Complete and Incomplete Canalith Jam: Diagnosis of Atypical Variants of BPPV and Key Roles of Video-HIT
Introduction
Benign paroxysmal positional vertigo (BPPV) represents the most common vestibular disorder. Detachment of otoconia from utricular macula resulting in positional vertigo is currently accepted as the underlying pathophysiological explanation. The diagnosis can sometimes be challenging as presenting signs are related to the semicircular canal (SC) affected and whether otoliths float within membranous ducts (canalolithiasis), adhere to the cupula (cupulolithiasis) or remain entrapped plugging the canal (canalith jam). Although objective measures of vestibular function could be of extreme help in selected challenging cases, the diagnostic usefulness of instrumental tests in BPPV remains controversial. In fact, studies evaluating the ability of VEMPs and other tests assessing SCs function (caloric test, rotatory chair and video-HIT) to detect the affected ear or the involved canal in patients with BPPV have achieved mainly equivocal results. Few studies have investigated the role of the video-HIT in the diagnostic work-up of BPPV so far. Though some data still remain controversial, most investigations detected normal high-frequency vestibulo-ocular reflex (VOR) for the affected SC. Nevertheless, most studies focused on BPPV with typical paroxysmal positional nystagmus, where positional nystagmus results from the transitory activation/inhibition of the ampullary receptors and debris are thought to behave as free-floating particles within the canal, not altering the dynamic responses of the SC cupula to high accelerations. In these cases, diagnosis mainly relies on the interpretation of positional nystagmus, whereas instrumental tests play a marginal role.
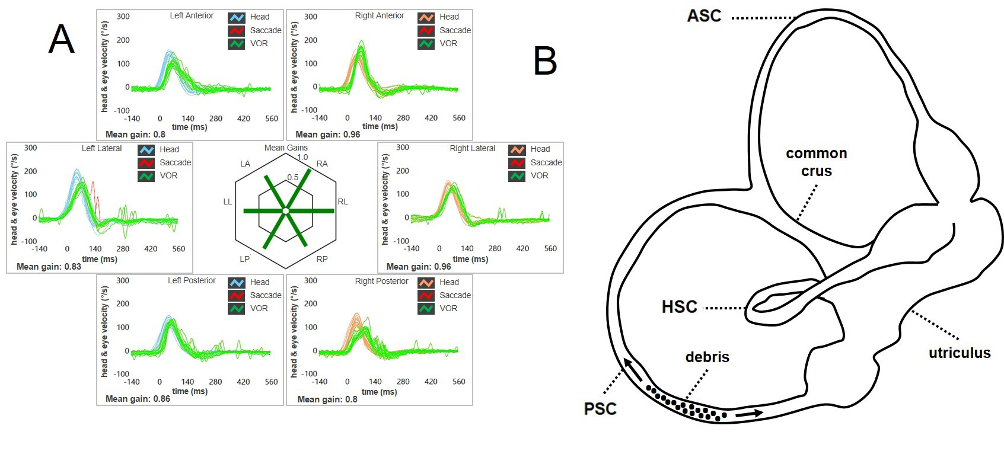
Figure 1. (A) VOR-gain for the affected canal (in this case the right PSC) is not affected on video-HIT, (B) as debris are free to float back and forth along the PSC membranous duct, not altering cupular dynamics in high-frequency ranges (Castellucci et al., 2021).
On the other hand, it may be hypothesized that in selected cases otoconia, depending on how and where they are disposed within the membranous ducts, may aggregate in clots and lead to endolymphatic flows altering cupular dynamic responses, resulting in challenging differential diagnosis with acute unilateral vestibular loss (AUVL) or with central nervous system (CNS) disorders. According to the recent literature, these situations include complete canalith jam (CJ) and a subsample of anterior SC(ASC)-BPPV and apogeotropic variants of posterior SC (PSC)-BPPV resulting in persistent positional downbeat nystagmus (DBN), where an incomplete (or positional) CJ might be hypothesized
AudiologyOnline: What are clinical-instrumental signs of a complete canalith jam? How can the video-HIT be useful for differential diagnosis?
