 From the desk of Gus Mueller
From the desk of Gus Mueller
We've all seen the Best Practice Guidelines for the selection and fitting of hearing aids. These guidelines typically include stages such as pre-testing, needs assessment, style and fitting considerations, selection of features, verification and validation. Part of validation typically is some sort of real-world self-assessment inventory. And with adults, we have several to choose from: the COSI, APHAB, SADL, HHIE, IOI-HA, PAL, Glasgow, SSQ, and the list goes on. But these scales are all for adults--what about the pediatric patient? Or, the infant or toddler who is fitted with hearing aids? Is there a systematic way to monitor their performance in the real world following the hearing aid fitting?
If you want to check out the latest research and clinical insights in the world of pediatric amplification, you can narrow your choices down to three or four locations fairly quickly. One of them is the University of Western Ontario. That's where our 20Q "Question Man" traveled this month to talk to Marlene Bagatto, AuD, to learn about a new pediatric monitoring protocol that she has been working on the past few years. Dr. Bagatto has been a member of the research team in the Child Amplification Laboratory at the University of Western Ontario for the past 12 years. As you know, this group is primarily known for their work with the Desired Sensation Level (DSL) Method for fitting hearing aids to children. You've probably seen Dr. Bagatto's publications regarding integrating auditory brainstem responses with the hearing aid prescription and infant-friendly ways of measuring the real-ear-to-coupler difference.
Dr. Bagatto has been a member of the research team in the Child Amplification Laboratory at the University of Western Ontario for the past 12 years. As you know, this group is primarily known for their work with the Desired Sensation Level (DSL) Method for fitting hearing aids to children. You've probably seen Dr. Bagatto's publications regarding integrating auditory brainstem responses with the hearing aid prescription and infant-friendly ways of measuring the real-ear-to-coupler difference.
Marlene not only conducts research, but she is involved in assisting practicing audiologists with pediatric fittings, and she continues to see young patients herself. I suspect that it was these activities that prompted her to focus her PhD work on the development and evaluation of an outcome evaluation guideline for infants and children who wear hearing aids. In this month's 20Q, she tells us about The University of Western Ontario Pediatric Audiological Monitoring Protocol (UWO PedAMP) and the evidence to support the guideline.
I hear that when Marlene is not thinking about the PedAMP, or writing the final sections of her dissertation, she can be found frolicking with her 100-pound Bernese Mountain Dog—a breed known for their excellent cart-pulling abilities. When it comes to pediatric amplification, we're thankful that Marlene and the audiology crew at UWO continue to do the "heavy pulling" for all of us.
Gus Mueller, Ph.D.
Contributing Editor
February 2012
To browse the complete collection of 20Q with Gus Mueller articles, please visit www.audiologyonline.com/20Q
 1. Outcome evaluation is a unique area in pediatrics, especially related to hearing aid fittings. What sparked your interest?
1. Outcome evaluation is a unique area in pediatrics, especially related to hearing aid fittings. What sparked your interest?
A few things actually. As a clinician working with infants and young children with hearing loss, I often find myself being asked by parents "How do I know if the hearing aids are helping my baby?" Being the clinician responsible for habilitation with hearing aids for these children, I also wanted to know the impact of the use of amplification. Especially for the young infants at the early stages of hearing aid use, I didn't have a systematic and evidence-based way to answer the parents' questions.
Another contributing factor was back in 2008 I participated in a focus group meeting arranged by my good friend and colleague Dr. Sheila Moodie at the University of Western Ontario. As you know from her recent 20Q article (Moodie, 2011), she has spent the last several years learning about knowledge translation and implementation science. In fact, they were the topics of her PhD dissertation. The focus group was the first meeting of the Network of Pediatric Audiologists of Canada, a group of pediatric audiologists from several provinces across Canada who are interested in participating in clinical research in various ways. One of the Network's roles at the meeting was to identify specific areas of clinical practice that needed better evidence to support their systematic use in the clinic.
