Learning Outcomes
After this course learners will be able to:
- Describe Δƒ thresholds in children.
- Compare and contrast the effect of APD or SLI on frequency discrimination ability.
- Understand the importance of supra threshold Δƒ detection.
Introduction
Frequency discrimination is important in everyday perception of phonemes, musical notes, or environmental sounds. Spectral processing is particularly important for language acquisition, with reports that poor frequency discrimination in young children places them at risk for difficult language learning and poor outcomes (e.g., Noonan, Bavin, Ong, & Grayden, 2007). In terms of speech perception, the detection of frequency differences (Δƒ) allows for distinguishing between formant frequencies (e.g., Bailey and Snowling, 2002) in phonemes such as the vowels or fricatives.
Background
Children with Auditory Processing Disorder (APD) or Specific Language Impairment (SLI) often have frequency discrimination deficits (e.g., Kleindienst and Musiek, 2011). Pediatric (C)APD/APD is a disorder of auditory perception in the absence of peripheral hearing loss; it is characterized by four main groups of symptoms: difficulty listening in noise, poor dichotic listening abilities, nonverbal auditory perceptual deficit and poor sound localization (ASHA, 2005; AAA, 2010). SLI is characterized by a difficulty in acquiring language, in the absence of known neurological disorder, or cognitive, emotional, or sensory deficit (Leonard, 2014). Poor detection of Δƒ is reported in these children (Rota-Donahue, Schwartz, Shafer, & Sussman, 2016). However, other reports of poor frequency discrimination in school aged children are sometimes conflicting (e.g., Ferguson, Hall, Riley, & Moore, 2011). This occurs mainly because speech-language pathologists examine different aspects of communication, without fully assessing auditory processing ability in a given child (e.g., McArthur & Bishop, 2005). As a result, the presence or absence of APD could explain the discrepant findings.
The prevalence of pediatric APD is approximately 3% of school-aged children (Musiek and Chermak, 2014), it is often associated with other developmental deficits (Rota-Donahue, Schwartz, Shafer and Sussman, 2014); and its diagnosis leads to appropriate treatment plans. Testing children with problems in school for APD is therefore important. Although often associated with language deficits, APD can be identified independently of SLI (e.g., Musiek & Chermak, 2014). Additionally, SLI can be present without APD, but it is often studied without the inclusion of auditory perception testing.
When clinical profiles are described thoroughly, children can be found to have APD-only, SLI-only, or both APD and SLI (e.g., Boscariol, Guimarães, Hage, Garcia, Schmutzler, Cendes, & Guerreiro, 2011; Rota-Donahue, Schwartz, Shafer, & Sussman, 2016). Other studies also suggest that pediatric APD and SLI can be co-morbid (see Bishop 2007 for a review). However, if there is some documented overlap between the two disorders, APD and SLI, their co-occurrence is common but not always present (e.g., Rosen, 2005). It is therefore essential to ensure that children with developmental impairments be tested for both language and auditory processing abilities (Musiek & Chermak, 2014), as recommended by the American Speech-Language Hearing Association (ASHA) and the American Academy of Audiology (AAA) (ASHA, 2005; AAA, 2010).
Pediatric auditory processing deficits are rarely isolated (Chermak, Musiek, & Weihing, 2017). Overall, atypical development can be seen as a spectrum of disabilities (Boscariol et al., 2011) going from mild, for children with only one disorder (i.e., APD-only or SLI-only), to more severe, for children with more than one disorder (i.e., both APD and SLI). Because of the frequent association of language impairment in children with APD, teasing apart the auditory portion of the auditory processing disorder from other deficits is problematic. One way to focus on auditory perception only is to use non-speech signals in relatively simple tasks, to avoid linguistic stimuli that involve both detection and higher order processing (Hickok & Poeppel, 2000). The detection of frequency differences is a basic auditory task that can measure nonverbal auditory processing. Frequency discrimination is an important aspect of auditory skills (Kleindienst & Musiek, 2011), especially in distinguishing between speech sounds. Frequency discrimination can be examined behaviorally and electrophysiologically, we will review Δƒ, detection in typically developing children and in children with APD or SLI.
