Editor's note: This is a transcript of an AudiologyOnline live expert seminar. Please download supplemental course materials.
GUS MUELLER: It is good to be back for another edition of Vanderbilt Journal Club. All of the Vanderbilt Audiology Journal Club sessions on AudiologyOnline feature a guest from the Vanderbilt University audiology faculty reviewing and discussing pertinent journal articles around a particular theme. Today's guest is Dr. Fred Bess and he will be talking about ethics. In addition, each Vandy Journal Club contains a few other special features relevant to audiology clinical practice that we hope keep these seminars lively and interesting, including What They're Reading at Vandy, Gus' Pick of the Month, and Vandy Vignettes. So let's get started.
What They're Reading at Vandy
In this segment, we feature an article recommended by one of our audiology team members at Vanderbilt. Today we're changing it up a bit by discussing what they're writing at Vandy. An interesting topic now in hearing aid research is how hearing aids might reduce fatigue and add to ease of listening. It is an area of hearing aid benefit that I think is very intriguing, and Vanderbilt's Dr. Ben Hornsby did an excellent job of reviewing the literature on that particular topic in his recent AudiologyOnline 20Q article entitled Hearing loss, hearing aids, and listening effort (January, 2012).
In this article, Ben points out some potentially beneficial things that hearing aids are doing that are sometimes difficult for us to measure in the clinic, and I think you will find some good counseling tips there as well.
Gus's Pick of the Month
As part of the Vanderbilt Audiology Journal Club sessions, I typically select a particular article that I've found interesting and clinically relevant. This month I chose something that I enjoyed reading that indirectly relates to Fred's topic of ethics.
We all have seen the marketing hype that occurs whenever new technology is introduced. Technology commonly is launched at the AudiologyNOW! meeting, and there is usually some supporting evidence for the technology. You will see quotes like, "The clinical trials showed significantly improved results", or, "Users preferred this new technology," and so on.
An article published last year in Ear & Hearing entitled The Placebo Effect and the Influence of Participant Expectation on Hearing Aid Trials (Dawes, Powell, & Munro, 2011) relates to product evaluation. This study was modeled very closely to what a hearing aid manufacturer might use with respect to sample size, degree of hearing loss and age of subjects. The participants were 20 experienced hearing aid users (54 to 80 years old; mean = 69), with bilateral downward sloping hearing loss. In this case, they used two hearing aids that were programmed identically, but one hearing aid had a yellow case and the other hearing aid had a beige case. The participants were told that one of the two hearing aids had new technology. They purposely did not blind the subjects to the technology. Understand that this happens in some clinical research with hearing aids as well, as when size or style is different, blinding isn't possible. Sometimes the examiner may say, "We have something new for you to try." The authors used three comparative measures. One was a speech-recognition-in-noise test, one was a sound quality rating, and the other was an overall personal preference rating.
First was the Four Alternative Auditory Feature (FAAF) speech-in-noise test. You might think this is a completely objective test, however, the results showed that there was, in fact, a placebo effect. There was about a two percent difference in favor of the "new" technology over the "conventional" technology, which was indeed statistically significant. These data can be plotted various ways, as I've done on my next two slides (Figure 1). The upper chart is one representation of the data, while the lower slide shows the data as you might see plotted by an ambitious marketing department at a manufacturer's booth or in a brochure.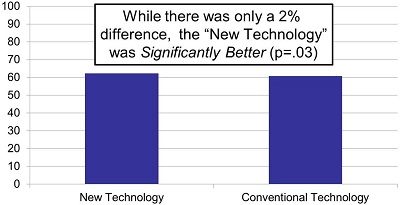
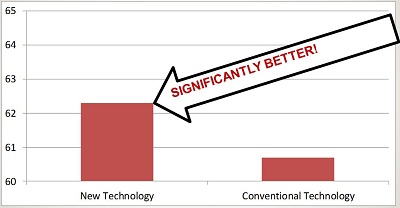
Figure 1. Two different representations of the same data, a 2% difference on the speech-in-noise test. In the lower slide, the 2% difference has a much larger visual representation, making it appear more significant.
For the sound quality ratings, the subjects judged the comfort, clarity, and overall impression of both instruments. Differences in all the sound quality ratings between the technologies were significant, with the "new" technology receiving the higher ratings. Remember again that these were the same hearing aids, programmed identically, just in different colored housings. At the end of the study, the examiners asked the subjects, "If you had to wear one of these hearing aids for next five years, which would you choose?" Seventy-five percent of the subjects picked the hearing aid with the "new" technology. Twenty-five percent said there was no difference, and not one person picked the hearing aid that had the "old" technology.