2. So did this Canadian Network come up with anything?
They most certainly did, which again relates to the topic of our conversation. The main area they identified was the need for well-developed, normed and validated outcome measures for infants and young children who wear hearing aids that would actually be feasible to implement in a clinical setting. They understood the importance of measuring outcomes, and some even used available tools, but they reported they did not know which tools were best to use that also had meaning for their work. It was during these meetings that I decided to focus my PhD work on developing an outcome evaluation guideline for infants and young children who wear hearing aids. The guideline is called the University of Western Ontario Pediatric Audiological Monitoring Protocol (UWO PedAMP). It is targeted for infants and young children up to six years of age who wear hearing aids and focuses on auditory-related outcomes. The guideline is a tool for audiologists to implement following the verification stage of the hearing aid fitting process.
3. Well, thanks for yet another acronym! What types of outcome evaluation tools did you include in the guideline?
The tools included in the UWO PedAMP (you're welcome!) can be divided into two groups: 1) clinical process outcomes and 2) functional outcomes. Clinical process outcomes relate to the hearing aid verification information for each child. Functional outcomes relate to the child's auditory development or auditory performance. Interpreting functional outcomes for a child is not useful unless you know about the quality of the hearing aid fitting. We know early intervention with hearing aids is important for outcome (e.g., Sininger, Grimes, & Christensen, 2010; Yoshinaga-Itano, Sedey, Coulter, & Mehl, 1998), and this is something that should be tracked. However, what has been lacking in many Early Hearing Detection and Intervention (EHDI) programs is a description of the quality of the hearing aid fitting.
We have comprehensive pediatric hearing aid fitting protocols that describe the components of the hearing aid fitting process (e.g., American Academy of Audiology, 2003; Bagatto, Scollie, Hyde, & Seewald, 2010). They commonly include a recommendation for a real-ear-to-coupler difference (RECD) measurement and coupler-based verification in order to match the hearing aid gain and output to pediatric prescriptive targets. These components are important to ensure good audibility of speech across a broad frequency range while maintaining comfort for loud sounds so as to support speech and language development. However, if these components are not completed, and completed well, the quality of the hearing aid fitting could have a negative impact on the child's outcome with intervention.
Therefore, the UWO PedAMP provides a systematic way of documenting the hearing aid fitting details, or clinical process outcomes, so that when the functional outcomes are obtained, they can be interpreted more accurately. It is true that poor functional outcomes may occur, but it is important to know from the beginning of the outcome evaluation process whether this is due to a poor match to prescriptive targets and not something inherently developmental with the child. After all, isn't the fit-to-targets the first thing we check when we see a child in the clinic for a second opinion? Evaluating both types of outcomes provides a comprehensive guideline for outcome evaluation in the pediatric hearing aid fitting process.
4. Sounds like a good idea, and a lot of work. What hearing aid fitting details are you monitoring as clinical process outcomes?
I understand it sounds daunting at first, but the hearing aid fitting details that are monitored are already items that the pediatric audiologist obtained during the verification stage of the hearing aid fitting process. That is, the clinician will have measured or predicted the RECD, verified the match to targets for soft and average-level speech inputs, and measured the maximum power output (MPO). More specifically, the fit-to-targets is not tracked, but the Speech Intelligibility Index (SII) of the fitting for soft and average speech inputs is assessed for each hearing aid fitting. The SII is the proportion of speech that is available to the hearing aid user (American National Standards Institute, 1997) and is found within several probe-microphone hearing aid fitting systems (e.g., Audioscan®, Interacoustics®). A summary of these details is provided in Table 1 below. For the RECD, the clinician can indicate whether it was measured, predicted, or if other ear or previously measured values were used. An SII value from 0 to 100 is documented for a soft speech input as well as an average speech input. Finally, the clinician should indicate whether the MPO was verified to targets by choosing 'Yes' or not verified by choosing 'No'. Following verification of the hearing aid, the audiologist simply documents the details of the fitting in the table.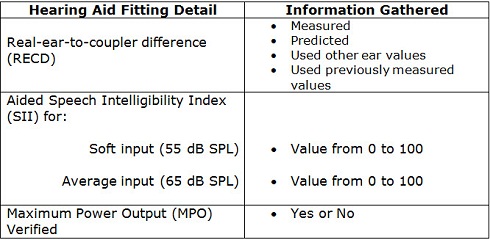
Table 1. Summary of hearing aid fitting details gathered for the UWO PedAMP.