Andrea Castellucci: In the case of a complete CJ, otoliths are assumed to aggregate in a clot which entirely plugs the canal lumen (either spontaneously or as a result of inappropriate repositioning procedures for BPPV). This condition is supposed to prevent physiologic endolymphatic flows and result in a continuous positive or negative pressure between the clot itself and the cupula. This scenario results in a persistent deflection of the cupula (bending either toward the utricle, in case of positive pressure, or away from the utricle, in case of negative pressure) accounting for spontaneous nystagmus irrespective to head position changes. In this situation, both high- and low-frequency responses of the ampulla of the affected SC should be impaired. Accordingly, horizontal SC (HSC)-CJ has demonstrated to result in spontaneous horizontal nystagmus (either paretic or irritative depending on the direction of the cupular bending) with ipsilateral canal paresis on caloric irrigations and selective impairment for HSC VOR-gain at the video-HIT. Once the CJ has restored and debris have dispersed along the canal (usually after impulsive therapeutic maneuvers along the plane of the affected SC, reinforced by robust head shakings and mastoid vibrations), both caloric and head impulse responses normalize. In this case, a selective HSC impairment on video-HIT may be of extreme help in differentiating spontaneous horizontal nystagmus due to an HSC-CJ from a similar scenario due to an AUVL, where an additional impairment for the ASC and the utricular afferents, at least, should be expected in the acute stage. In case of irritative spontaneous horizontal nystagmus (beating toward the lesioned site), the use of the video-HIT in addition to other instrumental tests (such as audiometry, and cervical/ocular VEMPs) could help in the differential diagnosis with an hydropic attack in Menière’s disease in the ictal stage.
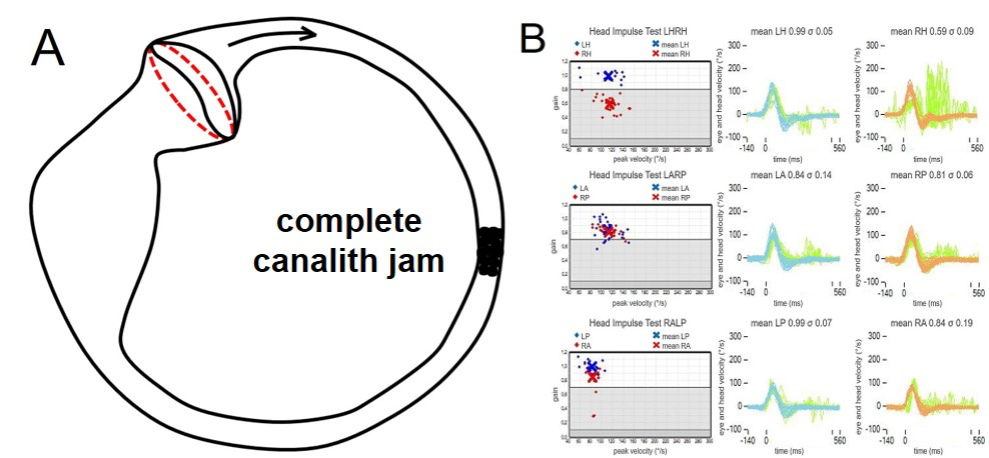
Figure 2. (A) An otolith clot within the HSC can likely plug the canal and continuously exert a negative pressure over the cupula, leading to a spontaneous horizontal paretic nystagmus mimicking an AUVL. (B) This condition results in a selective VOR-gain impairment for the affected HSC (in this case, right HSC - canalith jam) (Castellucci et al., 2019).
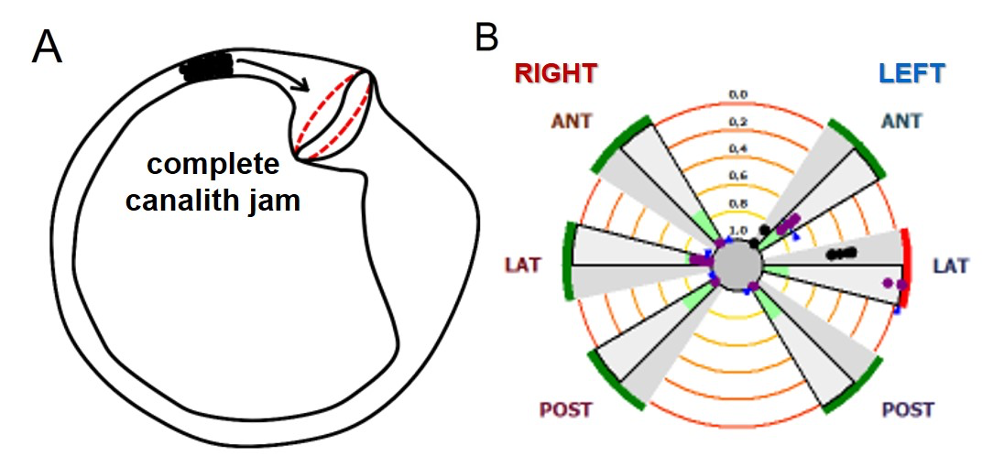
Figure 3. (A) Alternatively, an otolith clot might also continuously exert a positive pressure over the cupula, leading to a spontaneous horizontal irritative nystagmus mimicking an acute attack of Menière’s disease. (B) This condition can also result in a selective VOR-gain impairment for the affected HSC (in this case, left HSC - canalith jam) (Castellucci et al., 2019).
