Jensen, N., Samra, B., Best, S., Wilson, C., & Taylor, B. (2025). Improving speech understanding in noisy group conversation: 86% of participants performed better with Signia Integrated Xperience versus key competitor. AudiologyOnline, Article 29273. www.audiologyonline.com
Study design: This lab-based study used a similar design as outlined above but compared Signia IX’s RTCE to a leading competitor hearing aid that uses a DNN-based denoiser. Twenty-seven adults with hearing loss participated in the study. Each person was fitted with Signia IX and with a competitor’s device. A modified OLSA speech-in-noise test measured the speech reception threshold (SRT50) — a signal-to-noise ratio at which a listener can correctly repeat 50% of test sentences. Talkers on the recorded OLSA test were positioned at different angles to stimulate conversations, and café-like noise played in the background to mimic real conversation conditions.
Results: The mean SRT50 with Signia IX was 1.5 dB better (lower) than with the competitor device. This improvement corresponds to an approximate 24% increase in speech understanding in noisy group conversation. Additionally, 86 % of participants performed better with Signia IX than with the competitor. On average, Signia IX with RTCE yielded an improvement in speech-in-noise performance compared with the competitor device, with a mean SRT50 advantage of 1.5 dB, alongwith 86% of participants demonstrating better performance. Under the specific test conditions, this SRT shift corresponds to an estimated increase in speech understanding; however, real-world perceptual benefit is likely to vary across individuals and listening environments and is dependent upon several individual factors such as quality of the fit, the performance-intensity function of the individual, and expectations of the wearer.
How do the findings from these two lab-based speech intelligibility in noise studies relate to clinical practice? These two studies build on technical SNR results from the benchtop study by showing that, in carefully controlled conditions, the SNR advantages of IX can translate into real improvements in speech understanding in complex noisy environments. A 1.5 dB improvement is scientifically valid and directionally positive — but may sit near the lower boundary of what is perceptually meaningful in everyday listening. Recall from McShefferty et al (2016) that a just-meaningful difference (JMD) in SNR — the smallest change that listeners perceive as meaningfully better in real-world listening is ~3dB. Although a small, measured SNR improvement of 1.5 dB as demonstrated in this study does not automatically equate to a perceptually meaningful benefit, an individual’s benefit will vary and depend on several factors including individual slope of the psychometric function, cognitive factors, and the overall quality of the fitting (i.e., optimizing audibility through prescriptive gain target matching and selecting the proper acoustic coupling).
A third study, conducted at the ORCA US laboratory and published in a peer-reviewed journal, used the mismatch negativity (MMN) response, which can be extracted from EEG measurements. The MMN response is generally accepted as a proxy for phoneme discrimination and was used in this study to evaluate the efficacy of RTCE.
Slugocki, C., Kuk, F., & Korhonen, P. (2024a). Using the mismatch negativity to evaluate hearing aid directional enhancement based on multistream architecture. Ear and Hearing, 46(3), 747–757.
Study design: This study used the mismatch negativity (MMN) response to investigate whether IX processing enhances pre-attentive neural detection of speech contrasts in noise. Mismatch negativity (MMN) is an event-related potential (ERP) component measured with EEG. It reflects the brain’s automatic, pre-attentive detection of a change in an ongoing sound stream. A key question was whether RCTE in IX improves bottom-up encoding of speech signals under noisy, multi-talker conditions compared to when the feature is deactivated.
Fifteen older adults (mean age ~72.7 years, range 40–88; 8 females) with moderate-to-severe sensorineural hearing loss were fitted with IX hearing aids, and testing was completed in two conditions: with RTCE either on or off. Participants first completed a phonemic discrimination task (two-alternative forced choice) to find the signal-to-noise ratio (SNR) at which they could reliably distinguish /ba/ vs /da/ syllables in continuous background noise, set up is shown in Figure 4. This individualized SNR was then used in the electrophysiological portion. During passive listening, participants heard an oddball sequence of monosyllabic speech tokens (standards and deviants) presented from two loudspeakers in front (0° and −30° azimuth) within ongoing noise – an arrangement that simulates turn-taking in a group conversation with alternating talkers. The participant’s EEG was recorded while they listened without actively responding. MMN was derived from the difference between responses to deviant and standard stimuli. Figure 4 is an illustration of the lab setup for this study.
