The vast majority of patients with hearing loss visiting a typical audiology private practice or hearing aid dispensing office can be helped with hearing aids. Conventional wisdom would suggest that very few, if any, cochlear implant candidates are seen in private practices today since severe and profound hearing losses comprise a small percentage of overall hearing loss. And if so, there is a variety of hearing aid technology - such as frequency compression and/or frequency transposition, directional microphones and FM compatibility - that might help these people. So, why should a dispensing professional care about cochlear implant candidacy or referral?
In the case of severe-to-profound hearing loss, professionals need to provide information on cochlear implants for those who might benefit. Cochlear implant candidacy has changed over the years, just as cochlear implant technology has developed and improved. For the appropriate candidates, cochlear implants may offer the chance at vast improvement in speech recognition over what even the best hearing aids and assistive technology offers them. Professionals have a responsibility to inform patients of all their options, and that means understanding who is a current candidate for a cochlear implant and understanding the potential benefits. In addition to informing patients of their treatment options because it is in the best interest of the patients, professionals also risk losing patients if they are not informed about cochlear implants. Consumers are becoming savvier in this information age in regard to researching health information. They are looking to their health professionals whether it is their physician, audiologist, or hearing care professional to provide current information and options regarding their treatment choices.
This article will describe the current cochlear implant candidacy criteria, and provide information for dispensing professionals on the potential benefits of cochlear implants. In addition to providing basic information on cochlear implant performance and issues surrounding the cochlear implant process, some current data indicating a lack of referrals for cochlear implant evaluations will be presented. In providing up to date information and dispelling common myths about this technology, this article makes a case for increased referrals for cochlear implant evaluations from dispensing professionals for their patients who may benefit.
Who Are Good Candidates?
To determine who is a candidate for a cochlear implant, first consider who is a hearing aid candidate. As a dispensing audiologist, there is information and resources available to help determine this, and to guide best practices for hearing aid fitting. The Joint Committee on Clinical Practice Algorithms (2000), the American Academy of Audiology Guidelines for the Audiological Management of Adult Hearing Impairment (2006), and textbooks such as Valente (2000) and Dillon (2001) are readily available, just to name a few. In addition to performing comprehensive evaluations including recorded speech measures, measured (not predicted) frequency-specific loudness discomfort levels (LDLs) (Mueller & Bentler, 1994), audiologists also have at their disposal measures of speech in noise with the QuickSIN (Killion, Niquette, Gudmundsen, Revit and Banerjee, S. 2004) and the COSI (Dillon, James, & Ginis, 1997) to help identify patient specific wants and needs.
Today's tools for hearing aid fitting include verification equipment using probe microphone measurements to ensure that soft sounds are audible and that 90 dB SPL inputs do not exceed LDLs. Frequency and gain of today's hearing instruments can be easily adjusted to soft, medium, and loud signal levels using NAL-NL1 (Dillon, 1999) targets. While the probe is in the ear and stimuli are presented, different hearing aid programs can be activated and measured. Steady state composite noise can be introduced to verify adaptive features such as noise suppression. Other features such as directional microphone technology is available to improve speech understanding in noise, and this can be easily verified in the audiology clinic. Finally, a variety of counseling and validation tools are available to help with orientation and fine-tuning.
Even with all these twenty-first century tools available, every audiologist has had the experience of having patients who cannot be sufficiently helped with hearing aids. No matter how experienced the audiologist or how much evidence-based science is applied to the hearing aid fitting, there are going to be some patients who are simply not satisfied. Others may feel the benefit from hearing aids does not justify the cost. This paper is about the people for whom hearing aids are just not enough.
Despite the available information and resources, there are no universally accepted criteria regarding who is a good hearing aid candidate or what defines a successful hearing aid fitting. Patient satisfaction may be related as much to clinician counseling and technical skill as it is to hearing aid fit. Candidacy is as much a function of lifestyle and motivation as it is of the audiogram. A 45 dB HL high-frequency hearing loss might not bother a retiree with a sedentary lifestyle but could be devastating to a practicing audiologist who cannot adequately hear her patients or a teacher who cannot hear his students.