5. Sounds good, but when I look at the aided SII values, I often have trouble deciding what is "good enough".
We have something that might help you. We developed a worksheet that allows the clinician to compare the aided SII value to a set of normative values that we gathered with the help of the Network (Moodie, 2009, 2010). By "normative" I mean that these values could be considered typical SII values for a child with a particular pure tone average. They were obtained by gathering the SII values from 161 pediatric hearing aid fittings using the Desired Sensation Level (DSL) Method. All of the fittings were roughly within 5 dB of the prescribed DSL target. This allows the clinician to determine whether the aided SII value is appropriate for the patient's degree of hearing loss (see Figure 1). It is a general indicator of the quality of the hearing aid fitting and does not replace the clinician's judgment of the match to prescriptive targets. However, these hearing aid fitting details provide an overall idea of the quality of the hearing aid fitting to more clearly interpret the functional outcomes. Collecting these as part of an outcome evaluation guideline in a clinic or EHDI program can allow tracking of an individual child's fitting as well as overall program data. Wouldn't it be great to know how often the RECD is measured in your clinic or EHDI program? Or, how close the SII values come to provided norms? Knowing the details of the hearing aid fitting provides information about the quality of the hearing aid fittings for the program as well as the individual child in addition to providing a basis for functional outcome interpretation.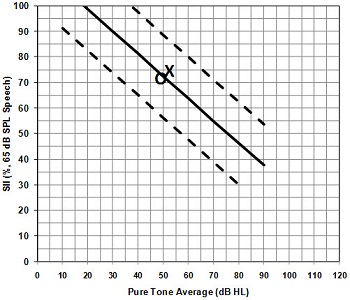
Figure 1. A portion of the Aided Speech Intelligibility Index (SII) Normative Values Worksheet from the UWO PedAMP. The solid line indicates the average aided SII values for an average (65 dB SPL) speech input and the dashed lines indicate the 95% confidence intervals, plotted by pure tone average (PTA) of the frequencies 500, 1000 and 2000 Hz. A 'typical' aided SII value should fall within the dashed lines when plotted by PTA. The 'O' and 'X' represent the aided individual SII values for an average speech input for the right and left hearing aid fitting of a child with a PTA of around 50 dB HL.
6. That's great! Let's move on to the functional outcome evaluation tools you mentioned.
Sure. We chose to focus on subjective caregiver-report functional outcome evaluation tools in the first version of the UWO PedAMP. While objective measures such as speech detection and recognition tasks are important, they often are not suitable for very young infants or those children with other medical issues who we struggle to get behavioral audiograms from in the first place. Also, objective measures are considered by the World Health Organization to be capacity measures, or what the child can do in a clinical setting (World Health Organization [WHO], 2001).
While these are important and will be included in future versions of the UWO PedAMP, we decided to focus on subjective questionnaires initially. This is because they are considered performance measures, or what the child can do in the real world (WHO, 2001). Having the child's caregiver report on the child's auditory behaviors using a questionnaire will provide rich information that cannot be obtained through objective speech measures. Additionally, the questionnaires can be completed for infants who are too young to perform speech tests reliably as well as for those children who have other medical issues that impact their ability to perform objective behavioral tests. Finally, with the introduction of this guideline, we wanted to include tools that could be implemented with relative ease so that clinical uptake would be more successful.