Typically Developing (TD) Children
Behavioral Δƒ Detection
Behaviorally, the detection of Δƒ was first measured in adults. Results showed that participants were able to detect small frequency differences in the order of 1% of the base frequency, or 10 Hz at 1000 Hz (Yost, 2007). However, Δƒ threshold in typically developing children were not established. In 2010, AudiologyOnline published an article on the topic (Rota-Donahue, 2010) to fill a gap in the literature. Indeed, until then there was little information regarding the smallest difference in frequency typically developing children could detect. To answer that question children - classified as being typically developing because they had no known neurological deficits, and had passed screenings for hearing loss, attention deficits, APD and SLI - were involved in a psychoacoustic experiment that included continuous tones with and without a frequency change (Rota-Donahue, 2010). There were two base frequencies: 500 Hz and 3000 Hz, and several magnitudes of frequency differences in the continuous tones: between 0.25 % and 40 % of the base frequency. The results showed that typically developing children had Δƒ thresholds close to adults’ Δƒ thresholds, in the order of 1% of the base frequency at 500 Hz and 3000 Hz.
Two years earlier, Moore and his colleagues (Moore, Ferguson, Halliday, and Riley, 2008) had already studied frequency discrimination in school-aged children and had found that “good performers” were able to detect small frequency differences, in the order of 1% of the base frequency at 1000 Hz. However, other children were “poorer performers” and had Δƒ thresholds in the order of 10% of the base frequency. Since these participants were not tested for developmental deficits, interpretation of the results is difficult, as the presence or absence of APD or SLI might have influenced the findings. Earlier studies on frequency discrimination in children also showed great variability and the authors suggested that the variation in methods and psychoacoustics measures could explain the mixed results (e.g., Sutcliffe and Bishop, 2005).
Brain Bases of Frequency Discrimination
The Auditory Evoked Potential (AEP) component of choice in the detection of small frequency differences is the Mismatch Negativity (MMN), because it can be elicited by small Δƒs. The MMN is elicited when a deviant stimulus is introduced in a chain of identical stimuli. The waveforms for the standard stimulus and for the deviant stimulus are averaged separately. Around 100 ms after the onset of the stimulus, a larger negativity of the response is observed for the deviant stimuli. This negativity is called MMN; the difference waveform is obtained by subtracting the deviant from the standard. The MMN in response to different Δƒs was first recorded in adults, Δƒthresholds in the order of 2% of the base frequency, or 20 Hz at 1000 Hz (Sams, Paavilainen, Alho, & Naatanen, 1985). Subsequent studies also showed that children could detect small Δƒs, using the MMN (Ahmmed, Clarke, & Adams, 2008; Rota-Donahue et al., 2014).
Responses in changes in frequency can also be recorded using the P1-N1-P2. This obligatory auditory evoked response is obtained about 100 ms after the onset of the stimulus. The first peak P1 is observed around 50 ms, N1 around 80 ms, and P2 around 180 ms. The important factor in children is that the morphology is different. During childhood P1 shifts in latency and the complex has only two peaks; the valley N1 emerges only around 9 years of age (Ponton et al., 2008). When this obligatory response is elicited by a change in a stimulus it is called the Acoustic Change Complex (Martin & Boothroyd, 1999). In children in can be elicited by Δƒs in the order of 1% of the base frequency at 500 Hz, and 3000 Hz (Rota-Donahue, 2010).
The same obligatory response recorded at the level of the temporal electrodes T7 and T8 is called the T-complex. Three peaks are seen between 70 ms and 160 ms: Na,Ta and Tb. Currently this site of recording is considered important, as it is closest to the auditory cortex on each side of the head (Shafer, Schwartz and Martin, 2011).