Similarly, a selective PSC impairment at the video-HIT has demonstrated to represent an important clue in differentiating spontaneous DBN due to CNS pathologies involving cerebellar and brainstem pathways from a PSC-CJ. In fact, while in the former condition other end-organs dysfunctions and/or additional signs of central origin (i.e., purely vertical spontaneous DBN not reduced by visual fixation, gaze-evoked nystagmus, impairment of saccadic eye movements and smooth pursuit, etc.) should be expected, in the case of PSC-CJ debris are thought to aggregate in a clot and remain entrapped in a narrow tract of the non-ampullary arm of the PSC. In this position, the otolith clot is thought to exert a continuous positive pressure on the cupula, persistently inhibiting only the PSC afferents. The sole PSC-VOR gain impairment at the video-HIT, the slight contralateral torsional component of spontaneous DBN due to an unopposed activation of the contralateral ASC, the lack of CNS signs and a possible recent history consistent with ipsilesional BPPV should help in the diagnosis of PCS-CJ. In these cases, impulsive physical treatment along the affected SC (such as Semont / Demi Semont / quick liberatory rotation maneuver), with the aid of mastoid vibrations and head shakings, and prolonged positioning with the head toward the healthy side should result either in a conversion of spontaneous DBN into a paroxysmal positional nystagmus (due to a dispersion of otoconia along the ampullary arm of the PSC) or in a receding of nystagmus (likely due to a replacement of otoconia within the utricle). In both conditions, PSC VOR-gain values at the video-HIT should normalize.
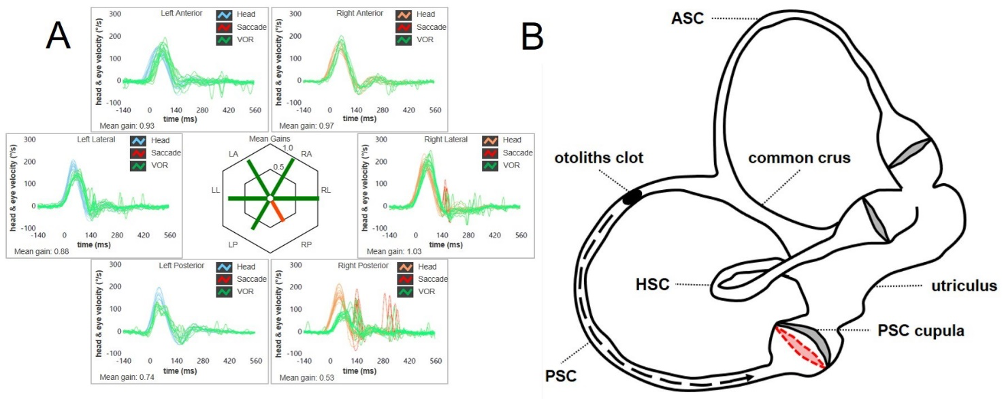
Figure 4. (A) A selective reduction for PSC VOR-gain at the video-HIT (in this case the right PSC) may result in spontaneous downbeating nystagmus with slight contralesional torsional component. (B) An otoconial plug entrapped within the PSC membranous duct could likely exert a continuous positive pressure on the cupula, preventing dynamic responses for the ampullary afferents of the right PSC by blocking endolymphatic flows (Castellucci et al., 2021).
Anecdotally, it has been described how otoconia might aggregate and settle in the ASC where they could remain entrapped due to stenotic tracts of the membranous duct, leading to an ASC-CJ. In this case, a spontaneous upbeating nystagmus could be detected, as debris are thought to exert a continuous positive pressure on the cupula, persistently inhibiting the ASC afferents. This condition mimics an acute CNS disorder and a careful examination excluding CNS involvement should always be given. Even in this case, the detection of a selective reduction of the affected ASC VOR-gain, slight contralesional torsional components of spontaneous upbeat nystagmus due to an unopposed activity of the contralesional PSC, a possible recent history of ipsilesional BPPV and the lack of coexistent oculomotor signs of CSN involvement should orient towards an otolithic etiology. After impulsive physical therapy aimed at unlocking the entrapped otoliths from the ASC, ASC VOR-gain should restored at the video-HIT.
