Introduction
Traditional electronic hearing aids have evolved to various stages of development since their inception in the late 19th century. The continuing development of electronic amplification using technologies such as carbon microphones, vacuum tubes, battery technology, transistors, digital technology, miniaturization, multi-channel processing, directional microphones, adaptability, open fittings, connectivity, and other significant advances in amplification have all contributed to the development of products that offer substantial assistance to the hearing impaired. Today's digital hearing instruments are extremely advanced, offering maximum performance and reducing many of the difficulties encountered in earlier designs. These devices can provide solutions for hearing loss that are well beyond the expectations of hearing impaired individuals.
Current benefits of amplification notwithstanding, these products do not enjoy universal acceptance by the hearing impaired population. George (2006) indicates that only 21.4% of the estimated twenty-eight million Americans with hearing loss utilize amplification regularly (Figure 1). In an interview by Strom (2005), Kochkin indicates that of the approximately 6 million hearing instrument users, 35-50% are not satisfied with the benefit from their instruments. The typical reasons for reduced use of amplification vary, but poor sound quality, feedback, limited frequency range, occlusion, pain or irritation, moisture, social stigma, and cosmetic issues are frequently cited as major concerns.
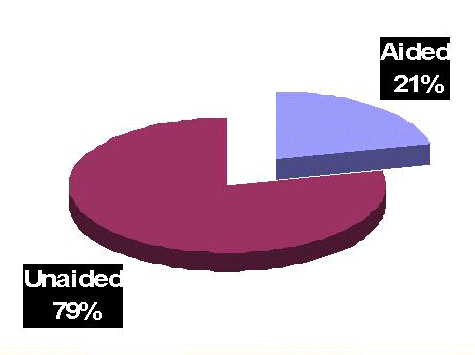
In the past 10 years, improvements in hearing devices have substantially helped feedback control, widening the frequency range, and, to some degree, sound quality; however, patients still experience the stigma and practical problems of using these devices. Recognizing that stigma and sound quality will always be issues for those that use traditional hearing instruments, research has pursued a quest to find an efficient, practical method of middle ear implantation that would counteract many of the difficulties of hearing aid use.
Where Did Middle Ear Implant Products Originate From?
Middle ear implants started in 1935 when Wilska (1935) experimented with iron particles placed on the tympanic membrane. Wilska generated a magnetic field from an electromagnetic coil inside an earphone, which caused the iron filings to vibrate in synchrony with the magnetic field. This vibration in turn caused the eardrum to vibrate, which allowed sound to be transduced to the inner ear in the normal fashion. In the late 1950s, Rutschmann (1959) successfully stimulated the ossicles by gluing 10 mg magnets onto the umbo. This configuration used an electromagnetic coil to create a modulated magnetic field and vibrate the ossicles. Devices actually placed into the middle ear did not appear until the 1970s (Goode, 1970, 1988; Fredrickson et al., 1973; Nunelly et al., 1976).
These early experiments paved the way to the development of middle ear implant devices that were included in actual Food and Drug Administration (FDA) clinical trials.
Types of Transducers Utilized in Middle Ear Devices
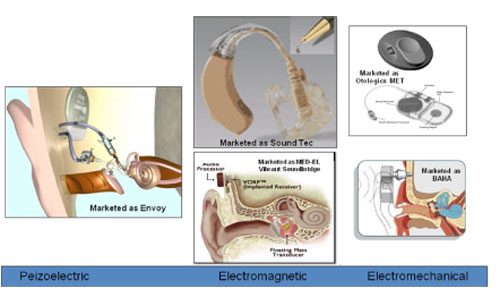
Three general types of transducers are used in middle ear implants, each with advantages and disadvantages related to power, efficiency, frequency response, and reliability. The types of transducers used in middle ear implants are shown in Figure 2 and consist of the following:
- Piezoelectric,
- Electromagnetic, and
- Electromechanical.
Piezoelectric devices make use of the properties of piezoelectric materials or crystals. Specifically, the property that when a voltage is applied, a deformation or bending of the material results. This deformation provides the mechanical energy to stimulate the ossicular chain or inner ear. Piezoelectric devices are found in two configurations, the monomorph and bimorph. The monomorph uses expansion and contraction directly to provide the displacement, whereas a bimorph uses two pieces of piezoelectric material bonded together with opposite polarities, causing the structure to bend.