There is a mind-boggling range of instruments for an infinite variety of hearing impairments. It can be challenging for dispensing professionals to keep up with the continuous introduction of new products and the constant bombardment of manufacturers' claims. However, it is the responsibility of audiologists and hearing instrument specialists to keep current on technology and patient options so that the best possible solutions can be provided to the patients.
Cochlear implants can be a solution for those who do not benefit or are no longer satisfied with the limited benefit they receive from hearing aids. Unlike hearing aids, the audiological candidacy criteria for cochlear implantation are well defined as approved by the FDA;it is based on the pure-tone audiogram and the patients' aided speech recognition scores. Of course, there are many other factors that go into the decision to receive an implant beyond these objective measures, including age at onset of the hearing loss, duration of deafness, etiology, motivation, family support, and access to rehabilitation. These factors should be discussed on an individual basis with an experienced cochlear implant team. For now, let us focus on identifying those patients we see who might be better served by a cochlear implant than hearing aids, and thus should be referred to a cochlear implant team for further evaluation.
Cochlear Implant Criteria
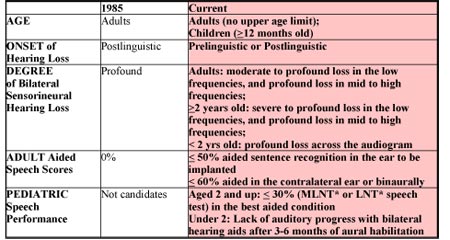
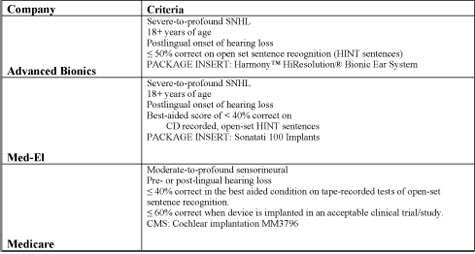
The criteria for cochlear implantation have changed dramatically over the years. The first multi-channel cochlear implant device was approved for use in the United States in 1985. At that time, only post-lingually deafened adults with bilateral profound sensorineural hearing loss and 0% aided speech recognition scores were considered candidates. Cochlear implants were not approved for children in the U.S. until 1990. Today, the criteria span the age range from infant to geriatric, and include those with lesser degrees of hearing loss and higher speech recognition scores. The current criteria for Cochlear™ Nucleus® cochlear implants as well as criteria used in 1985, are shown side by side in Table 1. Table 2 provides adult cochlear implant criteria based on sentence recognition performance (HINT sentences in quiet) for two other manufacturers and Medicare.
Click Here to View Larger Version of Table 1 (PDF)
Table 1. Criteria for CochlearTM Nucleus® cochlear implants, in 1985 and current. *(Multisyllabic Lexical Neighborhood Test;Lexical Neighborhood Test)
Click Here to View Larger Version of Table 2 (PDF)
Table 2. Adult cochlear implant criteria based on sentence recognition performance (HINT sentences in quiet) for two manufacturers and Medicare. Adapted from Gifford & Dorman, 2008.
The fact that the audiologic criteria for cochlear implant candidacy have expanded compared with those of the past is especially important information for professionals who dispense hearing aids, who may not be aware of the information. Adult patients can have only moderate hearing loss in the low frequencies, sloping to profound loss in the mid to high frequencies, and still be excellent candidates for a cochlear implant. It is no longer a requirement that one have "no useable hearing." Aided sentence recognition of 50% or less in the ear to be implanted and 60% or less in the contralateral ear or bilaterally ("best aided condition") is the current criterion for adults. Typically, cochlear implant centers will use recorded HINT sentences presented in quiet, typically at conversational level. Before a patient is considered a cochlear implant candidate, he or she must have been given an opportunity to use well-fit amplification.
In terms of age criteria for candidacy, the advantage of implanting an infant who is still in the critical language acquisition stage may seem obvious, but what about the 82-year-old who is socially isolated by a hearing loss? Older adults show no increase in operative time or length of stay in the hospital (Labadie, Carrasco, Gilmer, & Pillsbury, 2000). Speech perception in the implanted elderly is comparable to that of younger recipients (Kelsall, Shallop, & Burnelli, 1995;Vermeire, et al., 2005). Cochlear implants have a significant positive impact on the quality of life of older patients (Francis, Chee, Yeagle, Cheng and Niparko, 2002). There is no upper age limit for cochlear implant candidacy.