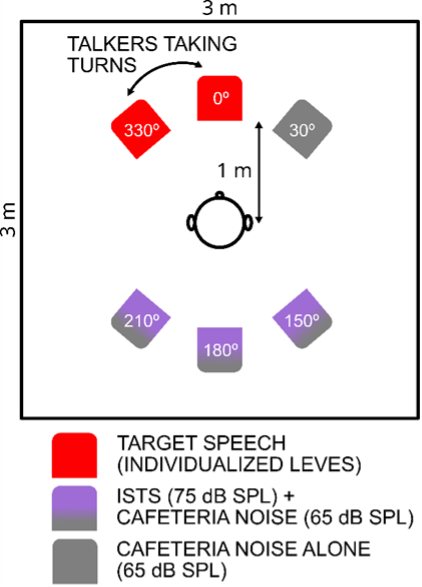
Fig. 4. A schematic of the test set-up for MMN study.
Results: The MMN response was significantly larger when the hearing aids were in the RTCE-ON condition compared to RTCE-OFF. This suggests that RTCE enhances neural sensitivity to phonemic contrasts in noise. These results suggest that RTCE enhances preattentive neural detection of speech contrasts in noisy, multi-talker listening situations, as shown by increased MMN response.
Track 2: Listening Effort
Another potential benefit of improving the wearer’s SNR is a reduction in listening effort. To evaluate Signia’s IX effectiveness in this area, one additional study was conducted.
Slugocki, C., Kuk, F., & Korhonen, P. (2024b). Using alpha-band power to evaluate hearing aid directionality based on multistream architecture. American Journal of Audiology, 33(4), 1164–1175.
Study design: A second study from the ORCA-US lab and also published in a peer-reviewed journal used a different electrophysiologic measure to evaluate the impact RTCE might have on listening effort. This study tested whether RTCE affected neural oscillations in the alpha-band (≈ 8–12 Hz) during speech-in-noise (SiN) listening. Alpha-band power is believed to be a proxy for listening effort, with higher power typically associated with more challenging listening conditions.
Thirteen older adults with sensorineural hearing loss (mean age ≈ 73.5 years; range 62–82 years; 6 women). They were fitted with Signia IX hearing aids and testing was completed in two conditions: with RTCE either on or off. Participants wore the study hearing aids and first completed an adaptive speech in noise test to find the individualized SNR at which they achieved ~50 % correct sentence understanding (speech reception threshold, SRT-50). After those individualized SNRs were determined, sentences presented in noise from two frontal azimuths (0° and −30°) to simulate turn-taking between speakers, and EEG was recorded while participants performed the speech in noise task under both RTCE-OFF and RTCE-ON conditions.
Results: Alpha power was significantly lower when participants listened in the RTCE-ON condition compared to RTCE-OFF. This finding represents a 50% reduction in neural activity associated with listening effort when RTCE is activated. These results are illustrated in Figure 5.
How do the findings from these two lab-based listening effort studies relate to clinical practice? Alpha-band EEG measures might be a useful proxy of listening effort. Results of this objective measure of neuronal activity suggest that RTCE provides a slight edge in improving listening effort compared to hearing aids without this feature.
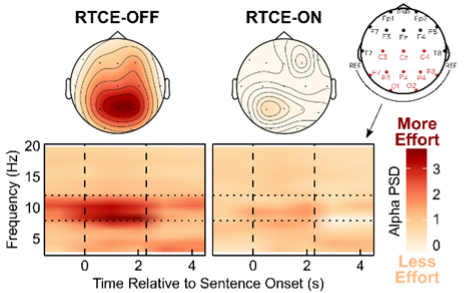
Figure 5. Spectrograms (lower panels) comparing average alpha-band activity measured in RTCE-OFF and RTCE-ON conditions.