Electromagnetic transduction devices consist of a magnet, generally rare earth (either samarium cobalt or neodymium iron boron), and an energizing coil. The magnet is attached to the ossicular chain, tympanic membrane, or the inner ear (round window or fenestra). A fluctuating magnetic field is generated when the coil is energized by a signal, which corresponds to an acoustic input. This magnetic field causes the magnet to vibrate. The vibrating magnet then, in turn, causes movement of either the ossicular chain or the cochlear fluids directly. The force generated is inversely proportional to the square of the distance between the coil and magnet (e.g., doubling the distance between the magnet and coil results in an output of one-fourth the force); therefore, these two components must be maintained in close proximity to one another to realize an efficient system.
Electromechanical transduction is actually a variation of electromagnetic transduction. In electromagnetic devices, it is often difficult to control the spatial relationship of the magnet and coil. Because the magnet is attached to one portion of the anatomy and the coil attached to another, the patient may observe a wide variation in performance. As the relationship between the coil and the magnet changes, it results in a variance of the frequency response and a significant fluctuation of output levels. An electromechanical device has an energizing coil and a magnet that are housed within an assembly. This optimizes the spatial and geometric relationship to avoid variability. The electromechanical transducer directly connects to the ossicular chain to transmit the mechanical energy produced.
Attempts at Middle Ear Devices
Indications for middle ear implants vary widely among the many different devices under investigation, and patient selection criteria greatly depend on the type of transducer and the method of implementation. Chasin (1997, 1998, 2002) indicates that several groups have conducted pre-clinical research in the field of middle ear implants and have collected data using a variety of approaches and techniques. The results obtained by otologic researchers demonstrated much promise, and several of the efforts have led to human clinical trials.
The early experiments of Fredrickson et al. (1973) are some of the more prominent pre-clinical research in middle ear implantable devices. Experimenting with electromagnetic technologies in rhesus monkeys, Fredrickson and colleagues demonstrated that an implantable apparatus could provide a safe, efficient transmission of acoustic energy. In their classic work, a magnet was attached to the head of a monkey's stapes; then, an adjacent copper wire created an electromagnetic induction and vibrated the magnet, inducing a signal into the hearing system.
The efficacy of these and other electromagnetic studies led Hough et al. (2001), in collaboration with SOUNDTEC, to conduct FDA clinical trials on an electromagnetic device that incorporated a magnet implanted at the incudostapedial joint. The electromagnetic coil and processor was integrated in a behind-the-ear or in-the-ear canal device.
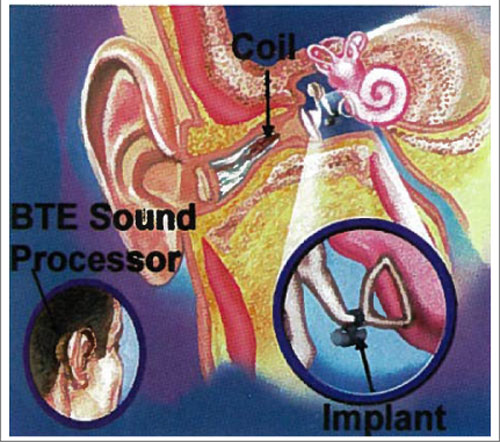
This first study led to a second FDA study reported by Matthews (2002). In this clinical trial, the SOUNDTEC Direct System provided higher functional gain, a significant increase in speech discrimination in quiet, and comparable speech discrimination in background noise. Although it was reported that patients preferred the implant (to an acoustic hearing aid), as evidenced by APHAB questionnaire scores, the obvious disadvantage of this device is the sustained occluded ear canal, which is one of the major complaints of acoustic hearing aids. Difficulties with magnets, output, and gain have caused this device to be discontinued and withdrawn from the market.
Symphonix Devices in San Jose, California marketed the Vibrant Soundbridge, which received both FDA approval in the U.S. in August 2000 and the European CE-mark in March 1998 (Snik & Cremers, 1999, 2001; Sterkers et al., 2003). The company went out of business in 2002 but returned in March 2003 as the Med-EL Vibrant Soundbridge. The Med-EL Vibrant Soundbridge is a semi-implantable device that uses an electromagnetic design referred to as the "floating mass transducer" (FMT).
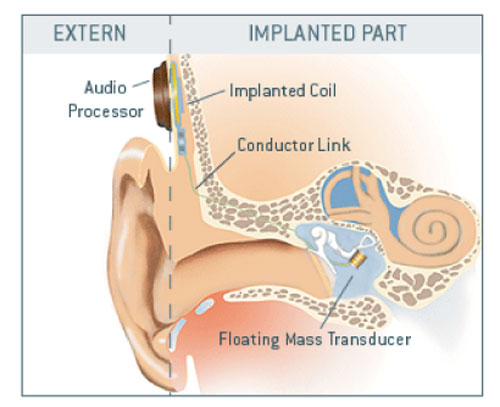
The FMT is crimped around the long process of the incus by a titanium strap. Vibration of the device is transmitted to the ossicles, thereby delivering stimuli to the middle and, subsequently, inner ear. In the Symphonix Phase II FDA study, 81 patients were followed for at least nine months post-surgery, and performance with the Vibrant Soundbridge and the patient's personal hearing aid were compared. Results indicated that with the implant, the patients experienced improved sound clarity and overall sound quality, better overall fit and comfort, reduced feedback, comparable or increased functional gain, improved perceived benefit in many listening situations, and reduced cerumen and moisture. The results also showed that patients could understand speech approximately the same with the implant as with conventional hearing aids.