Audiology Practice and the Potential for Conflicts of Interest
On to our guest today, Dr. Fred Bess. Fred Bess is certainly one of the leaders in the field of audiology and has been for the last several decades. For nearly 30 years he was the chair of the Hearing and Speech Department at Vanderbilt's Bill Wilkerson Center. He stepped down from that position a couple of years ago, but continued working, and now serves as the Director of the National Center for Childhood Deafness at Vanderbilt. He continues to do great work and is an inspiration to all of us.
FRED BESS: Thanks, Gus. I welcome the opportunity to share with you all some information that I hope is of value to you. We are talking about something unique to the other great Vandy Journal Clubs sessions we have had. Today, I won't be talking about technology or any sophisticated methodology. There are no complex analyses. Today, we are talking about basic things like ethics and conflicts of interest. We will talk about perceptions and attitudes. Much of what we know in this area comes from surveys of physicians, students or audiologists, and they are asked about attitudes, perceptions, and behaviors.
I think that most of us have had some challenges in our history with regard to conflicts of interest. I know that I have. You have to recognize that when I entered the profession of audiology there were only 300 audiologists in the United States, and when we were providing clinical services we did not dispense hearing aids. It was considered unethical.
As a consequence, we evaluated different hearing aids, identified the one we thought was the most appropriate, and then referred the patient on to a hearing aid dispenser to receive the hearing aid. Most of the time we never knew if they ever got the aid, and if they did we did not know if they received the one that we had recommended. Nevertheless, we thought that we were lily-white because we were within the ethical guidelines established by the American Speech-Language-Hearing Association (ASHA).
Because we did not personally dispense the hearing aids, we made contacts with all the hearing aid dispensers in our areas. We met with them on a frequent basis. We would have lunch with them. We occasionally would meet with them at the ASHA meeting, so, in a sense, there was a conflict of interest. I, for one, have had some situations in the past relative to conflicts of interest in a time when we did not know very much about this area. If I had this to do over again, I certainly would do some of these things differently.
Ethics and Conflict of Interest
One of the unique aspects of audiology is that it is assumed by our society that we put our clients' interests ahead of our own, and, in fact, that society permits us, as a profession, to have a great degree of self-governance. We have tremendous leeway to regulate and govern our own profession, and the way we do that essentially is through a code of ethics. In fact, the most readily accessible standard for conduct is our code of ethics. A code of ethics orders that a trust relationship exist between members of a profession and their patients. It sets a minimal standard. It determines those who are included and those who are excluded from the profession, and if we find ourselves deviating from that code we either need to amend the code of ethics, or we need to change our behavior. The code of ethics is a very important component of our profession. It governs what we do and defines who we are.
What are the differences that exist between morals versus ethics? To many, the two terms are synonymous, but they do have subtle differences. Morals define personal character. Morals are conforming to a standard of "right behavior." Ethics is conforming to an accepted professional standard or code of conduct. When we look at the differences between the two terms, consider, if you will, a criminal defense lawyer. Although the lawyer's personal moral code finds murder immoral, ethics demand that the accused be defended. Consider also, if you will, a physician who does not believe in abortion. Abortion is legal and therefore medically ethical, although many may find it immoral.
There are a few other things that we need to remember about ethics. If something is illegal it is most likely also unethical, but many unethical behaviors are not illegal. If your patients perceive an action as unethical, it could be considered unethical. In fact, there are a number of people who believe strongly that if something is perceived as unethical, it is unethical. Finally, conflicts of interest in clinical practice are considered unethical.
Let's talk about conflicts of interest for just a few moments. Conflicts occur when the interests of audiologists are out-of-sync with the interests of their patients. Our primary interest as audiologists is to provide the best possible services we can to our patients. When this poor alignment repeats itself over time, when it is institutionalized, and/or when it becomes systemic, then it not only compromises the quality of care to those that we are here to serve, but it also undermines the integrity of the audiology profession.
There are a few things that we need to remember about conflicts of interest. When the reimbursement of clinicians appears to encourage decisions that are not in the best interest of their patients, a conflict exists. When industry influences our decisions, whether through such activities as gifts, support of continuing education units (CEUs), travel, entertainment or sales incentives, a conflict exists. We will focus on the industry influences of clinicians today. We will look at some of the published literature, primarily in the medical field, about implications that exist as a consequence of some of these activities.
Finally, it is important to know that recognition and acknowledgement of the potential for conflicts of interest are the very first steps in effective intervention. It is important, too, to recognize that the AAA Code of Ethics specifies that individuals shall not participate in activities that constitute a conflict of professional interest. There are many organizations related to audiology and communication disorders that have their own codes of ethics - ASHA, the Academy of Doctors of Audiology, the Academy of Rehabilitative Audiology, Educational Audiology, and the list goes on. Some of the codes are modified to meet the unique needs of specialty groups.