Question 3: The “Benefit” Question: What are the expected patient benefits in everyday, real-world listening conditions with this feature compared to an existing feature or comparable feature from a competitor?
In an EBP paradigm, real-world benefit is the highest level of evidence. Because many variables, such as noise and reverberation levels, wear-time, and talker intensity levels cannot be controlled, it is also the most challenging research to conduct.
Folkeard, P., Jensen, N.S., Parsi, H.K., Bilert, S., & Scollie, S. (2024). Hearing at the mall: multibeam processing technology improves hearing group conversations in a real-world food court environment. American Journal of Audiology, 33, 782–792.
Study design: Researchers at Western University evaluated whether RTCE improves real-world group conversation listening for people with hearing loss in a noisy, reverberant food court setting (a busy mall environment) and published their findings in a peer-reviewed journal. The focus was on how well wearers perceived conversational success when multiple talkers and environmental noise in the food court were present. Twenty participants were fitted bilaterally with Signia Pure Charge&Go IX RIC devices that were programmed with two different beamforming strategies, identical to the previously cited studies:
- The RTCE ON: included multiple adaptive beams in the front hemisphere to track speech from different directions and a rear beam to reduce noise.
- The RTCE OFF: a single adaptive front-facing beam and a rear-facing noise-reduction beam (i.e., split processing found in the older generation platform, Signia AX).
Each participant joined two researchers in a noisy and reverberant mall food court, engaging in a turn-taking conversation while wearing the hearing aids. After listening with each beamforming strategy, participants provided ratings for ten dimensions of listening (e.g., understanding speech, clarity, focus). Participants then rated preferences between the two programs across seven aspects of conversation (such as listening effort, sound quality, background noise awareness) while the examiner switched programs without disclosing which was active (single-blinded).
Results showed that, on average, participants gave positive subjective ratings for both the RTCE and comparison strategies across all domains. Pairwise comparisons showed that the multibeam strategy had significantly higher absolute ratings on five of the 10 criteria, including: Speech understanding, Clarity, Focus, Listening effort and Background noise reduction. Figure 6 illustrates the rating differences between the two processing strategies.
Additionally, when asked to choose between programs, participants significantly preferred the RTCE ON setting for all seven rated aspects of the conversation—including aspects like understanding, effort, and sound quality—compared to the RTCE OFF setting.
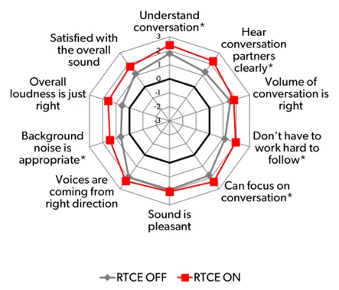
Figure 6. The subjective ratings differences between RTCE on and off.
How do the findings from this study relate to clinical practice? The results of this real-world study build on the four previously discussed laboratory studies and indicate that RTCE is effective in typical everyday life conversational situations.
Conclusions
When following an EBP approach, we encourage audiologists to evaluate the entire body of product research supporting that platform or feature. By asking the three questions posed above and applying a critical eye to all product claims, audiologists avoid brand-driven decision-making. Instead, they can select devices based on patient-centered needs, evidence, and other long-term support considerations -- activities that Signia has avidly supported for generations.
Although product claims often outpace the availability of peer-reviewed clinical evidence from independent sources, audiologists can still trust product research to be a valuable part of an EBP paradigm -- if that research is conducted in a responsible and transparent manner. We believe Signia’s product research achieves these goals and maintains a valuable place alongside other types of high-quality clinical research used in EBP decision-making. By conducting a combination of benchtop, laboratory and real-world studies, and publishing these studies in white papers, trade journals, as well as peer-reviewed publications, whenever feasible, audiologists can put more trust in the quality of the evidence generated by their industry partners. Finally, by conducting a diverse array of studies and publishing them across several channels, Signia enables audiologists to evaluate the validity, importance, and relevance of the evidence as it relates to their own practice.