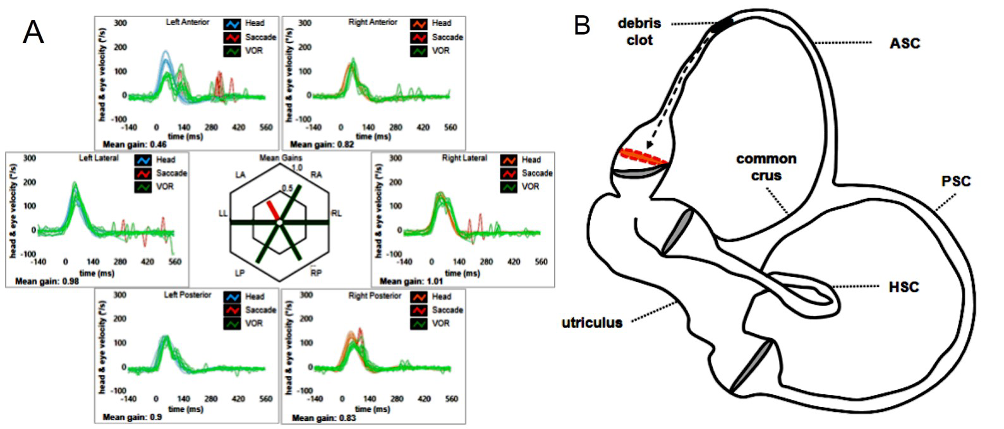
Figure 5. (A) A selective reduction for ASC VOR-gain at the video-HIT (in this case the left ASC) may result in spontaneous upbeating nystagmus with slight contralesional torsional components. (B) A debris clot within the membranous duct of the ASC could likely exert a continuous positive pressure on the cupula, preventing dynamic responses on the left ASC plane by blocking endolymphatic flows (Castellucci et al., 2022).
AudiologyOnline: Which are the clinical-instrumental signs of an incomplete canalith jam? How could the video-HIT be useful for differential diagnosis?
Andrea Castellucci: The second condition in which the video-HIT may play an important role in differential diagnosis is when BPPV results in persistent positional DBN. This type of nystagmus can be generated by both CNS and peripheral vestibular dysfunctions and it usually evoked when the patient is brought to the straight head hanging position and/or by Dix Hallpike maneuvers. In peripheral disorders, positional DBN has been mainly related to ASC-BPPV and, more recently, it has been hypothesized that it may also come from otoliths settling in the distal portion of the non-ampullary arm of PSC, close to the common crus (i.e., the “apogeotropic variant” of PSC-BPPV). Whereas in ASC-BPPV debris are thought to shift away from ASC-ampulla resulting in a utriculofugal deflection of the cupula and in an excitatory discharge of the superior ampullary nerve, in apogeotropic PSC-BPPV positioning maneuvers should move debris towards the ampulla producing a PSC inhibition, which in turn results in positional DBN with inconstant torsional components beating toward the uppermost ear. As both forms of BPPV result in positional DBN, it is frequently difficult to identify the affected ear because of the possible lack of torsional components. Moreover, apogeotropic PSC-BPPV is hardly distinguishable from BPPV involving the contralateral ASC since in both cases positional DBN results from the contraction of the same ocular muscles. Nevertheless, mostly in cases with persistent positional DBN, an “incomplete” or “positional” CJ has been hypothesized. Namely, debris are thought to be partially entrapped in narrow portions of the membranous duct, such as the distal portion of the non-ampullary arm of PSC, close to the common crus, or within possible structural stenosis of the ampullary arm of the ASC. When the patient is brought head-down into a diagnostic position, debris are thought to slowly move with gravity until they totally plug the canal lumen, thus leading to a continuous endolymphatic and a persistent cupular bending. When the patient is brought back upright, otoconia cease exerting continuous pressure of the cupula and nystagmus recedes. This condition could likely result in an “incomplete” or “positional” CJ behaving as a “low-pass filter”, i.e., allowing the cupula to be activated by low-frequency stimuli (otoconial shifts) while impeding the ampullary receptor to respond to high-frequency inputs (head impulses). This pathophysiological hypothesis has allowed explaining how patients with persistent positional DBN either due to apogeotropic PSC-BPPV or ASC-BPPV exhibited impaired VOR-gain values for the affected canal, that restore after repositioning maneuvers or after converting in a classical ipsilateral BPPV with paroxysmal nystagmus.