Children with APD or SLI
Behavioral Δƒ Detection
As reported above, Moore and colleagues showed that “poor performers” on a frequency discrimination had Δƒ thresholds in the order of 10% of the base frequency. In children with SLI who are not tested for APD, Δƒ thresholds are reported around 15 % of the base frequency (Nickisch & Massinger, 2009), or close to 1% of the base frequency (Hill, Hogben, & Bishop, 2005). The incomplete clinical profile could explain the difficulty in establishing Δƒ thresholds in children with SLI who are not tested for APD (Hill et al., 2005; Nickisch & Massinger, 2009). The discrepant reports could be due to the presence or absence of APD in these children. In a 2014 study, we analyzed detection of three magnitudes of Δƒfor 30 children. Our participants were tested for auditory processing and language impairment; they were also screened for hearing loss, attention deficits, non-verbal intelligence, and known neurological deficits. The detection of small frequency differences was analyzed for three magnitudes of Δƒs: a small Δƒ: 1000 Hz to 1020 Hz, a medium Δƒ: 1000 Hz to 1050 Hz, and a large Δƒ: 1000 Hz to 1150 Hz. To account for false alarm rate and hit rate, the sensitivity index d’ was calculated for each participant using the signal detection theory (Green and Swets, 1974). The sensitivity index d’ is reported below in figure 1.
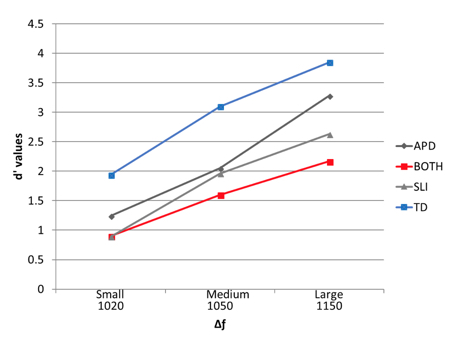
Figure 1: Average d’ values for three degrees of frequency differences: the small Δƒ, 1000 Hz to 1020 Hz; the medium Δƒ, 1000 Hz to 1050 Hz; and the large Δƒ, 1000 Hz to 1150 Hz., for the four groups of participants. A d’ value of 1 and above indicates discrimination above chance level. A perfect score on this scale is 4.6.
All groups performed better when the difference was large and worse when the difference was small. The TD group performed better than the children with APD-only, SLI-only or both, with children in the both group performing worse. The clinical groups were small, but when we accounted for the entire group of participants’ variances using a multiple regression analysis, APD status or SLI status affected frequency discrimination differently. Poor scores on a language test predicted poorer scores on the frequency discrimination for the large Δƒ. In contrast, poor scores on an auditory processing test predicted poor scores on the frequency discrimination for the small Δƒ. These results suggested that this frequency discrimination task involved both an auditory perceptual aspect for the identification of the small Δƒ, and a higher cognitive aspect for the identification large Δƒ (Rota-Donahue, Schwartz, Shafer, & Sussman, 2016).
In addition, to showing that different clinical profiles such as SLI and APD affect frequency discrimination differently, this study also highlights the importance of testing suprathreshold Δƒdetection. As we know, suprathreshold perception is important because it is related to everyday listening. However, it is rarely reported in the literature, as Δƒ thresholds results prevail, so further studies in this area are needed.
Electrophysiology
In children with SLI, APD, or both, brain anomalies in the auditory cortex have been reported in fMRI studies (e.g., Boscariol et al., 2011) and electrophysiology (e.g., Bishop, 2007, for a review). Some MMN studies with children with SLI reported reduced MMN amplitude in children with SLI (e.g., Rinker & Kohls, 2007), while other studies showed no difference between the MMN of the clinical group and the control group (e.g., Uwer, Albrecht, & von Suchodoletz, 2002). Electrophysiologically Bishop reviewed the literature in 2007 and found reported anomalies the level of the obligatory P1, N1, P2. The author suggested that children with atypical development had immature or delayed development of the auditory pathways, as manifested by different latency and amplitude of the P1 in children with SLI when compared to their age-matched peers. Reduced MMN amplitudes are also reported in these children
A more recent study (Ahmmed et al., 2008) found some MMN amplitude differences in response to Δƒs elicited at two rates of presentation. In that study, children with SLI and age-matched peers participated in a behavioral gap detection task. Results showed that children with SLI who performed poorly on the gap detection task had reduced MMN amplitude compared to controls. However, children with SLI who performed well on the gap detection task had MMN amplitude comparable to that of the children in the control group. In general children who performed poorly on the gap detection task had the worst MMN morphology. Although these participants were given a gap detection test, that is part of the behavioral battery for APD (ASHA, 2005; AAA 2010), they were not tested further for auditory processing disorder. Thus, interpretation of these findings remains uncertain because the presence or absence of APD might have influenced these results.