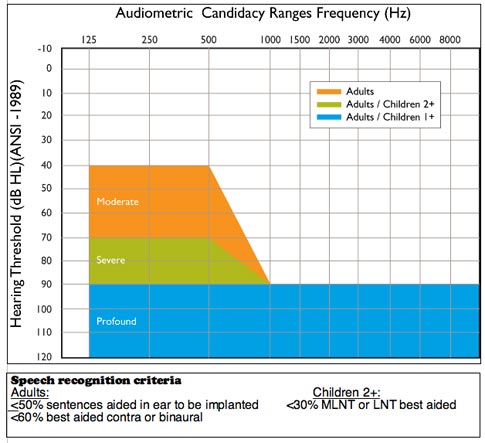
Figure 1. Audiogram range and speech recognition criteria by age group for Nucleus® cochlear implant candidates.
Cochlear Implant Performance, Surgery and Reimbursement
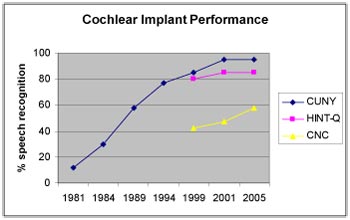
Performance with cochlear implants is much better today than it was 25 years ago. In the early 1980s, the best one could hope for was low double-digit open set sentence scores (Figure 2). Over the years, implant technology has advanced in the same way as hearing aid technology. Cochlear implant speech processors and coding strategies have improved, much like noise reduction and adaptive algorithms in hearing aids. Some cochlear implant processors have the capability of multiple programs and some have directional microphones to provide recipients with greater comfort in diverse listening environments. Signal processing strategies and speech coding enhancements now enable many implant users to achieve high open-set word recognition scores and the ability to talk on the telephone- achievements that would have been inconceivable only a few years ago.
Click Here to View Larger Version of Figure 2 (PDF)
Figure 2. Advances in technology and signal processing in cochlear implants have resulted in improved performance outcomes. Shown are group mean scores for CUNY and HINT sentences in quiet and CNC monosyllabic words from multiple sources: Skinner et al. (1994), Skinner et al. (1991);Pijl et al. (2009).
It is because of these improved outcomes that the criteria for implantation have expanded. Previously, the best a patient would expect was perhaps 12% sentence recognition performance with an implant;therefore the prerequisite was 0% speech recognition. As per Figure 2, now that the mean performance on clinical trials has improved to 90% open-set sentence recognition scores in quiet, those who only get 60% or less with hearing aids can consider cochlear implants a viable option if they want to hear better than they do with hearing aid amplification.
In addition to current candidacy and performance questions, dispensing professionals may have limited information or misconceptions regarding cochlear implant surgery. It is not uncommon to hear audiologists and dispensers who have not had any recent training or education on cochlear implants state, "My patients don't want brain surgery for a hearing loss." Cochlear implantation is not brain surgery. It does take a skilled surgeon to place the electrodes in the right space in the cochlea. Most people tolerate the surgery very well and actually go home from the surgical suite or outpatient clinic the same day.
In terms of reimbursement, cochlear implant reimbursement as not the same as hearing aid reimbursement. While Medicare and most insurance companies will not pay for hearing aids this is not the case with cochlear implants. Medicare will, in fact, cover a cochlear implant, as will many private insurance carriers. Bilateral implants are also sometimes covered by insurance.
Overcoming Obstacles
One obstacle to referral of appropriate candidates for cochlear implantation is lack of awareness by both professionals and potential recipients of current candidacy criteria. In a survey taken at the AudiologyNOW! conference in Charlotte, NC in 2008 (Cochlear Americas, 2008a) 437 attendees answered questions regarding the identification and referral of patients for cochlear implant evaluation. Twenty percent (20%) indicated they had not seen a cochlear implant candidate in their practice in the last six months. Fifty-five percent (55%) said they had seen less than five candidates. Combined, 75% of those who took the survey believed that they saw less than five people in their practices who might benefit from a cochlear implant in the previous six months.
Attendees were also asked how many patients they had referred for a cochlear implant evaluation in the past six months. Forty-one percent (41%) said none, 50% said less than five. In total therefore, over 90% had referred less than five patients for an implant evaluation in six months. Given the number of current recipients versus the number of people who are candidates as discussed in the next section, these numbers seem low.