Seminal Events in the Evolution of Audiology
Two seminal events in the evolution of audiology were hearing aid dispensing and the inception of the American Academy of Audiology. There are few that would argue this point. Both of these events increased our professional autonomy, and, at the same time, our dependence on hearing aid manufacturers. Many of you probably have heard about the 33 founders of the American Academy of Audiology who went to Houston to deliberate on the feasibility of the development of an organization of, by, and for audiologists. At the end of the day after that particular meeting when we were all ready to return to our homes, we realized that we needed a budget. Each member of that group put $20 on the table; this was not a lot of money to run a professional organization of, by, and for audiologists, but, nevertheless, we did what we could.
We had few resources and, as a result, we drew on the assistance of manufacturers. They graciously agreed and supported us, and as time went on the dependence on hearing aid manufacturers grew, and we began to realize that our convention, which defines the Academy to many members, was starting to see a significant presence of the industry. Exhibits were getting very large and there were all kinds of promotional items at the various exhibits. People started to ask, "Is this a meeting of audiology professionals or is this a meeting of hearing aid manufacturers?"
At this point in time, the American Academy of Audiology began to get more involved, but along with this issue of involvement and presence of manufacturers, a study was done at Nova Southeastern by Teri Hamill and colleagues (Hawkins, Hamill, Van Vliet, & Freeman, 2002). They conducted a 20-item questionnaire that described a variety of professional and business activities. Hamill and colleagues surveyed participants, who were both patients and audiologists, to judge these activities based on the following classifications: nothing wrong with this; may not be in the patient's best interest; borders on unethical practice; clearly unethical.
The survey compared audiologists' perceptions or attitudes with those of patients. One scenario was, "The audiologist earns credits for each hearing aid purchased. The credits can be redeemed for gifts or cruises." The vast majority of the patients (50%) said this activity was clearly unethical, whereas only nineteen percent of the audiologists indicated the same. Additionally only nineteen percent of audiologists said it was borderline unethical, with the majority (32%) saying there was nothing wrong with this practice. An obvious discrepancy exists between the perception of patients versus the perception of audiologists.
Another scenario was, "The manufacturer gives a $100 traveler's check for each aid purchased." Again, the same response categories were there, and 64% of patients said this is clearly unethical, while only 26% of audiologists believed this was unethical. This data caused concern on the part of the Academy and prompted them to become more actively involved when it came to the issues of conflict of interest within our profession.
When you looked at the reactions from audiologists, most of them would say these types of activities are not a problem because they are always doing what is in the best interest for their patient. Others might think, "Stay out of our business. We want to do our thing. We are going to do the right thing," or even, "I don't want to hear this. Don't talk to me about it." Still, others might say, "I think that this has the potential to be a serious problem. We need to do something about it."
That is how we started to become more involved in conflict of interest and ethics issues as a profession. The prospect that we might not be serving patients as our primary interest or that we are compromising the integrity of our profession is concerning. The American Academy of Audiology (AAA) came forward and was very proactive in implementing guidelines since this time. We will talk more about that in a few moments.
Milestones in Ethics
There are some important ethical milestones worth mentioning. First are the audiologists who have made an ethical difference in our profession. One is Barry Elpern. He was an audiologist back in the 1960s at the University of Chicago. One cold, wintry day he was on his way home, and the freeway became impassable due to a blizzard. He was forced to sleep in his car that evening. The next day got out of his car and trekked his way back to his home, and after a few days of thinking he said, "This is no way to go through life in Chicago." Consequently, he packed up his family and moved them to Arizona. He found a job with a group of engineers who were interested in making hearing aids, and they asked him, because he was an audiologist, to start testing some individuals with the hearing aids to see if they worked and to look at differences and changes in technology. Before long, he was dispensing hearing aids for them and, as a consequence, he was drummed out of ASHA for breaching the code of ethics. It was unethical at that time for audiologists to dispense hearing aids. At that time, when you were dismissed from ASHA, you were essentially removed from the profession, too. Elpern persisted, and others soon joined on to help. After a period of time, ASHA modified its code of ethics to allow audiologists to dispense amplification. Our profession is very different today because of the work of Barry Elpern, and we should all be grateful that he challenged that code of ethics and made it possible for us to dispense hearing aids.
Another important person in the area of ethics for our profession is David Resnick. David Resnick could be called the Father of Codes of Ethics within our profession. He chaired the Committee on Ethics for ASHA and the American Academy of Audiology.
Brian Walden also played an important role within the American Academy of Audiology. In 2001, David Fabry, as president of the Academy at the time, put together a presidential task force to explore the status of conflicts of interest within the profession of audiology and he asked Brian Walden to head up that task force. It was my pleasure to serve on that task force with Brian. The bulk of the leadership and work effort came from Brian. He created a great document that was submitted to the American Academy of Audiology leadership with recommendations on what to do. Numerous efforts were launched as a consequence of that report.