7. I see. But there are a lot of questionnaires out there. How did you decide which ones to include in the guideline?
Taking guidance from the knowledge-to-action framework I mentioned earlier (Graham, et al., 2006; Moodie, et al., 2011), I applied what I learned from the knowledge inquiry stage (i.e., the Network focus group) and critically reviewed the available subjective outcome measures as opposed to developing new questionnaires. In this knowledge synthesis stage, I used a published grading system (Andresen, 2000) to rate the questionnaires available for infants and children with hearing loss from birth to six years of age. This was the age group that the Network clinicians identified as the population of interest for this initial guideline work. There are other age groups that would benefit from a guideline but this group was the primary focus for this first project. In addition to being suitable for children up to six years of age, a parent-report questionnaire was included in the critical review if it could be administered and scored by an audiologist and had specific items relating to auditory behaviors.
While there are many questionnaires available, very few have published norms for children with hearing loss who wear hearing aids. It is important to include questionnaires with norms so that they have clinical meaning when a score is calculated. Therefore, questionnaires scored more favorably if they had published normative data, good reliability and validity as well as low administrative and respondent burden. It was important for us to develop a guideline that is balanced in both statistical properties as well as clinical feasibility. Some evidence-based guidelines can be too rigid and complex and therefore are not easily adopted into clinical practice. We wanted to ensure that the chosen tools were scientifically and practically balanced. If you would like to read more about the critical review, it was recently published in a special issue of Trends in Amplification where more about the knowledge-to-action framework and how it was implemented in the development of the UWO PedAMP can be found (Bagatto, Moodie, Seewald, Bartlett, & Scollie, 2011).
8. The suspense is killing me—which questionnaires made the final cut for the UWO PedAMP?
Based on the critical review, there were two questionnaires that scored highly: 1) the LittlEARS® Auditory Questionnaire (Tsiakpini, et al., 2004) and; 2) the Parents' Evaluation of Aural/Oral Performance of Children (PEACH) (Ching & Hill, 2005b). The items in the LittlEARS are more appropriate for young infants and the PEACH items describe more complex scenerios that are suitable for toddlers and young children. Therefore, we included both in a two-stage developmental process within the guideline. In the UWO PedAMP, we suggest that the LittlEARS be administered until a certain score and age is reached and then the PEACH can be administered. This prevents caregivers of young infants from feeling discouraged by low scores on a questionnaire that is not yet developmentally appropriate for their child.
9. I like the name "LittlEARS" but to be honest, I don't know much about it.
The LittlEARS is a 35-item questionnaire that assesses auditory development. It was originally developed for children 24 months and younger. Caregivers provide yes/no answers to the questions which are provided in developmental order. The total 'yes' score is plotted on a score sheet at the point where age and score meet. The score sheet displays normative values, as well as minimum and maximum values, for the clinician to compare to. The norms have been developed with 218 German-speaking families of normal hearing children (Coninx, et al., 2009) and have been validated in 16 languages, including a validation study we did with Canadian families of English-speaking normal hearing children (Bagatto, Brown, Moodie, & Scollie, 2011). The LittlEARS scored highly in the critical review because it has published norms, is reliable, valid and is quite easy to administer and interpret.
10. Sounds fairly straightforward. How about the PEACH?
Well, this one requires a bit more explanation. There is a diary version of the PEACH (Ching & Hill, 2005a) which requires the family to observe their child with hearing aids for a period of a week in eleven scenerios in order to obtain an idea of the child's auditory performance in quiet and noisy situations. The caregiver must write down as many examples of the child's auditory behavior over that one week period. Then they are interviewed by the audiologist to obtain an understanding of the examples provided and perhaps gain more examples. The score is based on the number of examples provided by the caregiver in each scenario.