Luetje et al. (2002) evaluated 53 adults with moderate to severe sensorineural hearing loss using the Vibrant Soundbridge system and found that aided speech recognition was again comparable to a conventional hearing aid. Additionally, there were significant improvements in patient satisfaction and sound quality, and occlusion and feedback problems were reported to be virtually eliminated (Snik & Cremers, 1999; Sterkers et al., 2003). In the past few years, surgeons have performed approximately 1000 implantations in the U.S. and Europe under the Med-EL Vibrant Soundbridge or Symphonix brand names.
Another technique, pioneered by Yanagihara (1984), incorporated an implantable piezoelectric device attached to the head of the stapes to induce a signal into the hearing system. Yanagihara and his colleagues in Japan have accomplished the earliest human trials using a piezoelectric type device (Yanagihara et al., 1987, 2001; Suzuki et al., 1987; Gyo et al., 1990). Their device is intended for patients with conductive and sensorineural loss.
Collaborating with the Rion company, Yanagihara has performed clinical trials with a semi-implantable piezoelectric device on more than 80 patients with over 10 years of follow-up data (Suzuki et al., 1994; Yanagihara et al.,1995).
St. Croix Medical, Inc. (now Envoy Medical Corporation) has participated in extensive research over the past ten years on a semi-implantable hearing device and has now developed a totally implantable piezoelectric device known as the Envoy System (Kroll et al., 2002).
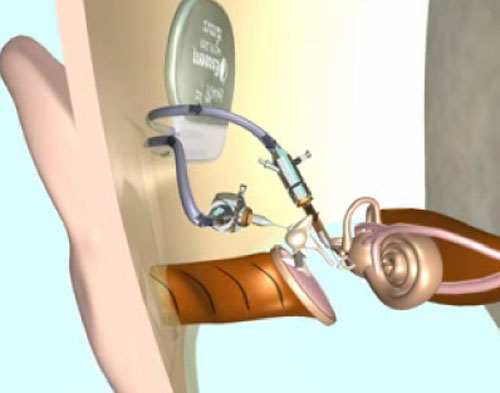
The drawback of the Envoy system is that it requires removal of the incus bone. The piezoelectric sensor and driver become the link between the malleus and stapes. Envoy Medical Corporation claims that it is possible to surgically restore the ossicular function of the middle ear by reattachment of the incus bone with a prosthesis. Operationally, the Envoy implant incorporates the eardrum as the microphone, and the malleus detects the mechanical signal. The piezoelectric transducer converts the mechanical signal to electrical, filters and amplifies, and transmits the vibratory signal via the stapes to the cochlea. The company is now actively enrolling patients to participate in further clinical trials in both the United States and Germany. Their results are referenced in numerous papers and lectures and have generally demonstrated positive patient satisfaction. The device does, however, have limited output capabilities that restrict its implantation to patients with mild-to-moderate hearing loss.
Recent studies by Snik et al. (2006) demonstrated the cost effectiveness of implantable hearing devices. They indicated that before the hearing impaired will utilize amplification in record numbers, these devices will need to be completely unseen, hiding the stigma generated by microphones, amplifiers, batteries, and the stimulator. In the past couple of years, totally implantable middle ear instruments have become available (Envoy and Otologics) that totally disguise the use of amplification and allow the user freedom from moisture problems, earmolds, and other practical difficulties of using amplification.
Development of the MET Transducer
Although some otologic researchers felt that these early electromagnetic and peizoelectric techniques would offer an effective and efficient middle ear implant, John Frederickson, then Professor and Head of Otolaryngology at Washington University, St Louis, Missouri, expressed concern that these early methods lacked bandwidth and acoustic output, reducing their practical application. With funding from Washington University and Storz Instruments, Frederickson and colleagues developed and refined the electromechanical motorized transducer (Figure 6) and continued experiments with various transducer placement sites within the middle ear. They evaluated placement of an electromechanical, motorized transducer (now called the Middle Ear Transducer [MET]) projecting into a laser-drilled hole in the incus of rhesus monkeys in 1995.