The ethical milestones for AAA are as follows. First, David Resnick crafts the first code of ethics in 1991, and upon order of a AAA presidential task force in 2001, a new code of ethics was drafted. Next, an awareness campaign was launched by Angela Loavenbruck, who was the next president of the American Academy of Audiology in 2002-2003. She set out to educate our membership on the issues of ethics and conflicts of interest within the profession.
Around 2002-2003, the Academy established the Ethical Practices Board. They have been very active and influential in educating our membership. They established the first Ethical Practice Guidelines in 2003, as well as the newest revision that came out in 2011. I would encourage all of you to take a look at those guidelines. They are available on the Academy's Web site.
The ban on giveaways from manufacturers at the AudiologyNOW! convention is something new as of this year, 2012. This is a proactive stance which states very clearly that we are concerned about ethics and the influence or perception of influence that some of these items can have on our profession.
Journal Articles
Much of what we are going to be talking about regarding ethics is based upon what we have learned from medicine. The magnitude of the conflicts of interest problem in physician-industry relationships is rather significant because a lot of money is spent, approximately $8,000 to $10,000 per physician per year. The pharmaceutical industry spends 30 to 40 percent of its revenues on selling products. There are 90,000 drug representatives, which is 1 for every 4.7 office-based physicians. Interactions with the industry include receipts of gifts, drug samples, free meals, travel, entertainment, subsidized attendance to meetings and honoraria to speak. Nine-hundred million dollars of the one billion dollars spent annually on continuing medical education is provided by the pharmaceutical industry. These numbers are staggering.
There are some parallels here with audiology and some things that are different. For example, the hearing aid industry does spend money to sell its products although it is nowhere near as much as what is spent in the pharmaceutical industry. We do have 25 to 30 percent of employees who are dedicated to marketing in the hearing aid industry. We do know that the interactions with audiologists and industry are fairly common and include gifts, free meals, subsidized travel to meetings and some other things. We also know that there are millions of dollars spent annually on continuing education activities. On the other hand, the magnitude of the problem is not anywhere near as great as pharmaceuticals, I do not believe. I do not have data to support that, but intuitively you cannot help but think that the magnitude is nowhere near as great.
An interesting difference is that in the pharmaceutical industry the representatives are typically business majors with experience in sales. In audiology, many of the manufacturer representatives are audiologists. They are one of ours; in many cases they are our colleagues and friends. This situation is significantly different from the medical and pharmaceutical industries.
The majority of the articles we will discuss today are surveys that looked at attitudes and perceptions, as well as behaviors. The first is Physicians in the Pharmaceutical Industry: Is a Gift Ever Just a Gift? (Wazana, 2000). This article was published in the Journal of the American Medical Association, and at the time there was controversy within the medical profession relative to conflicts of interest. That controversy is no different from what we see in audiology or other professions. There are some people in medicine that think all the focus on ethics and conflicts of interest is overkill when it should not be a big deal, while others advocate that it is a serious problem.
This particular article was written in response to these heated debates, in an effort to obtain information on the issues. The author wanted to know about the attitudes toward the physician-pharmaceutical relationship, and the impact of that relationship on the knowledge, attitudes and behaviors of physicians. To do this, he conducted a critical examination of the evidence by searching Medline for articles that were published between 1994 and 2000. He also had an Internet database that was searched by experts in this area to provide additional information. In total, there were 538 studies. We cannot even get close to that in audiology.
After criteria-based elimination, there were 29 peer-reviewed studies; these were studies that had analytic designs and used control groups in the analysis. Sixteen of the studies addressed physician-industry interaction, 16 identified attitudes toward the interaction, and 16 examined the effect of interaction on the practitioner. Of course some of the studies did all three and some did one or two. The author found that physician interactions with pharmaceutical reps were generally endorsed and, interestingly, began in the medical school. It is a learned behavior to some extent. It began in the medical school and it continued well into practice.
Gifts and number of gifts received correlated with the belief that gifts do not influence behavior. Drug-company-sponsored continuing medical education (CME) events preferentially highlighted their own drugs, and attending sponsored CME events and accepting funding for travel or lodging were associated with increased prescription rates of the sponsor's medication.
This was important because it was one of the early scientific studies used to show the negative impact associated with frequent physician-industry interactions. It demonstrated that the pattern of physician-industry interactions began in school, and that there is a need for the development of policies and guidelines for both associations and for medical schools.
The second journal article I selected to discuss today is entitled A National Survey of Physician-Industry Relationships (Campbell, Gruen, Mountford, Miller, Cleary, et al., 2007). It appeared in the New England Journal of Medicine in 2007. The authors wanted to know what physicians received from the industry, how often the physicians met with industry representatives and what characteristics are associated with the frequency and the nature of physician-industry relationships. They analyzed a survey of United States' physicians that was conducted by the Institute on Medicine as a Profession (IMAP). They surveyed 3,167 physicians in 6 specialties. They included within this large survey three questions that focused on physician-industry relations. One question specifically asked, "Which of the following have you received in the last year from drug, device or other medically-related companies?" The responses from which to choose included: food or beverage in the workplace; drug samples; honoraria for speaking; payment for consulting services; free tickets to sporting events; costs of travel, lodging and meals; payment for service or other expenses for attending meetings; and gifts received for prescribing practices.