The PEACH rated highly in the critical review because it has been normed with children with normal hearing as well as those who have hearing loss and wear hearing aids. What did not get rated as highly was the feasibility of the diary version of the PEACH. Because the scoring relies on the number of examples, it is important that the caregiver spend time providing as many as he/she can, as well as the audiologist being a skilled interviewer. In our pilot work with the PEACH Diary, many families did not complete the diary or provided very few examples. Also, the clinicians found the interview portion took up a lot of clinical time. However, there is a Rating Scale version of the PEACH that is available (Ching & Hill, 2005b) that the Network clinicians found more feasible to include in the UWO PedAMP. The PEACH Rating Scale includes all of the scenarios from the Diary, but the caregiver simply rates the frequency with which they saw the behavior over the last week on a five point rating scale. The scores are added up and a percentage is calculated for the quiet and noise subscales.
11. How does the clinician know if the score is within the normative range on these questionnaires?
Good question, it certainly is important to know how well the child scored compared to published norms. The authors of the LittlEARS provided a score sheet that displays the average normative values as well as the minimum and maximum values as a function of age in months. The age range goes up to 24 months with this score sheet. Through our work with this questionnaire, we administered it to caregivers of children with various hearing and medical profiles. We learned that the LittlEARS is sensitive to other medical issues besides hearing loss that can affect the score. We didn't want the family to have to complete the PEACH for a child who is not developmentally ready for the items on that questionnaire. Therefore, we modified the existing LittlEARS score sheet to extend the age range to 48 months. The norms remain the same since they have been validated for use with Canadian English-speaking families (Bagatto, Brown, et al., 2011).
The PEACH is a little different. Instructions for how to calculate the PEACH score are available, but there was no way to compare the child's score to published norms as with the LittlEARS. We really wanted the tools included in the UWO PedAMP to be clinically meaningful and utilizable. Therefore, we created a score sheet to assist clinicians with the interpretation of the scores. We used data from the published norms as well as scores we obtained through using the PEACH in research studies (Ching & Hill, 2007; Ching, et al., 2010). This allowed us to plot performance ranges that the clinician could compare the child's scores to, all summarized on a single score sheet.
12. So the LittlEARS assesses auditory development and the PEACH Rating Scale assesses auditory performance. Is that all the questionnaires?
Actually there is one more. We believed that we also should assess service delivery and program quality. For this reason, our lab worked together with the managers and consultants of the Ontario Infant Hearing Program to develop an eleven item questionnaire that addresses the acceptance and use of hearing aids, auditory performance for different levels of sound (e.g., soft and average speech), effectiveness of service delivery and overall satisfaction. It is called the Infant Hearing Program Amplification Benefit Questionnaire (we'll spare you the acronym for this one!). The questions have a rating scale answer format, with the exception of the last question which is open-ended and asks the caregiver how they think hearing aid services could be improved. The questionnaire was based on the International Outcome Inventory for Hearing Aids (IOI-HA; Cox & Alexander, 2002), but tailored for EHDI programs. We believed it was important to address hearing aid services so that the program could obtain an idea of how it is doing. This may help support the quality and potential future funding of the EHDI program. There are no norms or score sheet provided as it is really a satisfaction and quality assurance survey, but it does a good job of identifying areas of need for the family in terms of the services being provided.
13. You really thought of everything! Where can I find the questionnaires and score sheets?
It was important for us to do everything possible to support the clinical implementation of the UWO PedAMP. Therefore, we developed a training manual with an accompanying video (Bagatto, Moodie, & Scollie, 2010). The manual provides the relevant background on both the clinical process outcomes as well as the functional outcomes. It includes all of the score sheets, where the questionnaires can be found, frequently asked questions, case studies and recent publications. We have posted the manual on our DSL website (www.dslio.com). It is important to note that since we did not develop the LittlEARS and the PEACH, they are not posted but information about where to find them are in the manual as well as in Table 2 below.
Table 2. Information about where to find UWO PedAMP information and tools.