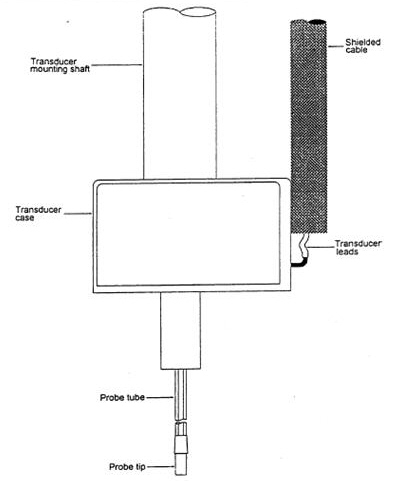
At this time, Fredrickson, Coticchia and Khosla demonstrated that there could be a safe, efficient method of transmitting sound energy to the ossicular chain. The benefit of the MET was demonstrated by pre/post acoustic and mechanical (bone conducted) auditory brainstem responses (ABRs) conducted on the rhesus monkeys implanted with the MET device. In evaluating the implanted monkeys, Fredrickson et al (1995) found no significant pre/post changes in latency/intensity functions, suggesting that the implantation of the middle ear transducer did not cause detectable conductive or sensorineural hearing loss. Further, they also demonstrated the fidelity of the implanted device by detection of distortion product otoacoustic emissions (DPOAE) generated through the implanted device.
Development of the Otologics Fully Implantable Hearing System
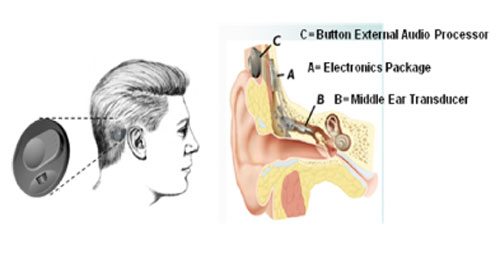
In 1996, Washington University and John Frederickson sold the MET technology to Otologics, LLC, and the company moved to Boulder, Colorado to continue development. By 1998, the new company had a semi-implantable product to offer for FDA investigation, and the MET semi-implantable instrument was on sale in Europe by 2000. As with most middle ear implants (Vibrant Soundbridge (formerly Symphonix), the Envoy, Sound Tec, and others) the Otologics MET was first offered in clinical trial both in Europe and the United States in a semi-implantable format. Although conceptually similar, semi-implantable instruments have an outside processor and an internal stimulator using one of the three technologies. Figure 7 presents the Otologics MET semi-implantable middle ear device where a button processor connects magnetically to an internal stimulator.
The FDA clinical trial process in the United States would have had significant costs; therefore, Otologics chose to not offer their semi-implantable device to the US market and concentrated totally on the research and development of the first fully implantable hearing device. The Otologics Fully Implantable MET received European CE-mark in October 2006 and is marketed as the Carina in Europe.
After an intensive research and development process in the Boulder facility, the Carina now incorporates a microphone, a speech processor, battery, and stimulating transducer into a prosthesis that can be totally placed under the skin behind the ear, avoiding most fitting and cosmetic issues. The device offers the same freedom and comfort of the natural auditory system and individuals can use it in environments not suitable for conventional hearing aids such as showering, swimming, and sporting activities.
The Current Device
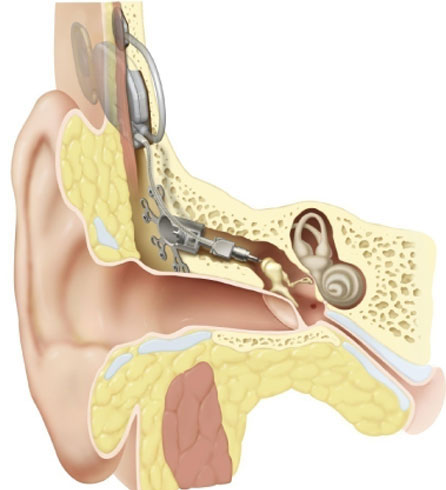
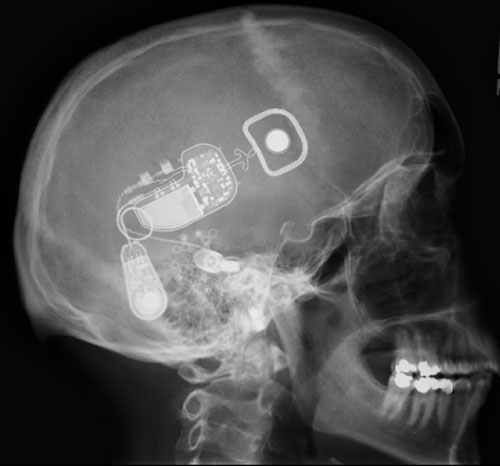
The Otologics MET Fully Implantable Ossicular Stimulator consists of three primary components: the implant (Figure 9A), the remote control (Figure 9B) and the charger (Figure 9 C/D).