It is logical that if professionals are not aware of current cochlear implant candidacy criteria, reimbursement, benefits and other up to date issues surrounding cochlear implants, they do not know how to identify candidates. Furthermore, it is probable that if professionals have no experience or contact with a cochlear implant center, they do not know how or where to refer. It is every dispensing professional's responsibility to know this information and overcome these obstacles in order to provide appropriate patients with their options for treatment.
Awareness among consumers is not much better. In a large multi-center clinical study, the average time from onset of severe-to-profound hearing loss until receiving a cochlear implant was 12 years (Balkany et al., 2007). However, once people become aware of cochlear implants and discover they are candidates, there is little hesitation to proceed. Cochlear Americas surveyed 1,501 cochlear implant recipients and found that most of them received their implant in less than one year once it was determined that they were candidates (Figure 3).
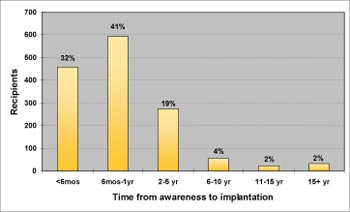
Click Here to View Larger Version of Figure 3 (PDF)
Figure 3. Time between awareness of cochlear implant candidacy and actual implantation,
n = 1501. From Cochlear Americas. (2008b).
In another survey by Cochlear Americas, cochlear implant recipients (n=594) were asked what professional referred them for their initial cochlear implant evaluation (Cochlear Americas, 2008c). Less than half were referred by audiologists or dispensers. Nearly 20% reported they were not referred by any professional. These individuals found out about cochlear implants from friends, family, the Internet, or another source (Table 3).

Click Here to View Larger Version of Table 3 (PDF)
Table 3. Responses to survey question, "Who recommended that you consider a cochlear implant evaluation?" Note that while n = 594 in this survey, total in table is greater than 594 because some people surveyed selected more than one professional. From Cochlear Americas (2008c).
These data indicate that there is a disconnect between professionals and cochlear implant candidates. One would think that most people who end up with a cochlear implant have been in contact with an audiologist or hearing instrument specialist at some point in the history of their hearing loss. The fact that over half of cochlear implant candidates do not hear about implants from a hearing care professional gives cause for reflection. The hearing healthcare profession seems to be missing the mark when it comes to informing people about cochlear implant technology when hearing aids are not enough.
How Many Cochlear Implant Candidates Are There?
How many patient files in a typical dispensing practice meet FDA-approved criteria for Nucleus™ cochlear implants? Most professionals probably have several such files. How many return to the dispensing office because they are not satisfied or do not receive adequate benefit from hearing aids?
Patient satisfaction in hearing aid fitting is an enigma. Since most dispensing professionals do not use standardized outcome measures, the majority may assume a customer who does not return an aid for credit is a satisfied customer. This assumption may be wrong. According to Kochkin (2005b;2005c) there are about 7.4 million hearing aid owners in the United States. Of these, 6.2 million are active users who wear their hearing aids at least once a year. The study also indicates that over 21% of hearing aid owners are not satisfied. For a hearing aid to be satisfactory, Kochkin suggests that people have to hear better aided in 70% of listening environments versus unaided. Seven percent (7%) of active users report that their hearing aids do not improve hearing in any situation. Further, while hearing aids provide some benefit over a wide range of hearing loss, 20% of patients with the worst hearing thresholds (more significant hearing losses) rate satisfaction with amplification lower than those with less severe hearing loss (Kochkin, 2005a). While overall customer satisfaction with hearing aids and the professionals who fit them is relatively high, there is much room for improvement. Could improvements be provided for the 21% of patients who are dissatisfied and for the 7% who report no benefit from hearing aids in any environment? Some of these people may have reached a degree of hearing loss where hearing aids are no longer adequate. Despite the best hearing aid technology and fitting, a referral for a cochlear implant evaluation may be indicated.