14. Do you think that the average clinician can implement this into their clinical practice?
Within the manual there is an administration grid. It lists the routine audiology appointments that the child and family generally attend when the child wears hearing aids. The outcome evaluation tools are designed to be completed at routine clinical appointments therefore, extra appointments are not necessary. The administration grid indicates when a particular outcome evaluation tool should be administered. One important rule is to know when to stop using the LittlEARS and administer the PEACH. As seen in the grid, when the child reaches a score of 27 or greater on the LittlEARS and is 24 months of age and older, then the PEACH should be administered. This rule is supported by data from a longitudinal intervention study we did using the UWO PedAMP with children in Ontario (Bagatto, Moodie, Malandrino, et al., 2011). It was noted through this clinical study that some children met the LittlEARS score criteria of 27, but were quite young; around 16 to 18 months of age. When the PEACH was administered, they scored poorly due to the developmental level of the PEACH items. Data from our recent work shows that children under 24 months of age score more poorly on the PEACH than children older than 24 months of age (Bagatto, Moodie, Malandrino, et al., 2011). Therefore, this administration rule was implemented for the functional outcome evaluation tools.
15. How much time will this add to a typical clinical appointment?
Good point—we know that "time" can be the main barrier for implementing new clinical procedures. According to the clinicians who participated in our clinical study, the LittlEARS takes the caregiver about six minutes to complete and the PEACH takes about ten minutes to complete. Including scoring time, interpretation and paperwork, the UWO PedAMP is estimated to take about 20 minutes. However, some clinicians have reported that the questionnaires include items that they would typically ask during the case history, but now they are provided in a systematic way. In addition, the questionnaires can be given to the caregiver in the waiting room or while the audiologist is assessing the hearing aids to save time. It is true that new clinical measures take time, but we believe that once they are implemented their value will be realized and outcome evaluation will become a routine part of pediatric audiology practice.
16. So how does using the UWO PedAMP fit into the pediatric hearing aid fitting process?
As you may be aware, there are several stages of the hearing aid fitting process (Figure 2). Outcome evaluation is designed to be applied following verification of the child's hearing aids. It is hoped that the UWO PedAMP will provide information about the impact of the hearing aid fitting, especially in the early stages of hearing aid use. Especially with the use of clinical process outcomes as well as functional outcomes, the UWO PedAMP provides a systematic and evidence-based way of applying outcome evaluation tools in clinical practice.
Figure 2. The stages of the hearing aid fitting process. In Assessment, considerations for audiometry and ear canal acoustics are considered. In Selection, prescriptive targets are generated and appropriate electroacoustic and non-electroacoustic properties of the hearing aid to be fitted are considered. The verification stage involves matching the gain and output of the hearing aid to the prescribed targets. Finally, outcome evaluation is designed to systematically measure in the impact of the hearing aid fitting.
17. What about children who do not wear hearing aids? Can the UWO PedAMP be used with them?
Evaluation is equally important for those children identified with permanent hearing loss and do not wear hearing aids. In this case, you are not measuring "outcome" per se, but for instance, children with minimal/mild bilateral hearing loss, unilateral hearing loss, auditory neuropathy spectrum disorder, etc. and the parents have decided not to aid the child. It is very important to monitor the child's auditory development and performance even when they are not wearing hearing aids. Information gathered from the LittlEARS and the PEACH on these children can provide support for intervention, further testing or medical referrals.
18. Tell me more about the study you have been working on and how that supports this guideline?
The longitudinal intervention study involved four clinics with five pediatric audiologists collecting data using the tools within the UWO PedAMP. Participants were parents of infants and children with and without hearing aids and had various medical profiles. We did not exclude children with other medical issues or complex factors related to hearing aid use (i.e., inconsistent hearing aid use). We felt it was important to understand how these children performed on the outcome evaluation tools given that each group makes up about 30 to 40% of the clinical pediatric audiology population. In addition, they were part of the reason why we chose to implement questionnaires in the first version of the UWO PedAMP. The recently published work provides data for children with hearing aids of various levels of hearing loss and medical profiles for both the LittlEARS and the PEACH as well as hearing aid fitting details (Bagatto, Moodie, Malandrino, et al., 2011). Case studies are also included to support implementation of the guideline in a clinical setting. More data has been collected since this publication and will be presented in future articles.
