"70s Night" is popular in the United States. Just about every major sports team playing more than 25 games a season devotes one night per year to all the novelties of the 1970s. Wide collars, long hair, platform shoes, and disco are the staples of these popular events. While we certainly salute the creative burst of the 1970s (think of all the great films and music from that era), from an audiology perspective there is not much that we still use today in the clinic. Békésy audiometry, the SISI, and comparative hearing aid evaluations have all fallen by the wayside. The aim of this paper is not to bring back the '70s, but to showcase how some of the audiological tests from that era can be useful tools for validating outcomes of modern hearing aids.
A famous scientist from the '70s (the 1870s, that is) once said, "When you measure something, you automatically improve it." When it comes to fitting hearing aids, this statement has profound consequences. Taking the time to systematically measure outcome that is meaningful to patients should almost, by definition, improve outcomes. A good example of this is from the MarkeTrak patient surveys. Kochkin has noted that when dispensing professionals take the time to merely ask about patient satisfaction, it improves (2007). One goal of this report is to shed light on the possible ways outcome can be measured in a busy clinic.
Success with amplification, as many know, can be assessed using both self-reports, also called questionnaires, and laboratory measures. This report will focus on the laboratory measures of outcome available to calculate the various dimensions of hearing aid performance and benefit. Laboratory measures of outcome can best be described as a measurement of hearing aid performance or benefit, using either a subjective or objective test conducted in a contrived listening environment, like the test suite or sound booth.
Subjective measures of laboratory outcome involve asking the patient to give a judgment on a well-defined task or questionnaire. Objective measures, on the other hand, require the patient to complete a task in which a score, usually defined as a percent-correct, is obtained. When real-world and laboratory measures of outcome are combined, using both subjective and objective tests, the audiologist is able to obtain a more comprehensive and robust assessment of their intervention with amplification.
Table 1 is a summary of the various types of outcome measures with examples of each. According to survey data, 20% of audiologists do not use any type of outcome measurement to assess the success of hearing aids. Of the 20% who systematically measure outcomes, very few are likely to rely on a comprehensive battery of outcome tools to robustly assess results. Although the use of multiple outcome measures may seem time consuming, patient surveys indicate that their use improves overall satisfaction.
With a nearly 25% return and exchange rate, coupled with the fact that one-in-six hearing aids ends up in the drawer (Kochkin, 2007), professionals need to rethink how outcomes can be used to improve overall patient satisfaction. This paper is geared toward helping the busy clinician identify practical ways to measure hearing aid outcome in the laboratory.
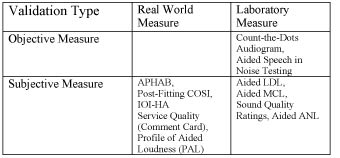
Table 1. Examples of various types of outcome measures in comprehensive battery of tests.
Before reviewing lab measures of outcome, it is important to discuss the differences between verification and validation. Verification techniques are used to confirm that the gain and output of the hearing aid is matching a prescriptive fitting target. Verification techniques, like probe microphone analysis, are really quality control measures, enabling the audiologist to know that the hearing aid is approximating a prescriptive fitting target. Although a proven starting point, verification techniques do not evaluate whether the hearing aids with the matched prescriptive target are benefiting the patient in everyday listening situations.
Hearing aid validation, on the other hand, refers to measuring how the hearing aids are benefiting the patient in real-world situations. Over the last few years, the emergence of evidence-based practice has emphasized the importance of measuring benefits in the real world. Because the laboratory is only an approximation of the real world, self-reports or questionnaires have become the favored manner in which to measure hearing aid outcome. In fact, self-reports of outcome have become the gold standard for measuring real-world benefit with hearing aids, and should be conducted systematically with every hearing aid fitting. For a review of self-reports of hearing aid outcomes, see Taylor (2007).
Even though self-reports of outcome are the gold standard for measuring success with hearing aids, there are several enduring reasons for conducting laboratory measures to assess hearing aid benefit. The legendary Raymond Carhart (1970), nearly 40 years ago, mentioned two reasons for assessing hearing aid outcome in the laboratory. Both are still valid today. The first reason is to measure everyday efficiency of communication with hearing aids. Since results on the pure-tone audiogram and unaided speech audiometry are not reflective of how patients communicate in the real world, aided laboratory tests have the potential to accurately and quickly evaluate outcomes. The second reason is to assess the practical gain (and today, many advanced features) the patient is receiving when wearing hearing aids.