The exact number of people who can benefit from cochlear implants can only be estimated. While several sources report prevalence of hearing loss in the U.S., only a few report by degree of loss. Agrawal and colleagues (2008) reported 29 million adults with hearing loss. Kochkin's MarkeTrak VII (2005b) indicates hearing loss in the U.S. topped 31 million people in 2005. The Center for Disease Control reports 37 million adults with hearing loss (Schoenborn & Heyman, 2008). Of these, 3.5% self report "a lot of trouble hearing or totally deaf." The Gallaudet Research Institute reports that one million people over the age of five are "functionally deaf" (Mitchell, 2005). Blanchfield (2001) estimates there are between 500,000 and 1,200,000 people with severe to profound hearing loss. Margolis and Saly (2008), reporting on 22,000 ears tested in a large medical center, found that 8% had severe hearing loss and 5% had profound loss. ASHA reports there are 250,000 to 1,000,000 potential cochlear implant candidates in the U.S. (2008). A reasonable estimate may be 1,000,000 people in the U.S. could benefit from cochlear implants. It is estimated that there are currently only about 50-60,000 cochlear implant recipients. It is likely that less than 10% of the people who could benefit from a cochlear implant actually have one. Compare this to a 20-25% penetration rate for hearing aids.
According to the NIDCD, 60% of cochlear implant recipients are adults and 40% are children (NIDCD, 2007). Even though the absolute percentage is higher in adults, the ratio of recipients to candidates is lower for adults than for children because hearing loss is more common in adults. Maybe professionals who see children are more convinced of the benefits of implanting children than professionals who see predominantly adult patients. The bottom line is that both groups are significantly underserved, and it is the responsibility of everyone who fits hearing aids to know who, when and where to refer potential implant candidates.
While professionals may feel that they do not see patients in their dispensing practices who are cochlear implant candidates, they should not be fooled by conventional wisdom. In a review of several thousand hearing aid charts across several clinics, it was independently found that, in fact, approximately 3% of the audiograms met the FDA criteria for cochlear implantation (Table 3). These were typical hearing aid dispensing practices treating mostly adult patients. It is possible that in large medical or ENT practices the percentage might even be higher. If a dispensing practice sees 100 patients in a month, it is reasonable to suggest that three of those might receive more help from a cochlear implant than from hearing aids. For an established practice, or for experienced professionals who have a few thousand patients in their databases, it is possible that dozens of those may have improved quality of life had they received a cochlear implant.
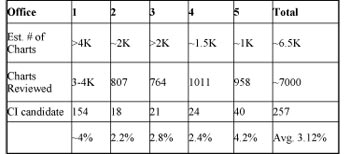
Click Here to View Larger Version of Table 4 (PDF)
Table 4. Cochlear implant candidates in a typical hearing aid practice. Results from independent review by author of existing files in five hearing aid offices (unpublished).
What's In It For Me?
Some would suggest that for professionals who own a dispensing practice or get paid based on the sale of hearing aids, the prospect of referring a patient for a cochlear implant might be bittersweet. Consider that the number one driver of customer satisfaction in the dispensing office is when professionals provide solutions to patients' hearing problems. When hearing aids do not solve the problem and a cochlear implant is recommended, professionals may become the driver of that patient's satisfaction. When patients are satisfied they tell their friends. Only a small percentage of people with hearing impairment will be implant candidates. Any goodwill generated by referring a patient for something as life changing as a cochlear implant is likely to result in more traffic to the professional's office instead of less.
Patients who get the least benefit from hearing aids may take up the most time in a dispensing practice. Efforts at reprogramming, fine-tuning and adjustments may still result in a return for credit. Even if a patient does not return an aid for credit but ends up putting it in the drawer, the result may be negative word of mouth about the professional's ability and about hearing aids in general. This is further incentive to refer for a cochlear implant evaluation for appropriate patients.
For well-satisfied hearing aid patients, there is no reason to refer for an evaluation at a cochlear implant center. Just because someone meets candidacy criteria does not necessarily make him or her a good cochlear implant candidate. Imagine that a satisfied hearing aid customer refers to his dispensing professional one of his friends who is wearing hearing aids purchased from the Internet. A professional evaluation reveals a pure-tone audiogram that falls in the shaded area on the audiogram in Figure 1. Sentence recognition scores using HINT sentences in quiet at 45 dB HL are 15% unaided, and there is no improvement when using his hearing aids, that do not meet the gain and output requirements of his audiogram. If the current hearing aids cannot be effectively adjusted new, appropriately-selected and fit hearing aids may be recommended. Once the new hearing instruments are fit, the patient reports he hears much better. A repeat of the HINT test using the new instruments reveals a sentence recognition score of 56%. Although it is not as improved as the professional would like, the patient is ecstatic with the new hearing aids. Is he a good cochlear implant candidate?