19. Who has been using the UWO PedAMP?
The UWO PedAMP was introduced in stages to the Ontario Infant Hearing Program starting in December 2009. It was fully implemented through in-person training sessions by July 2010. Training has also occurred in Winnipeg, Manitoba, Canada and the British Columbia Early Hearing Program has shown interest in adopting the guideline. Training has been conducted at individual clinics within North America. However, with the availability of our training manual and video, it is possible for clinicians to implement it anytime. Our group is available to support implementation, just as we are with the DSL Method.
20. So what's your prediction? Is this something that will become routine with pediatric amplification?
It is my hope that it will. Many clinicians already realize the importance of tracking the impact of hearing aids for infants and young children, especially during the early stages of amplification use. They just needed some guidance regarding which tools to use and how to extract meaning from the scores they obtained. And the families want to know how their child is doing. By including them in outcome evaluation, they become more involved in the process as well as better observers of their child's auditory behaviors.
The UWO PedAMP provides a comprehensive, systematic, evidence-based guideline that is also clinically feasible. With the inclusion of visual tools to permit rapid scoring, clinicians can compare their patients' scores to available normative values for interpretation. This injects meaning into the outcome evaluation stage of the hearing aid fitting process. Supporting clinical uptake of this guideline was an important aspect of the development of the UWO PedAMP. Hopefully its implementation will improve the efficiency and effectiveness of service delivery for young patients with hearing loss.
Acknowledgements - Funding
This work was supported with funding from the Canadian Institutes of Health Research [Marlene Bagatto: 200811CGV-204713-174463]. This work has also been supported by Ontario Ministry of Children and Youth Services Infant Hearing Program, The Ontario Research Fund Early Researcher Award, Siemens Hearing Instruments, Canada and The Masonic Foundation of Ontario, Help-2-Hear Project.
References
American Academy of Audiology (Producer). (2003). American Academy of Audiology Pediatric Amplification Guidelines. Retrieved from www.audiology.org/resources/documentlibrary/Documents/pedamp.pdf
American National Standards Institute. (1997). Methods for the calculation of the Speech Intelligibility Index (ANSI S3.5-1997). New York, NY: Acoustical Society of America.
Andresen, E. (2000). Criteria for assessing the tools of disability outcomes research. Archives of Physical Medicine and Rehabilitation, 81(Suppl 2), S15-S20. doi: 10.1053/apmr.2000.20619
Bagatto, M., Scollie, S., Hyde, M., & Seewald, R. (2010). Protocol for the provision of amplification within the Ontario Infant Hearing Program. International Journal of Audiology, 49, S70-S79. doi: 10.3109/14992020903080751
Bagatto, M. P., Brown, C. L., Moodie, S. T., & Scollie, S. D. (2011). External validation of the LittlEARS auditory questionnaire with English-speaking families of Canadian children with normal hearing. International Journal of Pediatric Otorhinolaryngology, 75, 815-817. doi: 10.1016/j.ijporl.2011.03.014
Bagatto, M. P., Moodie, S. T., Malandrino, A. C., Richert, F. M., Clench, D. A., & Scollie, S. D. (2011). The University of Western Ontario Pediatric Audiological Monitoring Protocol (UWO PedAMP). Trends in Amplification, 15(1), 57 - 76. doi: 10.1177/1084713811420304
Bagatto, M. P., Moodie, S. T., & Scollie, S. D. (2010). The University of Western Ontario Pediatric Audiological Monitoring Protocol (UWO PedAMP) Version 1.0: Training Manual. The University of Western Ontario. London, Ontario.