Ninety-four percent of the physicians reported some type of relationship with industry. More than a third received reimbursement for meetings or continuing medical education (CME). More than a quarter received payments for consulting and lectures or enrolling patients in clinical trials. Lastly, physicians in solo, two-person or group practices met more frequently with industry representatives than physicians in hospitals or clinics.
This is an important study because it shows that physician-industry relationships continue to be very common in medicine. In fact, in this study it is more frequent than had been seen in previous studies. The relationships vary according to specialty. Pediatricians, for example, were significantly less likely to receive samples, reimbursements and payments than family practitioners. The industry appears to target opinion leaders. They go after people who can make a difference or a decision, like cardiologists. Many other physicians follow cardiologists in terms of prescription behavior. The study concluded that there is an urgent need to develop guidelines and recommendations specific to the context of each specialty and setting.
The next article is entitled Medical Students' Exposure To and Attitudes About Drug Company Interactions: A National Survey (Sierles, Brodkey, Cleary, McCurdy, Mintz, et al., 2005). They asked, "What is the frequency of medical students' exposure? What are the medical students' attitudes? How many medical students have policies regarding pharmaceutical representatives and students?" The group surveyed third-year medical students from eight different medical schools. The survey instrument sought demographic information, the student's perception regarding whether or not the school had a policy on physician-industry interactions, experiences with specific types of drug-related activities, and beliefs about whether their school had taught them anything about physician-industry relationships. They queried students about the frequency and nature of gifts, attitudes and their perceived appropriateness of drug-related gifts. They found that student interaction with industry was very common.
As a group, students appeared to be influenced by marketing strategies. More interestingly, most students believed that sponsored grand rounds and sponsored materials were helpful, that sponsored meals and textbooks were appropriate, that gifts could not influence them or their colleagues, and that students may be entitled to gifts because of financial hardship. Many students who perceived that gifts were inappropriate had actually received them, and of the eight schools, seven had no school-wide policies.
Seventy-seven percent of the medical students surveyed thought meals were appropriate. Seventy-one percent thought textbooks as gifts were appropriate. Forty-eight percent thought gifts less than $50 were appropriate. Why is this important? This study documents the nature and the frequency of physician-industry relationships in medical schools. Educational institutions play a very important role in student perceptions and attitudes towards physician-industry relationships. Schools need to set examples, and the faculty need to serve as role models.
Vandy Vignette: Judith Gravel
In each Vanderbilt Journal Club seminar, we take a quick break about halfway through the journal articles to share an audiology vignette from Vanderbilt University. Today, I am going to talk today about a lovely person by the name of Judy Gravel (Figure 2) who graduated from our program in 1985. I think that Judy was one of the best students to advance through the program at Vanderbilt University. She became a pioneer in pediatric audiology and, unfortunately, she passed away after a very courageous battle with cancer in 2008.
Figure 2. Judith Gravel, 1985.
In the 23 years from the time she left Vanderbilt University to the time that she passed on, Judy had authored more than 100 articles, chapters, and books. She had over 300 presentations in the U.S. and abroad. She had many of her presentations embedded in tutorial tapes. She served on 39 different task forces and was a consultant to the National Institutes of Health, Early Hearing Detection and Intervention, Maternal and Child Health Bureau and to corporations.
She was fiercely loyal to Vanderbilt University, and she served on our board of directors. The last time she was at Vanderbilt in 2008 she delivered the commencement speech to our graduate students within our program. The Outstanding Alumni Award carries the name now of Judy Gravel. We also have a scholarship that has been created in her name along with a lecture series. The inaugural lecture will take place in April 2012, and David Stapells will be the inaugural speaker to celebrate her incredible career. We loved her very much, and we miss her.
Journal Review Continued
Getting back to the journal articles, here is one that you all probably know about. It is Ethical Issues in Hearing Aids Revisited: A Survey (Hawkins, Hamill, & Kukula, 2006). It surveyed the attitudes and opinions of audiologists concerning the ethics of relationships with the industry. This is one of the few systematic articles that was conducted specifically with audiologists. They wanted to see if the attitudes and opinions of audiologists differed from that of consumers and if these attitudes and opinions changed since the original review in 2002 (Hawkins et. al).
The 2002 survey was used to collect information for the presidential task force on ethics and to get a sense of the frequency of contacts with industry and other kinds of conflicts of interest issues. The 2006 article included 182 audiologists and 42 patients. The authors administered a 17-item questionnaire via the Internet. The survey was completed by 1,633 members of the American Academy of Audiology. It described the professional business activities and the respondents were asked to rate the scenarios by the following categories: nothing wrong with this practice, may not be in the patient's best interest, borders on unethical practice and clearly unethical. Hawkins and colleagues published this information because they thought it would be useful in defining the activities that may present potential conflicts of interest to our membership.