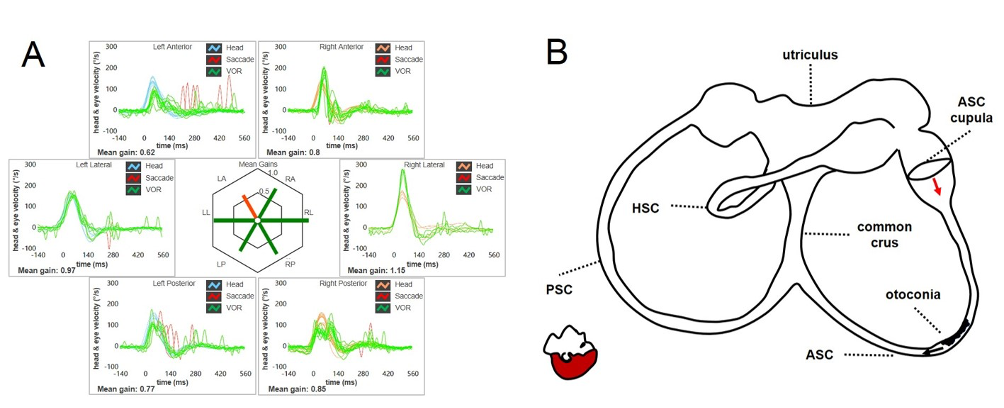
Figure 6. (A) ASC-BPPV may result in a selective reduction for high-frequency VOR-gain values for the involved canal (in this case the left ASC). (B) Debris could be located within a nearly horizontal tract of the canal, where possible narrowing or irregularities could allow them to settle without resulting in nystagmus in upright position. In this condition, otoconia are thought to partially occlude the canal lumen resulting in an incomplete CJ, preventing only high-frequency stimuli as detected by the video-HIT. After positioning tests, otoconia may slowly shift toward the non-ampullary tract of the canal below due to gravity and resulting endolymphatic movements should lead ASC cupula to bend ampullofugally (excitation), accounting for persistent positional downbeating nystagmus (Castellucci et al., 2020).
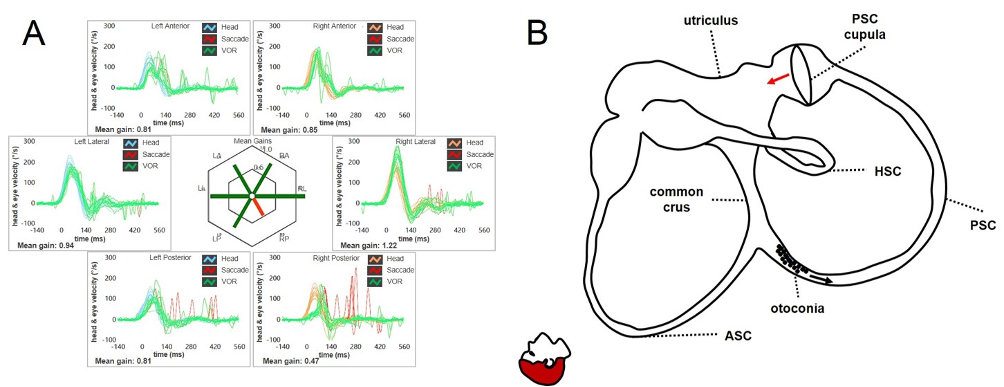
Figure 7. (A) Apogeotropic PSC-BPPV leading to a selective reduction for high-frequency VOR-gain for the affected canal (in this case the right PSC). (B) Even in this case, otoliths are partially entrapped within a narrow tract of the non-ampullary arm of the canal nearly aligning with the horizontal axis, explaining the lack of spontaneous nystagmus. As in the case of ASC-BPPV, even in this condition the canal is thought to be partially plugged by otoconia, explaining high-frequency VOR-gain impairment for the involved canal. Once diagnostic head hanging position is obtained, gravity vector should slowly move debris toward the ampullary tract of the canal below, leading to an ampullopetal endolymphatic flow and resulting cupular bending should generate an inhibitory discharge for the PSC afferents, accounting for persistent positional downbeating nystagmus (Castellucci et al., 2020).