The implant component of the MET Fully Implantable Ossicular Stimulator System acts in conjunction with a charger and remote control and is shown in detail in Figure 10. It consists of a signal processing electronic capsule, a pendant microphone system, the internal battery, and an electromechanical transducer. Specifically, the electronics package consists of two digital signal processors, control circuitry, a battery, radio frequency coil, and a magnet.
The MET transducer is a highly modified version of the original transducer first investigated by Fredrickson et al. (1995). Basically, the operation of the Otologics MET Fully Implantable Device is rather simple in that sound is picked up through the skin by an extra sensitive pendant microphone.
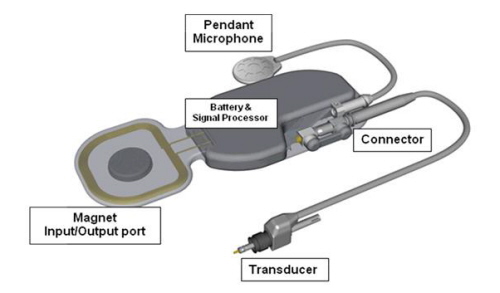
This is converted into an electrical signal, digitally processed according to the patient's hearing requirements, and conducted down a lead and into the transducer that is mounted in a laser-drilled hole in the body of the incus. The transducer translates the electrical signals into a mechanical motion that directly stimulates the ossicles and enables the wearer to perceive sound.
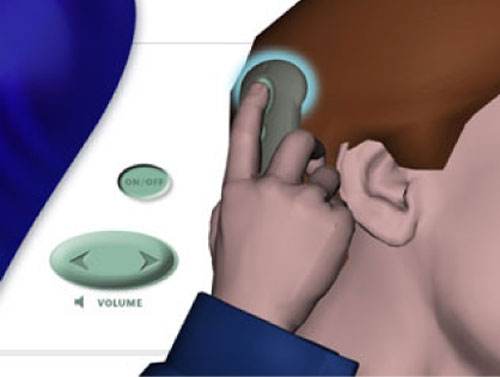
A remote control allows the wearer to turn the implant on and off and to adjust the volume. To use the remote control, the wearer simply holds the remote against the skin over the implant magnet (Figure 11).
The charger system consists of the base station, charging coil, and charger body (Figure 12).

Typically, the battery charger is plugged into an outlet and the battery fits inside the charger. Once charged, the battery may be used to charge the implant. The charging coil is placed over the magnetic attachment at the implant site; the charging time is typically one hour if the process is performed daily. The battery has a clip (Figure 13) that allows the charger to be attached to a belt or waistband of the wearer during charging, and the user can hear with the implant, turn it on and off, and adjust the volume during charging.

The Otologics Programming System
The programming system consists of fitting and diagnostic software; a radio frequency coil that, when placed over the implant site, magnetically adheres to the side of the wearer's head; and the NOAHlink™ programming interface, which is worn around the neck (Figure 14). Using the OtoFit™ Fitting Software, the NOAHlink interface receives signals from the computer through the wireless connection and sends the signals to the implant via the radio frequency coil.

Programming the implant is the same as programming traditional digital hearing aids. In addition, the Otologics Programming System provides the ability for extensive testing and diagnostics of the MET Fully-Implantable Ossicular Stimulator.
Surgery Overview: Sensorineural Implantation
The implant surgery is a relatively simple two hour procedure and not difficult in practiced hands. The incision is presented in Figure 15 and is similar to that for cochlear implants or other prosthetic devices.
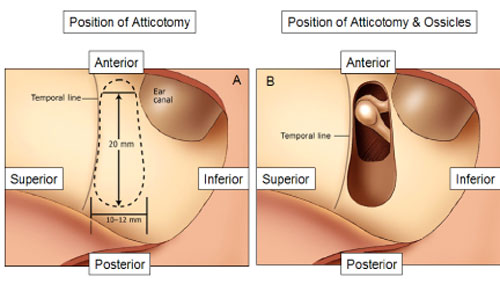
Once the incision is made, the surgeon drills an extended atticotomy between the temporal line and the ear canal, exposing the incus body and the head of the malleus (Figure 16A/B).
A mounting bracket is secured with bone screws and a laser guide is lined up with the body of the incus for the drilling of a .75 mm hole with a laser.
