In the United States, an estimated 60,000 people per year acquire Single Sided Deafness (SSD), typically caused by viral infections, Meniere's disease, head or ear trauma, or through surgical intervention to remove acoustic neuromas or other brain tumor removal procedures.
For people with SSD, there have only been limited options regarding audiologic rehabilitative treatments, and often, those living with SSD have been left without a choice or adequate hearing solution. Generally speaking, previous solutions for SSD have been limited to contralateral routing of signal (CROS) hearing aids and aural rehabilitative strategies, such as preferred listening and seating positions, and noise reduction strategies.
CROS hearing aids are available as either "wired" or "wireless." In essence, CROS hearing aids consist of an in-the-ear (ITE) or behind-the-ear (BTE) hearing aid shell, containing a receiver. The receiver unit is placed on the normal ear with an ITE or BTE shell (or ear hook) containing a microphone system located on the deaf ear. The sound is "heard" through the microphone system located on the side of the deaf ear, and sent via a wired or wireless system to the normal ear. An alternative to the CROS system, for people with two ears that are in need of amplification, is the BI-CROS. With the BI-CROS system, both ears have a microphone system in their shells, and again, the sound is perceived only on the side with the receiver, i.e., the better hearing ear.
However, performance, handling, and aesthetics of CROS and Bi-CROS systems have been considered poor by many professionals who have dispensed them, and by patients who have worn them. Most people offered CROS hearing aids do not purchase them, and of those who do, apparently only a percentage of the owners use them full-time, a year or two later.
For those who have not experienced SSD, it is difficult to appreciate the handicaps presented and the lifestyle changes that occur in tandem with this type of hearing loss. Many patients with SSD have to make major life changes as they may feel highly uncomfortable and no longer easily able to cope in everyday environments such as business meetings, busy restaurants, family gatherings, or even bicycling or jogging. People living with SSD often withdraw from social and professional activities. In group situations, loud sounds become overwhelming and simple things become major obstacles. Many times, those with SSD remove themselves socially and professionally from important activities, due to their discomfort and disability.
 Alternative to CROS Hearing Aids:
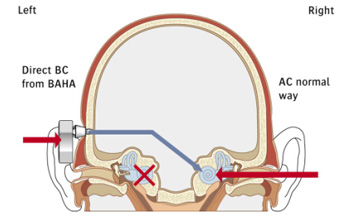
Alternative to CROS Hearing Aids:The Baha® System is an FDA-cleared solution for SSD. The Baha System provides a completely unique benefit. Consisting of a titanium implant placed in the mastoid bone behind the deaf ear, a connecting abutment, and a detachable sound processor, the Baha System transfers sound through bone conduction, stimulating the cochlea (the inner ear) of the normal hearing ear and allowing the wearer to hear and understand sounds originating from both sides.
Experiences from patients and professionals
Many patients and medical professionals have reported their positive experiences and results with Single Sided Deafness and the Baha System, and some of these are noted below. Please click on the interview (below) to view the entire text.
A chiropractor talks about his battle with a recurring acoustic neuroma removal and his experience with Single Sided Deafness and the BAHA system.
/interviews/interview-with-doug-metz-d-1648
A newspaper publisher shares her experience with the BAHA system and life after sudden hearing loss due to an acoustic neuroma.
https://www.healthyhearing.com/library/testimonial_content.asp?testimonial_id=141
A nurse left with SSD after a seizure talks about the difference the BAHA system has made in her life.
https://www.healthyhearing.com/library/testimonial_content.asp?testimonial_id=149
A teenager re-discovers the gift of hearing with the BAHA system after a head trauma left her with SSD.
https://www.healthyhearing.com/library/testimonial_content.asp?testimonial_id=163
Two surgeons speak about Bone Conduction Implants.
/interviews/interview-with-jack-j-wazen-1662
/interviews/interview-with-sujana-chandrasekhar-md-1655
An audiologist shares her perspective on SSD and the BAHA system.
/interviews/interview-with-donna-cody-audiologist-1636
The Baha System has been regarded as a successful treatment option in Europe since the late 1970s and was first FDA cleared in 1996 for use in the treatment of mixed and conductive hearing loss. In 1999, FDA indications were expanded and the Baha System was cleared for pediatric use in children ages five years and older. The system was cleared in 2001 for bilateral fittings and, in 2002, the Baha System was FDA cleared to treat Single Sided Deafness. Currently, the Baha System is worn by 25,000 people worldwide.
For more information about the BAHA system, click here.