The following table summarizes three possible scenarios accounting for different patterns of nystagmus in BPPV and vHIT measurements, with corresponding hypothetical pathomechanisms.
| Eye Movements | VOR-Gain for the Affected SC on vHIT | Proposed Underlying Mechanism | Endolymphatic Flows | |
Low-Frequency | High-Frequency | ||||
Typical Canalolithiasis | transient paroxysmal nystagmus in positioning test, reversing with gravity | generally normal | free-floating debris moving back and forth within the SC | preserved as debris are free to move in both directions along the SC | preserved as debris neither aggregate nor occlude the SC, thus they do not alter ampullary responses |
Incomplete (“Positional”) Canalith Jam | persistent pDBN in both head-down positionings, rarely reversing while upright | slightly reduced | debris are partially entrapped in a narrow tract of the SC, partially plugging the canal lumen | likely preserved as debris, even if partly blocked, are allowed to slowly shift towards the cupula or in the opposite direction in head-down positionings | reduced as debris likely impede high-frequency flows dampening ampullary responses to head impulses (behaving as a ”low-pass filter”) |
Complete Canalith Jam | spontaneous nystagmus, slightly increasing in positioning tests | markedly reduced | an otolith clot is completely entrapped within a narrow tract of the SC, entirely plugging the canal lumen | prevented due to the continuous endolymphatic pressure, leading to a constant cupular deflection | prevented due to the continuous endolymphatic pressure, leading to a constant cupular deflection |
These findings should encourage clinicians to include the video-HIT in the test battery of patients with spontaneous nystagmus and/or with persistent positional DBN, as it may be of extreme help in the differential diagnosis with AUVL or CNS pathologies, enabling the detection of the involved canal in BPPV, with important therapeutic and prognostic implications.
In conclusion, the video-HIT appears to play a minority role in the clinical management of typical BPPV where its routine use should not be recommended. Nevertheless, it is reasonable to assume that it may play a key role in atypical forms of BPPV, where spontaneous or persistent positional DBN are likely to result from altered dynamic responses of SC afferents, providing useful data for differential diagnosis from other peripheral and CNS pathologies.
Resources for More Information
For more information, visit https://www.inventis.it/en-na
References
Agus, G., Puxeddu, R., Demontis, G. P., & Puxeddu, P. (1995). Atypical "reversed" paroxysmal positioning nystagmus in benign paroxysmal positional vertigo. Acta Otolaryngologica Supplementum, 520(Pt 1), 143-147.
Bertholon, P., Bronstein, A. M., Davies, R. A., Rudge, P., & Thilo, K. V. (2002). Positional down beating nystagmus in 50 patients: Cerebellar disorders and possible anterior semicircular canalithiasis. Journal of Neurology, Neurosurgery, and Psychiatry, 72, 366-372.
Brandt, T. (1990). Positional and positioning vertigo and nystagmus. Journal of the Neurological Sciences, 95, 3-28.
Bronstein, A. M., Kaski, D., Cutfield, N., Buckwell, D., Banga, R., Ray, J., Chavda, S., & Irving, R. (2015). Head-jolting nystagmus: Occlusion of the horizontal semicircular canal induced by vigorous head shaking. JAMA Otolaryngology-Head & Neck Surgery, 141(8), 757-760.
Büki, B., Mandalà, M., & Nuti, D. (2014). Typical and atypical benign paroxysmal positional vertigo: Literature review and new theoretical considerations. Journal of Vestibular Research, 24, 415-423.
Büttner, U., Helmchen, C., & Brandt, T. (1999). Diagnostic criteria for central versus peripheral positioning nystagmus and vertigo: A review. Acta Oto-Laryngologica, 119, 1-5.
Califano, L., Capparuccia, P. G., Di Maria, D., Melillo, M. G., & Villari, D. (2003). Treatment of benign paroxysmal positional vertigo of posterior semicircular canal by "Quick Liberatory Rotation Manoeuvre". Acta Otorhinolaryngologica Italica, 23(3), 161-167.
Califano, L., Salafia, F., Mazzone, S., Melillo, M. G., & Califano, M. (2014). Anterior canal BPPV and apogeotropic posterior canal BPPV: Two rare forms of vertical canalolithiasis. Acta Otorhinolaryngologica Italica, 34, 189-197.
Califano, L., Iannella, R., Mazzone, S., Salafia, F., & Melillo, M. G. (2021). The video head impulse test in the acute stage of posterior canal benign paroxysmal positional vertigo. Acta Otorhinolaryngologica Italica, 41(1), 69-76.
Cambi, J., Astore, S., Mandalà, M., Trabalzini, F., & Nuti, D. (2013). Natural course of positional down-beating nystagmus of peripheral origin. Journal of Neurology, 260, 1489-1496.