A similar study was conducted in 2014 with thirty children tested for APD and SLI. Results showed no significant differences between groups at the level of the obligatory P1, N1, P2, at electrode FCZ, but some differences at temporal sites T7 and T8. The main component of interest in that report was the MMN. Weak MMN or small MMN amplitudes were recorded in atypical children. The most significantly finding was that children with both APD and SLI had the weakest MMN. Their overall MMN response was significantly worse than that of participants in the other groups (Rota-Donahue et al., 2014).
Conclusion
This review has confirmed the importance of spectral processing, especially in children with APD or SLI. Depending on the clinical profile different aspects of frequency processing are affected: difficulties at threshold - or with the smallest Δƒ- are associated with APD; while difficulty above threshold - where the detection of a Δƒis auditorily easier - is more likely to be seen in children with SLI. More studies using a range of frequency differences are needed to help isolate the nature of auditory processing deficits in children with APD vs. SLI, with the recording of Δƒ thresholds and the analysis of Δƒ suprathreshold. In sum, it is important to test children with developments problems for APD and SLI, as these disorders are often co-morbid. However, the main reason for assessing auditory processing in children is that the presence or absence of APD leads remediation plans tailored to the individual child’s needs.
Acknowledgement
This work was supported in part by a grant from the American Hearing Research Foundation. (Christine Rota-Donahue, Jan. 2015 to Dec. 2017).
References
Ahmmed, A. U., Clarke, E. M., & Adams, C. (2008). Mismatch negativity and frequency representational width in children with specific language impairment. Developmental Medical Child Neurology, 50(12), 938-944.
American Academy of Audiology. (2010). Clinical practice guidelines: diagnosis, treatment and management of children and adults with central auditory processing disorder. Clinical Practice Guidelines, 1-51.
American Speech-Language-Hearing Association. (2005). (Central) auditory processing disorders. Technical Report, 1-20.
Bailey, P.J., & Snowling, M.J. (2002). Auditory processing and the development of language and literacy. British Medical Bulletin, 63, 135-146.
Bishop, D. V. (2007). Using mismatch negativity to study central auditory processing in developmental language and literacy impairments: Where are we, and where should we be going. Psychological bulletin, 133(4),651-672.
Boscariol, M., Guimarães, C. A., Hage, S. R., Garcia, V. L., Schmutzler K. M., Cendes F., & Guerreiro, M. M. (2011). Auditory processing disorder in patients with language-learning impairment and correlation with malformation of cortical development. Brain Development, 33(10), 824-831.
Chermak, G. D., Musiek F. E., & Weihing J. (2017). Beyond controversies: the science behind auditory processing disorders. Hearing Review, 24(5), 20-24.
Ferguson, M. A., Hall, R. L., Riley, A., & Moore, D. R. (2011). Communication, listening, cognitive and speech perception skills in children with auditory processing disorder (APD) or specific language impairment (SLI). Journal of Speech, Language, and Hearing Research, 54, 211-227.
Green, D., & Swets, J. A. (1974). Signal detection theory and psychophysics. Huntington, NY: Robert Krieger Publishing Co.
Hickok G., & Poeppel, D. (2000). Towards a functional anatomy of speech perception. Trends in Cognitive Science, 4(4), 131-138.