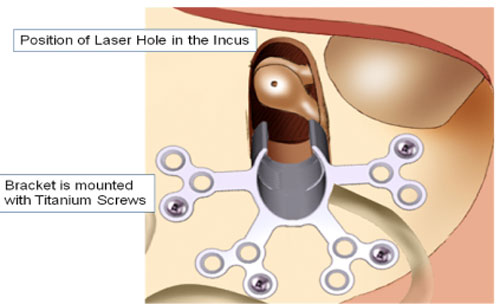
Once the surgeon drills the hole in the incus, the laser guide is removed. The transducer is carefully mounted with the aid of computer and software tools that allow the surgeon to know how much to load the transducer upon the ossicular chain.
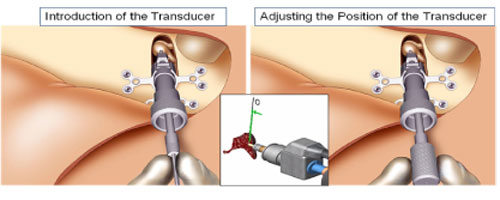
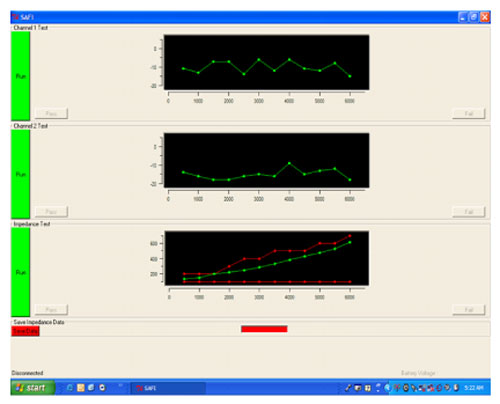
This is the delicate component of the implantation process requiring the use of a special tool called the Transducer Loading Assistant (TLA), which is shown in Figure 19. Use of the TLA insures proper loading of the transducer probe tip on the incus, thus providing excessive pressure (overloading) or insufficient pressure (underloading). Once the transducer is loaded onto the incus, the electronics package of the implant is tested using the diagnostics software, the Surgical Assistant FIMOS Implant (SAFI) that runs through the TLA (Figure 20).
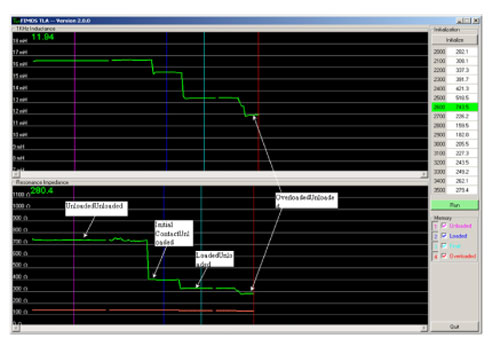
This provides real-time information to verify microphone input and internal electronics function, in addition to the correct loading of the transducer. Once tested, a template is used to drill a bone bed for the electronics package and the microphone.
The bone bed allows the electronics package to fit flush against the skull, maximizing the cosmetics of the implantation process. With the electronics package in place, the surgeon drills a bed for the microphone just posterior to the ear canal and thins the skin that will go over the microphone to 6 mm.
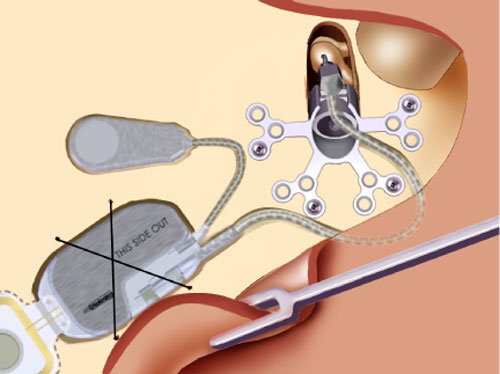
The thinning of the flap insures the proper skin thickness to allow the extra-sensitive microphone to pick up sound through the skin. Once all of the components are in position (Figure 22), the implant and microphone are secured with titanium straps, and the incision is closed with care to insure that the leads are not damaged during the process. The device is activated with the Otofit Software about 8 weeks after the surgery to allow for healing.
The MET-V Application for Conductive or Mixed Hearing Loss
Another application of the Otologics Fully Implantable MET device is now available for sale in Europe for conductive and mixed hearing loss. It is a variant of the MET, designated the MET V (Figure 23).
