Welcome. I am going to start by giving you a general background to bone-anchored hearing systems (BAHS). That is the terminology that we are using now to refer to any bone-anchored hearing system in the market today. The word Baha, Bone Anchored Hearing Aid, is a proprietary terminology that is owned by Cochlear Americas, so I will be using the generic term BAHS throughout my presentation.
I will address the candidacy criteria for BAHS and what hearing loss is indicated for this particular device. Then I will touch on some key design and technological features of the Ponto BAHS from Oticon Medical, as well as challenges we have seen and where we think this technology headed in the future.
Bone Anchored Basics
Let me start by going through some basic parts of the BAHS. A BAHS is a well-established hearing solution for conductive and mixed hearing losses and also for single-sided sensorineural hearing loss, commonly referred to as single-sided deafness (SSD). The bone-anchored hearing device has been used for almost three decades now for conductive hearing loss, mixed hearing loss, and SSD. It is based on two main principles: osseointegration and direct bone conduction. The device is composed of three main parts: a titanium implant, an external abutment, and a sound processor.
A sound processor is what many people regard as the hearing aid part of the whole system. The titanium implant is a screw or the fixture that a surgeon implants in the skull. The abutment is like a bridge that connects the sound processor to the titanium fixture or the implant in the skull.
Osseointegration is one of the principles on which the BAHS is based on. Osseointegration is where there is direct structural and functional connection between the living bone and the surface of a load-carrying implant, which is the titanium fixture. The titanium fixture fuses with the bone. That is a very unique characteristic of titanium. This osseointegration principle has been used in other implants as well, namely dental implants. The BAHS came later than dental implants, but we are using the same principles of osseointegration.
The other principle, direct bone conduction (Figure 1), is also very important because it is the principle that allows us to use this device for the types of hearing losses mentioned. There is stimulation through vibration from the sound processor onto the skull, and the vibrations are transmitted to the abutment, to the implant, to the bone, and then to both cochleae across the skull. Direct bone conduction gives very clear sound because the cochlea is a bony structure in the skull, so it is directly stimulated.
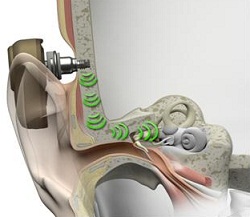
Figure 1. Pathway from the skull to the cochlea through direct bone conduction.
Pre-operative testing is possible with this method. We can see prior to a surgery how well patients might do post-operatively. There is high wearing comfort compared to alternative interventions. The surgery itself is very simple and straight forward, and takes, on average, about fifteen minutes.
Bone-Anchored Hearing System: Candidacy
The hardware of the bone-anchored system comprises the fixture and the abutment. The Ponto sound processor is worn on the outside of the head on the abutment. Lastly, there is the software that is used to fit the sound processor. Our platform is the Genie Medical Software.
In 2009, Oticon Medical Ponto was born. Prior to that time, there was only one other bone-anchored hearing system on the market, the Cochlear Baha. The Ponto System has a coupling system, which enables the sound processor to be attached to the abutment, which is like a bridge between the sound processor and the titanium implant.
This device, like any other, had to obtain Food and Drug Administration (FDA) clearance. In the United States, because this is an implantable device, it requires very strict FDA clearances. In 1996, this device was cleared to treat mixed and conductive hearing losses. In 1999, it was cleared for pediatric use in children age five and older. In 2001, it was cleared for bilateral fittings. In 2002, it was cleared for use in patients with SSD.
The FDA candidacy also stipulates that for mixed and conductive hearing losses, it should only be implanted on people five years or older. That said, for children under the age of five years, the sound processor can still be used on a head or soft band. So while it can be used in children, they have to be five years or older. The average bone conduction thresholds for people seeking bilateral implantation should be equal to or better than 45 dB HL and symmetrical between ears according to the FDA indications. This means that there should be less than a 10 dB difference at 500, 1000, 2000 and 4000 Hz on average or less than 15 dB at any one of those frequencies. That indication has changed somewhat since the introduction of more powerful bone anchored devices, which I will discuss later. Speech discrimination scores should be greater or equal to 60% correct.
Individuals with conductive and mixed hearing losses, as opposed to those with SSD, benefit most from a bone anchored hearing device. Candidates include those with chronic otitis media, congenital atresia, various cranio-facial syndromes such Treacher Collins and Goldenhar, cholesteatomas and middle ear dysfunctions and diseases. The hearing loss can be unilateral or bilateral.
If the air-bone gap is greater than 30 dB, then speech recognition is likely to be better with a bone-anchored hearing device than with a traditional hearing aid. The bone conduction thresholds should be up to, on average, 55 dB. The FDA indication states 45 dB, but we are able to fit up to 55 dB HL average bone conduction thresholds with the new Ponto Pro Power device.
Conductive and Mixed Hearing Loss
How do candidates with mixed and conductive hearing loss hear in the unaided condition? They often have bilateral hearing losses, and as such, significant difficulties following conversations. When fitted, these people become very dependent on the bone-anchored processors, one for both ears. They have not been able to function well in daily conversations, and they become dependent on the technology because of the audibility it restores.