The content areas included in this study were business incentives from hearing aid manufacturers, entertainment, small gifts and visits from manufacturers, continuing education events sponsored by hearing aid manufacturers, and business practices. They found a significant positive change in attitudes from 2002 to 2006 regarding the acceptability of activities such as gifts and cruises. In 2002 we saw some people that did not think gifts and cruises were a bad thing; there was a very definite change in 2006 where many more audiologists found that unethical, and I believe that is a direct reflection of the education and the efforts of the American Academy of Audiology.
In 2006, there is little concern about sales reps bringing small items and gifts such as pens and notepads. All groups expressed more concern about expenses paid for out-of-town company-sponsored workshops. Discrepancies in attitudes continue to exist between audiologists and consumers, and there are wide differences of opinion that continue to exist among audiologists on some practice behaviors.
There are several things I would like to point out. There were significant improvements from 2002 to 2006 in some of the major areas, especially those that involved business incentives such as credits redeemable for gifts or cruises. The audiologists who were surveyed in 2006 responded more like the consumers who responded in 2002. This is very encouraging and does show that things are improving along those lines. This survey is important because it is the only existing large-scale evidence-based study exploring conflicts of interest in our profession. It demonstrates that the Academy's efforts to educate members about potential conflicts is working and identifies target areas for future discussions and educational training.
Commodore Award
GUS MUELLER: During each Vanderbilt Audiology Journal Club session, we designate one article as receiving the Commodore Award, named of course after Commodore Cornelius Vanderbilt, whose generous gift in 1873 resulted in the founding of Vanderbilt University. In the opinion of our presenter, the article receiving the Commodore Award has particular importance, significance, relevance or is unique is some way.
FRED BESS: For the Commodore Award today, I have selected an article entitled Effect of Exposure to Small Promotional Items on Treatment Preferences (Grande, Frosch, Perkins, & Kahn, 2009) that appeared in the Archives of Internal Medicine. I think this article is very interesting as it looked at the influence of small items that you might think are of little significance and could not possibly impact treatment. In this study, they asked two basic questions: will exposure to small pharmaceutical promotional items result in more favorable attitudes toward marketed products? And, will a policy that restricts pharmaceutical marketing mitigate this effect? It is the first study that I know of that examined the small items from manufacturers. The researchers looked at third- and fourth-year medical students from two schools. One school, University of Pennsylvania, had restrictive policies that prohibited gifts, meals and samples. The other school, the University of Miami, permitted marketing practices.
The authors used a randomized experimental design. The subjects were randomly assigned to a control group or an exposure condition. The exposure group was exposed to Lipitor-branded promotional items prior to completing a computer-based study instrument. Exposures included Lipitor logos on a clipboard when the subjects signed in and on note paper used to assign the subjects' IDs. The controls completed the same procedure but with a plain clipboard and plain notebook paper. The researchers tried to determine if the exposure to branded products had an effect on attitudes toward two prescriptive statins, Lipitor and Zocor. The outcome measures were implicit, or unconscious, attitudes and explicit, or self-reported, attitudes. We will only focus on the implicit attitudes. To get those attitudes, the authors used what was developed in marketing and psychology as the Implicit Association Test (IAT).
Conceptually the IAT, as they call it, pairs targets like Lipitor and Zocor with attributes like pleasant or unpleasant, and then requires the subjects to sort corresponding images and words. Differences in reaction times when pairing targets and attributes are a clue to implicit attitudes. For example, a greater speed and reaction when Lipitor and "pleasant" are paired or when Zocor and "unpleasant" are paired reflects a more favorable attitude toward Lipitor than Zocor. You can log on to Harvard's Web site for yourself and try similar demonstrations.
The authors found that students in both class years at both schools demonstrated implicit attitudes favoring Lipitor. There were significant differences between the exposed and control groups among the fourth-year students. At Miami, fourth-years exposed to Lipitor promotional items demonstrated much stronger preference to Lipitor compared to the control group.
In contrast, students at Penn, who had a rather comprehensive policy on conflicts of interest, exhibited a weaker preference toward Lipitor compared to the control. This paper showed that subtle branding exposures can be important and are indeed influential. It also suggests that institutional policies can make a difference; their influences on student attitudes toward marketing can lead to different responses to branded promotional items. The authors concluded that guidelines and new policies are very beneficial. I wanted to mention that Vanderbilt does have a policy on this, by the way.
Conclusion
What can we take away from these studies? We can conclude that interactions of physicians and audiologists with industry appear to be prevalent. Frequent interactions with industry can potentially affect professional behavior and decision-making. Student experiences and attitudes suggest that they are at risk for unrecognized influence of marketing. Gifts, even small gifts, can influence attitudes. Attitudes and behaviors are developed in educational programs, and institutions need to take note of this. Finally, more audiologists today are concerned about potential conflicts with industry than ever before. Thank you.