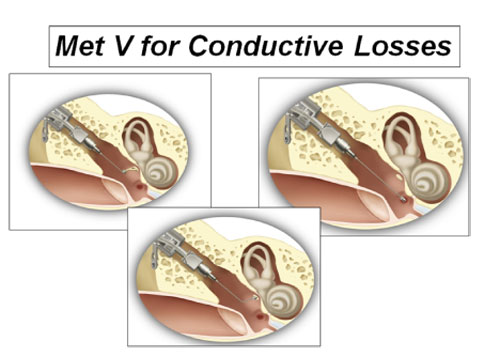
In theMET V, any one of several ossicular prosthetic attachments may be affixed to the vibrating transducer element in place of the ceramic tip. This modification allows the transducer to drive an incomplete, injured, or abnormal ossicular chain, as may result from middle ear disease, acoustic trauma, or congenital malformation. The attachments were designed for effective coupling to the long process of the incus or stapes capitulum (if present), to the stapes footplate when the stapes superstructure is absent, to the round window as in cases of stapes fixation, or to the oval window.
The ossicular prosthetic attachments used with the MET V are familiar to otologic surgeons and similar to prosthetics used in reconstructive surgery for decades. The incorporation of this family of attachments into the MET does not significantly change its function, but does greatly expand the options available to a surgeon treating cases of conductive and mixed loss from a broad variety of causes.
It is expected that the long-term stability of the ossicular attachment will be similar to outcomes observed for ossicular prostheses. However, one important difference from cases requiring total ossicular replacement prostheses is that total ossicular replacement requires interface with the tympanic membrane. This is the single most common cause of failure in these procedures, and the MET V requires no such interface.
Placing the partial ossicular replacement prosthesis (PORP) on the stapes at the incudo-stapedial joint requires that the surgeon implant the transducer through a mastoidectomy and facial recess. The facial recess is the most common surgical approach employed in cochlear implant surgery as well as for some chronic cholesteatoma conditions. The facial recess approach is also used when implanting some middle ear implant devices.
Candidacy for the Otologics Fully Implantable MET and MET-V Devices
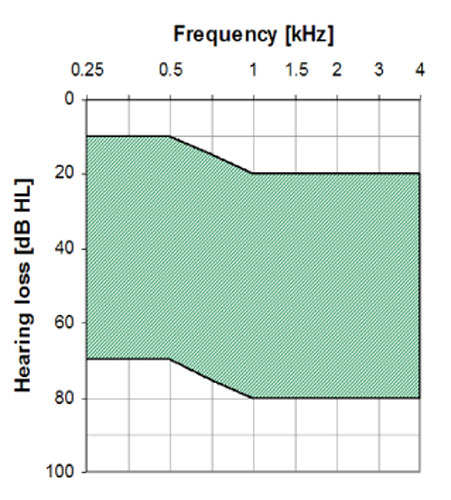
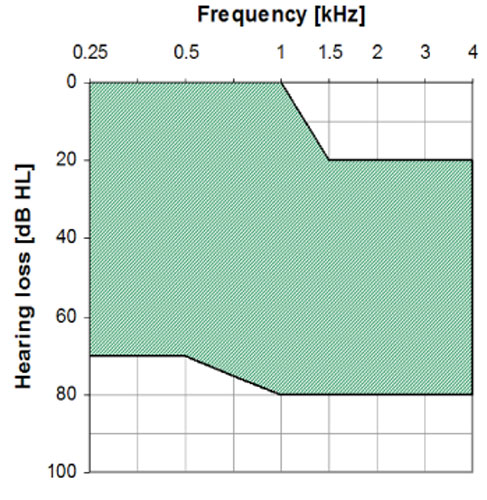
The MET Ossicular Stimulator is intended to compensate for auditory deficits in adults with a moderate to severe sensorineural hearing loss. The MET V Ossicular Stimulator is intended to compensate for mild to severe conductive or mixed type hearing loss due to congenital aural atresia or ossicular abnormalities. See Figures 24 and 25
Summary
This presentation discussed middle ear implants with specific emphasis on the Otologics Fully Implantable MET and MET-V devices. Although available in Europe, the Otologics Fully Implantable MET device is limited by Federal (US) law to investigational use. The MET V is currently not being investigated in the US. If patients are interested in obtaining information about involvement in the clinical trial for the Fully Implantable MET device in the US, they should go to www.otologics.com or call 800-390-5506 for further information.
References
Chasin, M. (1997). Current trends in implantable hearing aids. Trends in Amplification, 2(3), 84-107.
Chasin, M. (1998). Implantable hearing aids. Hear Rev, 5(2), 20-27.
Chasin, M. (2002). Bone anchored and middle ear implant hearing aids. Trends in Amplification, 6(2), 33-38.
Fredrickson, J., Tomlinson, D., David, E., & Odvist, M. (1973). Evaluation of an electromagnetic implantable hearing aid, Canadian Journal of Otolaryngology, 2, 53-62.