References
Connor, L., Dean, J., McNett, M., Tydings, D. M., Shrout, A., Gorsuch, P. F., Hole, A., Moore, L., Brown, R., Melnyk, B. M., & Gallagher-Ford, L. (2023). Evidence-based practice improves patient outcomes and healthcare system return on investment: Findings from a scoping review. Worldviews on Evidence-Based Nursing, 20(1), 6–15. https://doi.org/10.1111/wvn.12611
Cox, R. M. (2005). Evidence-based practice in provision of amplification. Journal of the American Academy of Audiology, 16(7), 419–438. https://doi.org/10.3766/jaaa.16.7.3
Folkeard, P., Jensen, N. S., Parsi, H. K., Bilert, S., & Scollie, S. (2024). Hearing at the mall: Multibeam processing technology improves hearing group conversations in a real-world food court environment. American Journal of Audiology, 33, 782–792. https://doi.org/10.1044/2024_AJA-23-00224
Jensen, N. S., Høydal, E. H., Branda, E., & Weber, J. (2021). Improving speech understanding with Signia AX and Augmented Focus [White paper]. Signia. https://www.signia-library.com
Jensen, N. S., Samra, B., Best, S., Wilson, C., & Taylor, B. (2025). Improving speech understanding in noisy group conversation: 86% of participants performed better with Signia Integrated Xperience versus key competitor. AudiologyOnline, Article 29273. https://www.audiologyonline.com
Jensen, N. S., Samra, B., Kamkar Parsi, H., Bilert, S., & Taylor, B. (2023). Power the conversation with Signia Integrated Xperience and RealTime Conversation Enhancement [White paper]. Signia. https://www.signia-library.com
Jensen, N. S., Wilson, C., Kamkar Parsi, H., Samra, B., Hain, J., Best, S., & Taylor, B. (2024). Signia IX delivers more than twice the speech enhancement benefit in a noisy group conversation than the closest competitors [White paper]. Signia. https://www.signia-library.com
Jensen, N. S., Wilson, C., Kamkar Parsi, H., & Taylor, B. (2023). Improving the signal-to-noise ratio in group conversations with Signia Integrated Xperience and RealTime Conversation Enhancement [White paper]. Signia. https://www.signia-library.com
Lehane, E., Leahy-Warren, P., O’Riordan, C., Savage, E., Drennan, J., O’Tuathaigh, C., O’Connor, M., Corrigan, M., Burke, F., Hayes, M., Lynch, H., Sahm, L., & Hegarty, J. (2019). Evidence-based practice education for healthcare professions: An expert view. BMJ Evidence-Based Medicine, 24(3), 103–108. https://doi.org/10.1136/bmjebm-2018-111019
McShefferty, D., Whitmer, W. M., & Akeroyd, M. A. (2016). The just-meaningful difference in speech-to-noise ratio. Trends in Hearing, 20, 1–10. https://doi.org/10.1177/2331216515626570
Sackett, D. L., Rosenberg, W. M., Gray, J. A., Haynes, R. B., & Richardson, W. S. (1996). Evidence-based medicine: What it is and what it isn't. BMJ, 312(7023), 71–72. https://doi.org/10.1136/bmj.312.7023.71
Slugocki, C., Kuk, F., & Korhonen, P. (2024a). Using alpha-band power to evaluate hearing aid directionality based on multistream architecture. American Journal of Audiology, 33(4), 1164–1175. https://doi.org/10.1044/2024_AJA-24-00057
Slugocki, C., Kuk, F., & Korhonen, P. (2024b). Using the mismatch negativity to evaluate hearing aid directional enhancement based on multistream architecture. Ear and Hearing, 46(3), 747–757. https://doi.org/10.1097/AUD.0000000000001463