References
Campbell, E.G., Gruen, R.L., Mountford, J., Miller, L.G., Cleary, P.D., & Blumenthal, D. (2007). A national survey of physician-industry relationships. The New England Journal of Medicine, 356, 1742-1750.
Hawkins, D.B., Hamill, T., & Kukula, J. (2006). Ethical issues in hearing aids revisited: a survey.
Hawkins, D., Hamill, T.A., VanVliet, D. & Freeman, B.A. (2002) Potential conflicts of interest as viewed by the audiologist and the hearing-impaired consumer.
Hornsby, B. (2012, January 9). 20Q: Hearing Loss, Hearing Aids, and Listening Effort. AudiologyOnline, Article 2409. Direct URL: www.audiologyonline.com/articles/article_detail.asp?article_id=2409 Retrieved May 1, 2012, from the Articles Archive on www.audiologyonline.com
Sierles, F.S., Brodkey, A.C., Cleary, L.M., McCurdy, F.A., Mintz, M., Frank, J., Lynn, D.J., Chao, J., Morgenstern, B.Z., Shore, W., & Woodard, J.L. (2005). Medical students' exposure to and attitudes about drug company interactions: a national survey. The
Wazana, A. (2000). Physicians and the pharmaceutical industry: is a gift ever just a gift? The Journal of the American Medical Association, 283(3), 373-380.
Audiology Practice and the Potential for Conflicts of Interest - Vanderbilt Audiology's Journal Club with Dr. Fred Bess
August 10, 2012
Related Courses
1
https://www.audiologyonline.com/audiology-ceus/course/ethical-business-practices-in-fitting-39188
Ethical Business Practices in Fitting Hearing Aids
The program of Ethical Business Practices is based on PCC (person-centered care) and helps the professional (audiologist, HCP, or HIS) determine the individual's best and most optimal hearing aid solution. The concept assists independent clinics not only in improving business results but also in creating clients through proactive word of mouth.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Ethical Business Practices in Fitting Hearing Aids
The program of Ethical Business Practices is based on PCC (person-centered care) and helps the professional (audiologist, HCP, or HIS) determine the individual's best and most optimal hearing aid solution. The concept assists independent clinics not only in improving business results but also in creating clients through proactive word of mouth.
39188
Online
PT60M
Ethical Business Practices in Fitting Hearing Aids
Presented by Oliver von Borstel
Course: #39188Level: Introductory1 Hour
AAA/0.1 Introductory; ACAud inc HAASA/1.0; AHIP/1.0; ASHA/0.1 Introductory, Related; BAA/1.0; CAA/1.0; Calif. SLPAB/1.0; IACET/0.1; IHS/1.0; NZAS/1.0; SAC/1.0
The program of Ethical Business Practices is based on PCC (person-centered care) and helps the professional (audiologist, HCP, or HIS) determine the individual's best and most optimal hearing aid solution. The concept assists independent clinics not only in improving business results but also in creating clients through proactive word of mouth.
2
https://www.audiologyonline.com/audiology-ceus/course/ethical-and-legal-requirements-audiology-30065
Ethical and Legal Requirements of Audiology Practice - Staying Compliant
In this presentation, we will discuss the foundations of ethics and compliance in audiology, as well as U.S. regulations. We will explore the AAA and ASHA Codes of Ethics, state ethical guidelines contained in several state licensure laws, Medicare, HIPAA, the FDA Guidelines, Stark laws, and Anti-kickback laws and explain, in detail, how they relate to specific scenarios we encounter in audiology.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Ethical and Legal Requirements of Audiology Practice - Staying Compliant
In this presentation, we will discuss the foundations of ethics and compliance in audiology, as well as U.S. regulations. We will explore the AAA and ASHA Codes of Ethics, state ethical guidelines contained in several state licensure laws, Medicare, HIPAA, the FDA Guidelines, Stark laws, and Anti-kickback laws and explain, in detail, how they relate to specific scenarios we encounter in audiology.
30065
Online
PT120M
Ethical and Legal Requirements of Audiology Practice - Staying Compliant
Presented by Kim Cavitt, AuD
Course: #30065Level: Intermediate2 Hours
AAA/0.2 Intermediate; ACAud inc HAASA/2.0; BAA/2.0; CAA/2.0; IACET/0.2; IHS/2.0; NZAS/2.0; SAC/2.0
In this presentation, we will discuss the foundations of ethics and compliance in audiology, as well as U.S. regulations. We will explore the AAA and ASHA Codes of Ethics, state ethical guidelines contained in several state licensure laws, Medicare, HIPAA, the FDA Guidelines, Stark laws, and Anti-kickback laws and explain, in detail, how they relate to specific scenarios we encounter in audiology.