Fredrickson, J., Cotcchia, J., & Kohosla, S. (1995). Investigations into an implantable electromagnetic hearing device for moderate to severe sensorineural loss. The Otolargologic Clinics of North America, 28(1), 107-120.
George, L. (2006). Report of the Medical Technology Assessment Working Group. Duke University, Durham, NC.
Goode, R.L. (1970). An implantable hearing aid. State of the art. Transactions of the American Academy of Ophthalmology and Otolaryngology, 74(1),128-139.
Goode, R.L. (1988). Electromagnetic implantable hearing aids. In: Suzuki, J.-I., ed. Advances in Audiology. Basel: Karger, 44-31.
Gyo, K., Yanagihara, N., Saiki, T., & Hinohira, Y. (1990). Present status and outlook of the implantable hearing aid. American Journal of Otology, 11(4), 250-253.
Hough, J., Dyer, R., Matthews, P., & Wood, M. (2001). Early clinical results: SOUNDTEC Implantable Hearing Device phase II study. Laryngoscope, 111, 1-8.
Kroll, K., Grant, I.L., & Javel, E. (2002) The Envoy Totally Implantable Hearing System, St. Croix Medical. Trends in Amplification, 6(2), 73-80.
Luetje, C.M., Brackman, D., Balkany, T.J., Maw, J., Baker, R.S., Kelsall, D., et al., (2002). Phase III clinical trial results with the Vibrant Soundbridge implantable middle ear hearing device: A prospective controlled multicenter study. Otolarygology Head and Neck Surgery, 126(2), 97-107.
Matthews, P., Wood, M.W., & Dyer Jr., R.K. (2002). Middle ear electromagnetic semi-implantable hearing device: Results of the phase II SoundTec Direct System clinical trial. Otology Neurotology 23(6), 895-903.
Rutschmann. (1959). Magnetic audition: Auditory stimulation by means of alternating magnetic fields acting on a permanent magnet fixed to the eardrum. IRE Transactions Med Electron, 6, 22-23.
Snik FM, & Cremers WRJ. (1999). First audiometric results with the Vibrant Soundbridge, A semi-implantable hearing device for sensorineural hearing loss. Audiology, 38, 335-8.
Snik, F.M. & Cremers, W.R.J. (2001). Vibrant semi-implantable hearing device with digital sound processing: Effective gain and speech perception. Arch Otolaryngol Head Neck Surg, 127, 1433-1437.
Snik, F.M., Moulder, J., Cremers, C., & Noten, J., (2006). Middle ear implants; patients´ satisfaction, 9th International Conference on Cochlear Implants, Vienna Austria, June 2006.
Sterkers O, Boucarra D, & Labassi S. (2003). A middle-ear implant, the Symphonix Vibrant Soundbridge; Retrospective study of the first 125 patients implanted in France. Otol Neurotol, 24, 427-36.
Strom, K., (2005). HR Interviews...Sergi Kochkin, Hearing Review, 12(10), Retrieved from the World Wide Web September 30, 2007.
Suzuki, J.-I, Yanagihara, N., & Kadera, K. (1987). The partially implantable middle ear implant, case reports. Annals of Otology, Rhinology, and Laryngology, 37,178-184.
Suzuki, J., Kodera, K., Nagai, K., & Yabe, T. (1994). Long-term clinical results of the partially implantable piezoelectric middle ear implant. Ear Nose Throat Journal, 73(2),104-107.
Wilska, A. (1935). Einmethode zur bestimmung der horsch wellanamplituden des trommelfells bei verscheiden frequenzen. Skandinavisches Archives of Physiology, 72,161-165.
Yanagihara, N., Suzuki, J., Gyo, K., Syono, H., & Ikeda, H. (1984). Development of an implantable hearing aid using a piezoelectric vibrator of bimorph design: state of the art. Otolaryngology Head and Neck Surgery, 92(6), 706-712.
Yanagihara N, Aritomo H, Yamanaka E, & Gyo K. (1987). Implantable hearing aid: Report of the first human applications. Archives of Otolaryngology: Head and Neck Surgery, 113(8), 869-872.
Yanagihara, N., Gyo, K., & Hinohira, Y. (1995). Partially implantable hearing aid using piezoelectric ceramic ossicular vibrator. Results of the implant operation and assessment of the hearing afforded by the device. Otoloryngology Clinics of North America, 28(1), 85-97.
Yanagihara, N., Sato, H., Hinohira, Y., Gyo, K., & Hori, K. (2001). Long-term results using a piezoelectric semi-implantable middle ear hearing device: The Rion device E-type. Otolaryngol Clin North Am, 34(2), 389-400.