Casani, A. P., Cerchiai, N., Dallan, I., & Sellari-Franceschini, S. (2011). Anterior canal lithiasis: Diagnosis and treatment. Otolaryngology-Head and Neck Surgery, 144, 412-418.
Castellucci, A., Malara, P., Brandolini, C., Del Vecchio, V., Giordano, D., Ghidini, A., Ferri, G. G., & Pirodda, A. (2019). Isolated horizontal canal hypofunction differentiating a canalith jam from an acute peripheral vestibular loss. American Journal of Otolaryngology, 40, 319-322.
Castellucci, A., Malara, P., Delmonte, S., & Ghidini, A. (2020). A possible role of video-head impulse test in detecting canal involvement in benign paroxysmal positional vertigo presenting with positional downbeat nystagmus. Otology & Neurotology, 41, 386-391.
Castellucci, A., Malara, P., Martellucci, S., Botti, C., Delmonte, S., Quaglieri, S., Rebecchi, E., Armato, E., Ralli, M., Manfrin, M. L., Ghidini, A., & Asprella Libonati, G. (2020). Feasibility of using the video-head impulse test to detect the involved canal in benign paroxysmal positional vertigo presenting with positional downbeat nystagmus. Frontiers in Neurology, 11, 578588.
Castellucci, A., Malara, P., & Ghidini, A. (2021). Spontaneous downbeat nystagmus in posterior semicircular canal benign paroxysmal positional vertigo: A canalith jam? Neurological Sciences, 42, 313-315.
Castellucci, A., Malara, P., Martellucci, S., Delmonte, S., & Ghidini, A. (2021). Fluctuating posterior canal function in benign paroxysmal positional vertigo depending on how and where otoconia are disposed. Otology & Neurotology, 42, e193-e198.
Castellucci, A., Piras, G., Del Vecchio, V., Ferri, G. G., Ghidini, A., & Brandolini, C. (2021). Which inner ear disorders lie behind a selective posterior semicircular canal hypofunction on video head impulse test? Otology & Neurotology, 42, 573-584.
Castellucci, A., Malara, P., Martellucci, S., Armato, E., & Califano, L. (2021). Possible pathomechanism behind the transient hypofunction of the affected canal in BPPV. European Archives of Oto-Rhino-Laryngology, 279(2), 1117-1118.
Castellucci, A., Botti, C., Martellucci, S., Malara, P., Delmonte, S., Lusetti, F., & Ghidini, A. (2022). Spontaneous upbeat nystagmus and selective anterior semicircular canal hypofunction on video head impulse test: A new variant of canalith jam? Journal of Audiology and Otology, 26(3), 153-159.
Chang, Y. S., Choi, J., & Chung, W. H. (2014). Persistent direction-fixed nystagmus following canalith repositioning maneuver for horizontal canal BPPV: A case of canalith jam. Clinical and Experimental Otorhinolaryngology, 7(2), 138-141.
Choi, J. Y., Kim, H. J., & Kim, J. S. (2018). Recent advances in head impulse test findings in central vestibular disorders. Neurology, 90, 602-612.
Çınar, Y., Bayram, A., Culfa, R., & Mutlu, C. (2018). Analyses with the video head impulse test during the canalith repositioning maneuver in patients with isolated posterior semicircular canal benign paroxysmal positional vertigo. Turkish Archives of Otorhinolaryngology, 56, 81-84.
Comacchio, F., Poletto, E., & Mion, M. (2018). Spontaneous canalith jam and apogeotropic horizontal canal benign paroxysmal positional vertigo: Considerations on a particular case mimicking an acute vestibular deficit. Otology & Neurotology, 39(9), e843-e848.
Elsherif, M., Eldeeb, D., & Eldeeb, M. (2021). Clinical significance of video head impulse test in benign paroxysmal positional vertigo: A meta-analysis. European Archives of Oto-Rhino-Laryngology, 278(12), 4645-4651.
Epley, J. M. (1997). Caveats in particle repositioning for treatment of canalithiasis (BPPV). Operative Techniques in Otolaryngology-Head and Neck Surgery, 8, 868-876.
Epley, J. M. (2001). Human experience with canalith repositioning maneuvers. Annals of the New York Academy of Sciences, 942, 179-191.
Fallahnezhad, T., Adel Ghahraman, M., Farahani, S., Hoseinabadi, R., & Jalaie, S. (2017). Vestibulo-ocular reflex abnormalities in posterior semicircular canal benign paroxysmal positional vertigo: A pilot study. Iranian Journal of Otorhinolaryngology, 29, 269-274.