Even though prescriptive fitting targets, verified with probe microphone measures, have supplanted traditional functional gain measures collected in the sound field, laboratory measures can show the patient in a sort of snapshot manner how they are doing with amplification. As patients become more savvy and well-informed about their treatment options, conducting laboratory measures is a proven strategy for building trust with patients. Seven tests that can be used as laboratory measures of outcome are outlined below:
1. Lab Measure of Audibility: Count-the-Dots Audiogram
Conducting functional gain or aided thresholds in the test suite and charting the scores on the Count-the-Dots audiogram is a handy way to show the patient how WDRC might be affecting speech understanding. The Count-the-Dots audiogram (Mueller & Killion, 1990) can be useful for demonstrating to patients if audibility for quiet speech sounds has been achieved. Although maximizing audibility does not always equate to an improvement in speech intelligibility, it is a well established fact that speech intelligibility is virtually impossible without sufficient audibility of speech.
Here's how the procedure works:
Step 1: Place the patient in the calibrated sound field, 1 meter from the speaker at 0 degrees azimuth.
Step 2: Using a blank Count-the-Dots audiogram, conduct sound field threshold testing in the unaided condition. Chart the results on the form.
Step 3: Conduct the threshold test in the aided condition and chart on the same form.
Step 4: Compare the unaided to the aided scores and discuss the results. Your conversation with the patient should focus on the improvement in audibility with hearing aids, and how it relates to intelligibility with the patient.
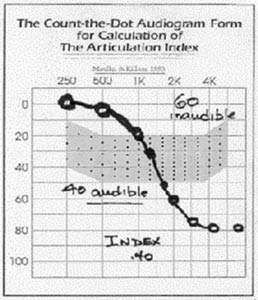
Figure 1. Example of the Count-the-Dots Audiogram (Mueller & Killion, 1990).
2. Lab Measure of Audibility and Intelligibility: Speech in Noise Testing
Since the Count-the-Dots audiogram lacks face validity, other lab procedures can be used to validate aided audibility. Speech-in-noise tests can be used to demonstrate the effects of improved audibility with hearing aids. The QuickSIN works very well for this procedure.
When the QuickSIN is conducted at a low intensity level, it provides the patient with meaningful information on how improved audibility usually translates into improved speech intelligibility in noise. To conduct this test, do the following:
Step 1: The patient is placed 1 meter from a single speaker at 0 degrees azimuth. Using the QuickSIN, unaided scores are obtained and charted.
Step 2: The aided condition sentences are presented at a typical level of soft conversation (i.e., 50 dB HL) as the noise level is increasing using an adaptive procedure of the QuickSIN.
Step 3: Relate the results of this test to real-world listening conditions that the patient experiences. One example of this procedure is shown in Figure 2. Note that the poor scores in the unaided condition are attributed to inaudibility, and when the hearing aid restores soft speech, the scores improve dramatically. Conducting speech-in-noise tests at low intensity levels is an effective way to demonstrate to patients the effect audibility has on intelligibility in noise.
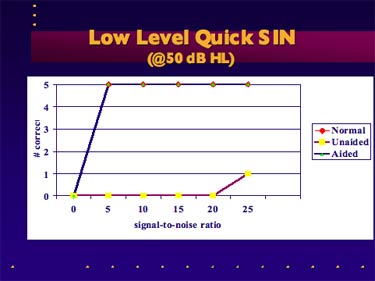
Figure 2. Example of QuickSIN as a lab measure of aided audibility for soft speech in noise for a patient with moderate, flat hearing loss.
3. Lab measure of comfort: Aided MCL
Lab measures can be used to quickly confirm that average levels of speech are comfortable. One way to demonstrate to patients that average levels of speech are comfortable when wearing their hearing aids is to conduct an aided sound field most comfortable loudness level (MCL). This procedure was outlined by Pascoe (1988) and incorporates the long-term average speech spectrum into measures of aided loudness. Given their precision, warble tones for this procedure are preferred over the use of live voice.