Hill, P. R., Hogben, J. H., & Bishop D. M. V. (2005). Auditory frequency discrimination in children with specific language impairment: a longitudinal study. Journal of Speech, Language, and Hearing Research, 48, 1136-1146.
Kleindienst, L., & Musiek, K. (2011). Do frequency discrimination deficits lead to specific language impairments? The Hearing Journal, 64, 4, 10-11.
Leonard, L.B. (2014). Children with Specific Language Impairment. 2nd ed. Boston, MA: MIT Press.
McArthur, G. M., & Bishop, D. V. (2005). Speech and non-speech processing in people with specific language impairment: A behavioural and electrophysiological study. Brain and language, 94(3),260-273.
Martin, B. A., & Boothroyd, A. (1999). Cortical, auditory, event-related potentials in response to periodic and aperiodic stimuli with the same spectral envelope. Ear & Hearing, 20(1), 33-44.
Moore, D. R., Ferguson, M. A., Halliday, L. F., & Riley, A. (2008). Frequency discrimination in children: perception, learning, and attention. Hearing Research, 238(1-2), 147-154.
Musiek, F. E., & Chermak, G. D. (2014). Handbook of Central Auditory Processing Disorder. Second Edition. San Diego: Plural Publishing.
Nickisch, A., & Massinger, C. (2009): Auditory processing in children with specific language impairments: are there deficits in frequency discrimination, temporal auditory processing or general auditory processing? Folia Phoäniatrica et Logopaedica, 6, 323-328.
Noonan K., Bavin E., Ong B., and Grayden D., 2007. Child Language Seminar, 2007.
Ponton, C., Eggermont, J. J., Khosla, D., Kwong, B., & Don, M. (2002). Maturation of human central auditory system activity: separating auditory evoked potentials by dipole source modeling. Clinical Neurophysiology: Official Journal of the International Federation of Clinical Neurophysiology, 113(3),407-420.
Rinker, T., Kohls, G., Richter, C., Maas,V., Schulz, E., & Schecker, M. (2007). Abnormal frequency discrimination in children with SLI as indexed by mismatch negativity (MMN). Neuroscience Lett., 413,2, 99-104.
Rosen, S. (2005). “A riddle wrapped in a mystery inside an enigma”: defining central auditory processing disorder. American Journal of Audiology, 14,139-142.
Rota-Donahue, C. (2010). Neurophysiological and behavioral processing of small frequency changes in 8-11-year-old children. AudiologyOnline, 7/12/2010.
Rota-Donahue, C., Schwartz, R. G., Shafer V., & Sussman, E. S. (2014). Neurophysiological bases of frequency discrimination in children with Auditory Processing Disorder or Specific Language Impairment. Ann Harbor, MI: ProQuest LLC.
Rota-Donahue, C., Schwartz, R. G., Shafer V., & Sussman, E. S. (2016). Perception of small frequency differences in children with auditory processing disorder or specific language impairment. Journal of the American Academy of Audiology, 27, 489-497.
Sams, M., Paavilainen, P., Alho, K., & Naatanen, R. (1985). Auditory frequency discrimination and event-related potentials. Electroencephalography and Clinical Neurophysiology, 62(6), 437-448.
Shafer, V. L., Schwartz. R.G., & Martin, B. (2011). Evidence of deficient central speech processing in children with SLI: the T-complex. Clinical Neurophysiology 122(6),1137-1155.
Sutcliffe, P., & Bishop D. (2005). Psychophysical design influences frequency discrimination performance in young children. Journal Experimental Child Psychology, 91(3), 249-270.
Uwer, R., Albrecht, R., & von Suchodoletz, W. (2002). Automatic processing of tones and speech stimuli in children with specific language impairment. Developmental Medicine and Child Neurology, 44(8),527-532.
Yost, W. (2007). Fundamentals of hearing (fifth edition). Bingley, UK: Emerald.
Citation
Rota-Donahue, C. (2018). Frequency discrimination in children with auditory processing disorder or specific language impairment. AudiologyOnline, Article 23486. Retrieved from https://www.audiologyonline.com