Halmagyi, G. M., Chen, L., MacDougall, H. G., Weber, K. P., McGarvie, L. A., & Curthoys, I. S. (2017). The video head impulse test. Frontiers in Neurology, 8, 258.
Iida, M., Hitouji, K., & Takahashi, M. (2001). Vertical semicircular canal function: A study in patients with benign paroxysmal positional vertigo. Acta Oto-Laryngologica Supplementum, 545, 35-37.
Jeong, S. H., Kim, H. J., & Kim, J. S. (2013). Vestibular neuritis. Seminars in Neurology, 33(3), 185-194.
Ko, K. M., Song, M. H., Kim, J. H., & Shim, D. B. (2014). Persistent spontaneous nystagmus following canalith repositioning procedure in horizontal semicircular canal benign paroxysmal positional vertigo. JAMA Otolaryngology-Head & Neck Surgery, 140(3), 250-252.
Lopez-Escamez, J. A., Molina, M. I., & Gamiz, M. J. (2006). Anterior semicircular canal benign paroxysmal positional vertigo and positional downbeating nystagmus. American Journal of Otolaryngology, 27, 173-178.
Luis, L., Costa, J., Vaz Garcia, F., Valls-Solé, J., Brandt, T., & Schneider, E. (2013). Spontaneous plugging of the horizontal semicircular canal with reversible canal dysfunction and recovery of vestibular evoked myogenic potentials. Otology & Neurotology, 34, 743-747.
Mangabeira Albernaz, P. L., & Zuma e Maia, F. C. (2014). The video head impulse test. Acta Oto-Laryngologica, 134, 1245-1250.
Martellucci, S., Castellucci, A., Malara, P., Pagliuca, G., Clemenzi, V., Stolfa, A., Gallo, A., & Libonati, G. A. (2022). Spontaneous jamming of horizontal semicircular canal combined with canalolithiasis of contralateral posterior semicircular canal. Journal of Audiology and Otology, 26(1), 55-60.
Molnár, A., Maihoub, S., Tamás, L., & Szirmai, Á. (2022). A possible objective test to detect benign paroxysmal positional vertigo. The role of the caloric and video-head impulse tests in the diagnosis. Journal of Otolaryngology, 17(1), 46-49.
Navari, E., & Casani, A. P. (2020). Lesion patterns and possible implications for recovery in acute unilateral vestibulopathy. Otolaryngology-Head and Neck Surgery, 41, e250-e255.
Perez-Fernandez, N., Martinez-Lopez, M., & Manrique-Huarte, R. (2014). Vestibulo-ocular reflex in patients with superior semicircular canal benign paroxysmal positional vertigo (BPPV). Acta Oto-Laryngologica, 134, 485-490.
Saltürk, Z., & Yetişer, S. (2020). Video head impulse testing in patients with benign paroxysmal positional vertigo. Acta Oto-Laryngologica, 140(12), 977-981.
Schubert, M. C., Helminski, J., Zee, D. S., Cristiano, E., Giannone, A., Tortoriello, G., & Marcelli, V. (2020). Horizontal semicircular canal jam: Two new cases and possible mechanisms. Laryngoscope Investigative Otolaryngology, 5, 163-167.
Sekine, K., Imai, T., Morita, M., Nakamae, K., Miura, K., Fujioka, H., Kubo, T., Tamura, K., & Takeda, N. (2004). Vertical canal function in normal subjects and patients with benign paroxysmal positional vertigo. Acta Oto-Laryngologica, 124, 1046-1052.
Tarnutzer, A. A., Bockisch, C. J., Buffone, E., & Weber, K. P. (2017). Association of posterior semicircular canal hypofunction on video-head-impulse testing with other vestibulo-cochlear deficits. Clinical Neurophysiology, 128(8), 1532-1541.
Vannucchi, P., Pecci, R., & Giannoni, B. (2012). Posterior semicircular canal benign paroxysmal positional vertigo presenting with torsional downbeating nystagmus: An apogeotropic variant. International Journal of Otolaryngology, 2012, 413603.
Vannucchi, P., Pecci, R., Giannoni, B., Di Giustino, F., Santimone, R., & Mengucci, A. (2015). Apogeotropic posterior semicircular canal benign paroxysmal positional vertigo: Some clinical and therapeutic considerations. Audiology Research, 5, 130.
Von Brevern, M., Clarke, A. H., & Lempert, T. (2001). Continuous vertigo and spontaneous nystagmus due to canalolithiasis of the horizontal canal. Neurology, 56(5), 684-686.