A long-term average speech spectrum has the following approximate intensity level (dB HL) in the sound field:

Table 2. Average speech spectrum intensity level in dB HL at discrete frequencies (Pascoe, 1988).
Using the dB HL levels for average levels of speech in Table 2, aided MCL measures can be conducted in the following manner:
Step 1: Place the patient in the sound field 1 meter from the speaker with hearing aids turned on.
Step 2: Using the IHAFF loudness chart (Figure 3 below), instruct the patient to rate the loudness of warble tones presented at the intensity levels shown in Table 2.
Step 3: Use an ascending procedure to deliver the warble tones. When the patient gives you a consistent response for comfort, record the result on the audiogram.
Since we are checking to ensure that average intensity levels for speech are comfortable in aided conditions, the patient should judge the sounds to be comfortable (#4 on the IHAFF chart). If the patient does not rate these sounds as comfortable, the audiologist should consider tweaking the gain-for-average until a comfort level (#4) is achieved. Although this test is an approximation, it can serve as an effective way to demonstrate in the lab that average speech is indeed comfortable in the aided condition.
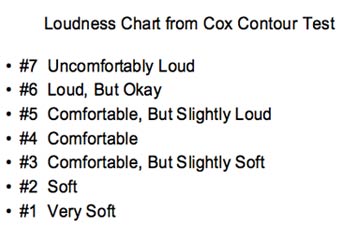
Figure 3. IHAFF Loudness Chart.
4. Lab Measure of Sound Quality: Aided Sound Quality Judgments
When it comes to fitting and assessing hearing aids, the term "good sound quality" is rather vague. We know, for example, that aided speech intelligibility can be excellent, and patients still struggle with issues related to sound quality, especially when listening to music. Nielson (1992) defined aided sound quality as "attributes in the auditory perception that describe naturalness and timbre. Speech intelligibility is not part of sound quality."
There are a number of ways that sound quality can be evaluated in the clinic. The lab method described here was originally reported by Gabrielson and Sjogren (1979) and has been demonstrated to be a valid procedure for making sound quality judgments. It requires the patient to listen to a recorded conversational speech passage or music at a comfortable listening level and favorable signal-to-noise ratio of +10 SNR.
The following sound quality rating procedure can be used to validate sound quality of speech or music. This lab measure should only be completed once audibility for quiet and average levels of speech have been verified. In other words, subjective lab measures of sound quality, like the one described below, can be used to tweak the hearing aid fitting in order to maximize sound quality for either conversational speech or music.
Step 1: Patient is placed in the sound field 1 meter from the speaker while wearing their hearing aids in the 'on' position.
Step 2: A one-to-two minute passage of either conversational speech or music is played through the speaker. Using the IHAFF loudness scale (Figure 3) and an ascending procedure, a "comfortable" (#4) level is ascertained.
Step 3: The patient is asked to rate the following dimensions of sound quality on a 0 (very poor) to 100 (very good) scale: clearness, sharpness, fullness, and overall impression. The scale shown in Figure 4 below can be given to the patient as a guide for making their ratings for the four dimensions.
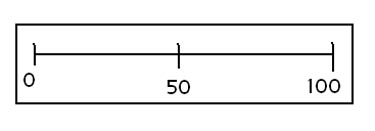
Figure 4. Sound quality rating scale for one of the four dimensions of sound quality.
Step 4: Once the patient has been instructed to provide their rating for those four dimensions, the patient listens to the passage for approximately one minute, until he can make a subjective rating. The patient then uses the 0 to 100 scale (Figure 4) to make their rating.
Step 5: For ratings less than 70, further adjustment of the acoustic parameters of the hearing aid may be needed. Given the subjective nature of this procedure, the audiologist must carefully discuss ratings with the individual. For patients with good audibility of speech as documented on probe microphone measures and the Count-the-Dots audiogram, counseling may be needed to address expectations.
When sound quality ratings in the aided condition are compared to the unaided ratings using the same passages, a reasonably good indication of the impact amplification has on sound quality can be demonstrated to the patient. Because sound quality and speech intelligibility are thought to be two separate dimensions of hearing aid performance, taking the time to conduct sound quality ratings may help to systematically measure outcomes. For this reason, they should be used as part of a comprehensive hearing aid validation protocol.