3
https://www.audiologyonline.com/audiology-ceus/course/essential-ethics-in-audiology-today-37777
Essential Ethics in Audiology Today
Practical information on ethical conduct and decision making for audiologists. Specific topics include models for ethical decision making in health care professions, ethical provision of audiology services via teleaudiology, ensuring ethical behavior of audiology support personnel, and real-life case study examples of unethical behavior in audiology.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Essential Ethics in Audiology Today
Practical information on ethical conduct and decision making for audiologists. Specific topics include models for ethical decision making in health care professions, ethical provision of audiology services via teleaudiology, ensuring ethical behavior of audiology support personnel, and real-life case study examples of unethical behavior in audiology.
37777
Online
PT60M
Essential Ethics in Audiology Today
Presented by James W. Hall III, PhD
Course: #37777Level: Intermediate1 Hour
AAA/0.1 Intermediate; ACAud inc HAASA/1.0; AHIP/1.0; ASHA/0.1 Intermediate, Related; BAA/1.0; CAA/1.0; Calif. SLPAB/1.0; IACET/0.1; IHS/1.0; Kansas, LTS-S0035/1.0; NZAS/1.0; SAC/1.0
Practical information on ethical conduct and decision making for audiologists. Specific topics include models for ethical decision making in health care professions, ethical provision of audiology services via teleaudiology, ensuring ethical behavior of audiology support personnel, and real-life case study examples of unethical behavior in audiology.
4
https://www.audiologyonline.com/audiology-ceus/course/ethics-for-hearing-healthcare-professional-40089
Ethics for the Hearing Healthcare Professional
Professionals in the hearing healthcare industry must follow a code of ethics. Ethical guidelines can vary based on experiences and perspectives; therefore, each practice should create their own specific code of ethics. These should be based on professional organization’s guidelines such as the American Academy of Audiology or the American Speech-Language-Hearing Association. This course will discuss the difference between laws and ethics and the purpose of an ethics protocol in a hearing healthcare practice. An ethical decision-making matrix was also reviewed.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Ethics for the Hearing Healthcare Professional
Professionals in the hearing healthcare industry must follow a code of ethics. Ethical guidelines can vary based on experiences and perspectives; therefore, each practice should create their own specific code of ethics. These should be based on professional organization’s guidelines such as the American Academy of Audiology or the American Speech-Language-Hearing Association. This course will discuss the difference between laws and ethics and the purpose of an ethics protocol in a hearing healthcare practice. An ethical decision-making matrix was also reviewed.
40089
Online
PT60M
Ethics for the Hearing Healthcare Professional
Presented by Marjorie Klaskin, AuD, CCC-A
Course: #40089Level: Introductory1 Hour
AAA/0.1 Introductory; ACAud inc HAASA/1.0; ASHA/0.1 Introductory, Professional; BAA/1.0; CAA/1.0; Calif. SLPAB/1.0; IACET/0.1; IHS/1.0; Kansas, LTS-S0035/1.0; NZAS/1.0; SAC/1.0; TX TDLR, #142/1.0 Manufacturer, X
Professionals in the hearing healthcare industry must follow a code of ethics. Ethical guidelines can vary based on experiences and perspectives; therefore, each practice should create their own specific code of ethics. These should be based on professional organization’s guidelines such as the American Academy of Audiology or the American Speech-Language-Hearing Association. This course will discuss the difference between laws and ethics and the purpose of an ethics protocol in a hearing healthcare practice. An ethical decision-making matrix was also reviewed.
5
https://www.audiologyonline.com/audiology-ceus/course/eaudiology-ethical-considerations-in-audiology-40137
eAudiology: Ethical Considerations in Audiology Practice
This course is presented by members and a former member of the Academy’s Ethical Practices Committee (EPC). They review current ethical topics related to the audiology profession. Also included are discussions of ethical situations and the application of the Academy’s Code of Ethics to these hypothetical situations.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
eAudiology: Ethical Considerations in Audiology Practice
This course is presented by members and a former member of the Academy’s Ethical Practices Committee (EPC). They review current ethical topics related to the audiology profession. Also included are discussions of ethical situations and the application of the Academy’s Code of Ethics to these hypothetical situations.
40137
Online
PT60M
eAudiology: Ethical Considerations in Audiology Practice
Presented by Victor Bray, PhD, Alison M. Grimes, AuD, Devon Weist, AuD, Melissa Ferrello, AuD
Course: #40137Level: Intermediate1 Hour
AAA/0.1 Intermediate; Tier 1 (ABA Certificants)/0.1
This course is presented by members and a former member of the Academy’s Ethical Practices Committee (EPC). They review current ethical topics related to the audiology profession. Also included are discussions of ethical situations and the application of the Academy’s Code of Ethics to these hypothetical situations.