On the other hand, consider the patient who has been seen by his dispensing professional for 10 years. She was fit with her first set of CICs when she just started to notice her hearing was "not as good as it used to be." She was fit with BTEs when her hearing dropped further. She trusted the professional enough to buy power BTEs when she could not carry on a conversation using her mini BTEs and talking on the phone was completely out of the question. Her audiogram is now in the shaded area of Figure 1. Despite the professional's best efforts, her scores on the HINT never exceed 40% in the best aided condition. The new aids are definitely better, and she is satisfied with her professional and the services provided, but, unfortunately, she is afraid that she will probably quit her job or lost it because it is just too hard to hear, even with assistive technology. She reports that she is frustrated that she has to struggle to carry on a conversation with her daughter and grandchildren. She relies on her husband to answer the phone. When she stops by the dispensing office to pick up batteries, she tells the receptionist in confidence "I just wish there was something else available to help me". Is this patient a good cochlear implant candidate?
What Do I Do Next?
The first step for professionals is to be familiar with FDA-approved cochlear implant criteria (Joint Committee, 2000). It can be taped up in a conspicuous place so it is in plain view and not filed away and forgotten. Next, identify cochlear implant centers in your area and open lines of communication with them. Discuss with them about how you can share in the care of patients you both serve. About three quarters of those people who have a cochlear implant only have one. As implant criteria expands, people with more usable hearing are being implanted. That means more are likely to benefit from also using a hearing aid in the non-implanted ear (a bimodal device fitting). Finally, become an expert in fitting of bimodal devices (Huart & Sammeth, 2008). Few clinics have the time, equipment, and inclination to fit both hearing aids and cochlear implants. The audiologist who knows about cochlear implant criteria and is also proficient in bimodal fitting will have a distinct advantage in a competitive hearing aid market.
Acknowledgements: Dr. Carol Sammeth and Dr. Bobbi Scheinin for their review and comments. Dr. Aaron Parkinson for data in Figure 2.
References
Agrawal, Y., Platz, E., & Niparko, J. (2008). Prevalence of hearing loss and differences by demographic characteristics among U.S. adults. Archives of Internal Medicine, 168(14), 1522-1530.
American Academy of Audiology Task Force (2006). Guidelines for the audiological management of adult hearing impairment. Audiology Today, 18(5), 32-36.
American Speech-Language Hearing Association (2008). Incidence and prevalence of hearing loss and hearing aid use in the United States - 2008 edition. Retrieved March 25, 2009, from www.asha.org/members/research/reports/hearing.htm
Balkany, T., Hodges, A., Menapace, C. Hazard, L., Driscoll, C., Gantz, B., Kelsall, D., Luxford, W., McMenomy, S., & Neely, J. (2007). Nucleus Freedom North American clinical trial. Otolaryngology-Head and Neck Surgery, 136(5), 757-762.
Blanchfield, B., Feldman, J., Dunbar, J., & Gardner, E. (2001). The severely to profoundly hearing-impaired population in the United States: Prevalence estimates and demographics. Journal of the American Academy of Audiology, 12(4), 183-189.
Cochlear Americas (2008a). [AAA 2008 kiosk survey]. Unpublished data.
Cochlear Americas (2008b). [Cochlear Americas recipient survey]. May: N=1501. Unpublished data.
Cochlear Americas. (2008c). [Hearing aids and cochlear implant recipient survey]. Unpublished data.
Dillon H. (1999). NAL-NL1: A new procedure for fitting non-linear hearing aids. The Hearing Journal, 52, 10-16.
Dillon, H. (2001). Hearing Aids. New York: Thieme.
Dillon, H., James, A., & Ginis, J. (1997). Client Oriented Scale of Improvement (COSI) and its relationship to several other measures of benefit and satisfaction provided by hearing aids. Journal of the American Academy of Audiology, 8(1), 27-43.