5. Lab Measure of Directional Microphone Benefit: Speech-in-Noise Testing
Speech-in-noise tests can also be used to assess directional microphone benefit in the lab. When using speech-in-noise testing for evaluating directional microphones, the speech and noise need to be delivered via separate channels to two different speakers. Because speech and noise are recorded on separate tracks, either the HINT or QuickSIN can be used for this lab test.
In order to simulate real-world listening conditions, the speaker delivering speech to the patient should be placed 1 meter from the patient at 0 degrees azimuth. The speaker delivering the noise should be placed directly overhead, 1 meter above the patient. When using the QuickSIN, speech should be delivered at 65 dB HL;the noise automatically varies in 5 dB steps, starting at a signal-to-noise ratio of +25 dB. To obtain these measurements, follow these steps:
Step 1: With the hearing aids in the omnidirectional mode, a score is obtained on the QuickSIN at six different signal-to-noise ratios.
Step 2: With the hearing aids switched to the directional mode, a second score on the QuickSIN is obtained. The difference in scores is the directional benefit.
Step 3: Results can be compared to self reports of benefit and satisfaction in noise and can be discussed with the patient. Figure 5 is an example how the two scores are be charted. Because of the manner in which the speech and noise are delivered in the sound field, this lab test is a good estimation of the amount of directional benefit the patient is likely to receive in a typical noisy listening situation.
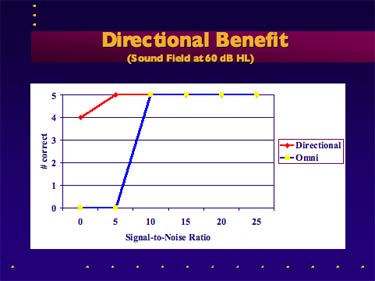
Figure 5. An example of directional benefit on the QuickSIN in the sound field
6. Lab measure of Discomfort: Aided LDL
Although unaided measures of loudness discomfort levels (LDL) have been equated with aided benefit (Mueller & Bentler, 2005), aided LDLs can be a useful tool for identifying discomfort associated with high hearing aid outputs. This is especially true during the initial adjustment period after the patient has been subjected to common loud environmental sounds. Additionally, binaural summation and channel summation are thought to affect measures of loudness discomfort with hearing aids. Therefore, aided LDLs may need to be evaluated in a systematic nature.
In order to complete aided loudness discomfort level testing, the audiologist needs a CD player and a speaker (stereo system or boom box), inexpensive sound level meter (SLM), and a CD with recordings of common noises. The procedure is conducted in the following manner:
Step 1: Before conducting the test, the audiologist needs to calibrate the CD through the stereo system. This is done by placing the SLM approximately 1 meter from the speaker and setting the volume of the stereo system so that the reading on the SLM reaches 85 dBA. The volume level on the boom box needs to be marked for future reference
Step 2: Place the patient 1 meter from the speaker while wearing the hearing aids. Using the IHAFF loudness chart (Figure 3), ask the patient to rate the loudness of various passages that have been calibrated to reach 85dBA on the SLM. (The patient can be instructed using the IHAFF LDL guidelines).
Step 3: Patient listens to the passage and rates the loudness level on the IHAFF scale. Patients should rate the passage to be a #5 or #6 on the chart. If the patient rates them as #7 or higher, the output needs to be adjusted on the hearing aids.
7. Laboratory Measure of Sound Annoyance: Aided ANL
The Acceptable Noise Level (ANL) test is used to measure annoyance from noise. When the test is conducted in the unaided condition, research has shown the ANL test to be a good predictor of hearing aid usage. The ANL can also be measured in the aided condition. When the aided ANL is compared to the unaided ANL score, the difference is a good lab measure of the effectiveness of digital noise reduction.
Step 1: Following the ANL test guidelines (Nabelek, et al., 2006), establish the unaided ANL score. Given the predictive nature of the unaided ANL, this test should be conducted during the pre-fitting evaluation.
Step 2: Using the same procedure, the aided ANL score is obtained.