Bagatto, M. P., Moodie, S. T., Seewald, R. C., Bartlett, D. J., & Scollie, S. D. (2011). A critical review of audiological outcome measures for infants and children. Trends in Amplification, 15(1), 23 - 33. doi: 10.1177/1084713811412056
Ching, T. Y., & Hill, M. (2005a). The Parents' Evaluation of Aural/Oral Performance of Children (PEACH) Diary. Australian Hearing. Chatswood, New South Wales, Australia. Retrieved from www.nal.gov.au/outcome-measures_tab_peach.shtml
Ching, T. Y., & Hill, M. (2005b). The Parents' Evaluation of Aural/Oral Performance of Children (PEACH) Rating Scale, from www.outcomes.nal.gov.au/LOCHI assessments.html
Ching, T. Y., & Hill, M. (2007). The parents' evaluation of aural/oral performance of children (PEACH) scale: Normative data. Journal of the American Academy of Audiology, 18, 220-235.
Ching, T. Y., Scollie, S. D., Dillon, H., Seewald, R. C., Britton, L., Steinberg, J., et al. (2010). Evaluation of the NAL-NAL1 and the DSL v.4.1 prescriptions for children: Paired-comparison intelligibility judgements and functional performance ratings. International Journal of Audiology, 49, S35-S48. doi: 10.3109/14992020903095791
Coninx, F., Weichbold, V., Tsiakpini, L., Autrique, E., Bescond, G., Tamas, L., et al. (2009). Validation of the LittlEARS Auditory Questionnaire in children with normal hearing. International Journal of Pediatric Otorhinolaryngology, 73, 1761-1768. doi: 10.1016/j.ijporl.2009.09.036
Cox, R. M., & Alexander, G. C. (2002). The International Outcome Inventory for Hearing Aids (IOI-HA): Psychometric properties of the English version. International Journal of Audiology, 41(1), 30-35.
Graham, I. D., Logan, J., Harrison, M. B., Straus, S. E., Tetroe, J., Caswell, W., et al. (2006). Lost in knowledge translation: Time for a map? Journal of Continuing Education in the Health Professions, 26, 13 - 24. doi: 10.1002/ehp.47
Moodie, S. T. (2009). Clinician fit-to-DSL targets: Preliminary data from a network study. AudiologyOnline, Recorded Course #14898. Retrieved from /audiology-ceus/course/hearing-aids-children-clinician-fit-to-dsl-targets-14898
Moodie, S. T. (2010, October). Engaging audiologists in research: The Network of Pediatric Audiologists of Canada. Canadian Academy of Audiology Presentation. Montreal, QC.
Moodie, S. T. (2011). 20Q: Moving from evidence to practice - Can knowledge translation and implementation science help audiology get there? AudiologyOnline, Article #2392. Retrieved from www.audiologyonline.com/articles/article_detail.asp?article_id=2392
Moodie, S. T., Kothari, A., Bagatto, M. P., Seewald, R. C., Miller, L., & Scollie, S. D. (2011). Knowledge translation in audiology: Promoting the clinical application of best evidence. Trends in Amplification, 15(1), 5 - 22. doi: 10.1177/1084713811420740
Sininger, Y., Grimes, A., & Christensen, E. (2010). Auditory development in early amplified children: Factors influencing auditory-based communication outcomes in children with hearing loss. Ear and Hearing, 31(2), 166-185.
Tsiakpini, L., Weichbold, V., Kuehn-Inacker, H., Coninx, F., D'Haese, P., & Almadin, S. (2004). LittlEARS Auditory Questionnaire. MED-EL. Innsbruck, Austria.
World Health Organization (Producer). (2001). International classification of functioning, disability and health. Retrieved from www.who.int/classifications/icf/en/
Yoshinaga-Itano, C., Sedey, A. L., Coulter, D. K., & Mehl, A. L. (1998). Language of early- and later-identified children with hearing loss. Pediatrics, 102(5), 1161-1171. doi: 10.1542/peds.102.5.1161