Francis, H., Chee, N., Yeagle, J., Cheng, A., Niparko, J.K.(2002). Impact of cochlear implants on the functional health status of older adults. Laryngoscope, 112(8), 1482 - 1488.
Gifford, R. & Dorman, M. (2008, April). Cochlear implant candidacy indications. Presentation at the American Academy of Audiology annual convention. Dallas, TX.
Huart, S. & Sammeth, C. (2008). Hearing aids plus cochlear implants: Optimizing the bimodal pediatric fitting. The Hearing Journal, 11(61), 54-58.
Joint Committee on Clinical Practice Algorithms and Statements. (2000, August). Audiology clinical practice algorithms and statements. Audiology Today, Special Issue 2000.
Kelsall, D., Shallop, J., & Burnelli, T. (1995). Cochlear implantation in the elderly. American Journal of Otology, 16(5), 609-615.
Killion, M.C., Niquette, P.A., Gudmundsen, G.I., Revit, L.J. and Banerjee, S. (2004). Development of a quick speech-in-noise test for measuring signal-to-noise ratio loss in normal hearing and hearing-impaired listeners. Journal of the Acoustical Society of America, 116, 2395-2405.
Kochkin, S. (2005a). MarkeTrak VII: Customer satisfaction with hearing aids in the digital age. The Hearing Journal, 58(9), 30-37.
Kochkin, S. (2005b). MarkeTrak VII: Hearing loss population tops 31 million. The Hearing Review, 12(7), 16-29.
Kochkin, S. (2005c). MakeTrak VII: Obstacles to adult non-user adoption of hearing aids. The Hearing Journal, 60(4), 27-43.
Labadie, R., Carrasco, V., Gilmer, C., & Pillsbury, H. (2000). Cochlear implant performance in senior citizens. Otolaryngology-Head and Neck Suregry, 123(4), 419-424.
Margolis, R. & Saly, G. (2008). Distribution of hearing loss characteristics in a clinical population. Ear & Hearing, 29(4), 524-532.
Mitchell, R.E. (2005). Retrieved from Gallaudet University, Gallaudet Research Institute Web site: gri.gallaudet.edu/Demographics/deaf-US.php
Mueller, G.A., & Bentler, R. (1994). Measurements of TD: How loud is allowed? The Hearing Journal, 10, 42-44.
National Institute on Deafness and Other Communication Disorders (NIDCD). (2007). Cochlear implants. Retrieved April 14, 2009 from NIDCD Web site: www.nidcd.nih.gov/health/hearing/coch.asp
Pijl, S. et al. (2008). The Nucleus Freedom Cochlear Implant System: Adult IDE Clinical Trial Results. Manuscript in preparation.
Schoenborn, C.A. & Heyman, K. (2008). Health disparities among adults with hearing loss: United States, 2000-2006. Centers for Disease Control. Retrieved March 25, 2009, from www.cdc.gov/nchs/products/pubs/pubd/hestats/hearing00-06/hearing00-06.pdf
Skinner, M.W., Clark, G.M., Whitford, L.A., Seligman, P.M., Staller, S.J., Shipp, D.B. et al. (1994). Evaluation of a new spectral peak coding strategy for the Nucleus 22 channel cochlear implant system. American Journal of Otology, 15(S), 15-27.
Skinner, M.W., Holden, L.K., Holden, T.A., Dowell, R.C., Seligman, P.M., Brimacombe, J.A. et al. (1991) Performance of postlinguistically deaf adults with the wearable speech processor (WSP III) and mini speech processor (MSP) of the Nucleus multi-electrode cochlear implant. Ear & Hearing, 12(1), 3- 22.
U.S. Department of Health and Human Services. Summary Health Statistics for U.S. Adults: National Health Interview Survey, 2006. Washington, DC: Author. Retrieved from www.cdc.gov/nchs/data/series/sr_10/sr10_235.pdf
Valente, M., Hosford-Dunn, H. & Roeser, R. (2000). Audiology Treatment. New York: Thieme.
Vermeire, K., Brokx, J.P.L, Wuyts, F.L., Cochet, E., Hofkens, A., & Van de Heyning, P. (2005). Quality of life benefit from cochlear implantation in the elderly, Otology & Neurotology, 26, 188-195.