Step 3: The difference in the unaided and aided ANL scores is the benefit. This benefit can be attributed to the Digital Noise Reduction (DNR) technology of the hearing aid. Mueller and colleagues (2006) have shown that certain types of DNR improve the aided ANL score by an average of 4 dB. This improved score in the aided condition is a good simulated demonstration for the patient showing the effectiveness of DNR technology in real-world listening conditions.
A comprehensive and practical outcome battery that drives patient satisfaction
Because personality, age, and other issues affect self-reports of hearing aid outcome (see Kricos, 2006), laboratory measures, like the ones outlined here, can serve as cross-checks when measuring overall hearing aid success. According to Humes (2004), there are three separate and unique dimensions of hearing aid outcome. Two of these dimensions include aided speech recognition benefit, and hearing aid usage & subjective benefit as measured on self-report scales. Each of these dimensions can be evaluated or cross-checked with the lab measures reviewed here.
Both research results and feedback from today's well-informed patient remind us that the hearing aid validation process can be improved from the combination of laboratory and real-world measures of outcome. Although self-reports of outcome will continue to be the gold standard in an evidence-based practice paradigm, objective measures of outcome in the lab certainly have their place. When both laboratory and self-report measures are combined, a more robust determination of hearing aid success is established. This leads to more confident audiologists who know their standard protocols are effective, and more importantly, produce more satisfied patients.
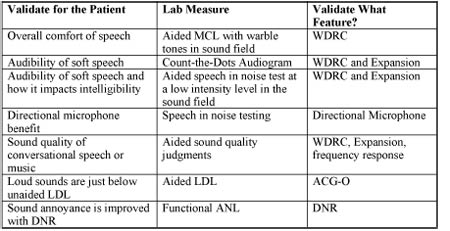
Table 3. Summary of lab measures used to validate various dimension of hearing aid performance and benefit, included is the advanced feature likely to contribute to lab results
The various measures of outcome and how they can be used to validate various advanced hearing aid features are summarized in Table 3. Lab measures of outcome should be used at any time to validate or demonstrate for the patient that advanced hearing aid features are working and that the hearing aids are performing in a satisfactory manner. The '70s might be long over, but as you can see, lab measures - many of which were born in that era - live on in the modern clinic. It is the responsibility of the audiologist to judiciously use the various laboratory measures reviewed here in order to comprehensively validate hearing aid success.
References
Carhart, R. (1970). Principle of competing message speech audiometry. 2nd Danavox Symposium.
Gabrielsson, A. & Sjogran, H. (1979). Perceived sound quality of hearing aids. Scandinavian Audiology, 8(3), 159-169.
Humes, L. (2004). As outcome measures proliferate, how do you choose which ones to use? The Hearing Journal, 57(4), 10-17.
Kochkin, S. (2007). MarkeTrak VII: obstacles to adult non-users adoption of hearing aids. The Hearing Journal, 60(4), 27- 43.
Kricos, P. (2006, January 16). Minimizing the effects of non-audiological variables on hearing aid outcomes. Audiology Online, Article 1527. Retrieved February 25, 2008, from the Articles Archive at Down www.audiologyonline.com/articles/
Nabelek, A., Freyaldenhoven, M., Tampas, J., Burchfield, S., & Meunchen, R. (2006). Acceptable noise level as a predictor of hearing aid use. Journal of the American Academy of Audiology,17(9), 626-639.
Nielson, L. (1992). Subjective evaluation of sound quality for normal-hearing and hearing-impaired listeners. 15th Danavox Workshop.
Mueller, G., & Bentler, R. (2005). Fitting hearing aids using clinical measures of loudness discomfort levels: an evidence-based review of effectiveness. Journal of the American Academy of Audiology, 16, 461-472.
Mueller, H.G., & Killion, M.C. (1990). An easy method for calculating the articulation index. The Hearing Journal, 43(9), 14-17.
Mueller, H.G., Weber, J., & Hornsby, B. (2006). The effects of digital noise reduction on the acceptance of background noise. Trends in Amplification, 10(2), 83-94.
Pascoe, D. (1988). Functional gain measurements. 13th Danavox Symposium.
Taylor, B. (2007, October 22). Self-Report Assessment of Hearing Aid Outcome - An Overview. Audiology Online, Article 1888. Retrieved February 25, 2008, from the Articles Archive at www.audiologyonline.com/articles/