These are often people with chronic otitis media, chronically draining ear, or external otitis, and they have a long track record of visits to the ear, nose, and throat (ENT) physician. There might also be candidates with microtia and atresia who can benefit from this device. These are all individuals with significant conductive hearing loss, but they tend to have normal cochlear function.
Figure 2 shows a picture of an audiogram with an air-bone gap and a representation of the skull and bone conduction pathway. The pink line indicates a gap between the air conduction thresholds and the bone conduction thresholds. If you have a bone-anchored device, the vibrations will be transmitted directly to the bone. In essence, you are bypassing the middle ear completely, and stimulating the inner ear directly. The air-borne gap is a nonissue if you have this device.
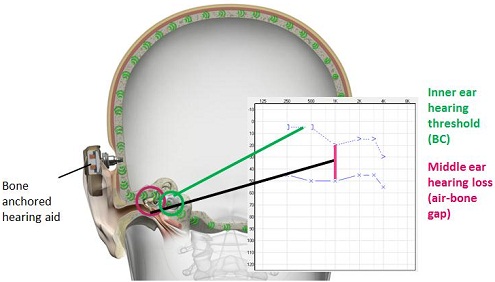
Figure 2. Illustration of the air-bone gap and the stimulation pathway of a bone-anchored device.
How does the bone-anchored device compare with traditional hearing aids? Traditional air-conduction hearing aids require higher gain because they have to overcome the air-bone gap. With the bone-anchored device, you do not need as much gain as a traditional hearing aid because you are bypassing the middle ear space where the air-bone gap occurs, and stimulate the cochlea directly instead. In most cases, very little gain is needed. You can achieve better sound quality because of direct bone conduction. Additionally, with normal ear anatomy, the ear canal remains open. So if there is a problem with draining ears, this is the best intervention to have because the ears can remain open, benefitting the patient on a medical level as well.
Another method that has been used for a long time to treat conductive and mixed hearing losses is the bone conduction hearing aid. It looks like the bone conduction vibrator we use in audiometric bone conduction attached to a metal headband. Compared to a traditional bone-conduction hearing aid, a bone-anchored device will not exert any pressure against the skin or skull because the implant resides on the skull. It is a percutaneous solution, which means it goes through the skin and directly to the bone, so there are no skin damping issues, either. Cosmetically, the bone-anchored device is going to be more appealing.
Compared to even a middle ear implant such as the MedEL Vibrant Soundbridge, we are able to pre-operatively assess the effectiveness of this device. We put the device onto a headband or softband and perform soundfield testing. Some skin damping occurs in this demonstration scenario, but patients are able to listen to the device and make some judgment as to whether they want the device or not. We can even do aided and unaided speech-in-noise testing in the soundfield to look at post-operative benefit and outcomes with this device.
Single-Sided Deafness
The two most common etiologies of SSD are acoustic neuroma and sudden hearing loss. There are other causes also, such as neurological degenerative diseases, genetics, ototoxic treatments, inner ear malformations and traumas.
The typical audiometric profile of SSD can be seen in Figure 3. There is normal hearing in one ear, defined as average air conduction thresholds of 20 dB or better. There is a profound sensorineural hearing loss in the other ear.
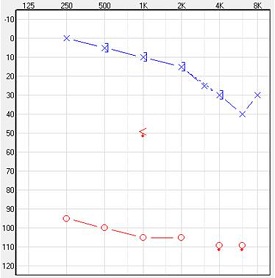
Figure 3. Audiometric data of an individual with SSD in the right ear.
How do candidates with SSD hear in the unaided condition? Some patients report that following a conversation in quiet is not a problem. Listening to the radio and TV is not an issue. Some can even attend concerts and theater shows comfortable. What is challenging, however, is understanding speech in group conversations and in noisy environments. These people may have difficulties hearing sounds directed to the deaf side for obvious reasons. There are also difficulties localizing sounds because the binaural capability has been compromised, and they are only getting input from one cochlea.
The benefit of a bone-anchored device in SSD was first reported in 2000 in just two patients (Vaneecloo, Hanson, Laroche, Vincent, & Dehaussy, 2000). But a year later, data was reported on 29 patients (Vaneecloo, Ruzza, Hanson, Gerard, Dehaussy, et al., 2001). In 2003, Niparko, Cox and Lustig published a study that compared the effectiveness of a bone-anchored hearing system with a traditional CROS (contralateral routing of signal) hearing aid. It was shown that head-shadow effect was compensated for, and patients were able to localize better with the bone-anchored device and that speech discrimination scores were improved. Another study (Wazen, Spitzer, Ghossaini, Fayad, Niparko, et al., 2003) compared a bone-anchored hearing device to a traditional CROS hearing aid, showing that the bone-anchored hearing device provided better speech discrimination in noise.
For SSD, the bone-anchored device can function as a CROS aid, but based on the principle of direct bone conduction. One cochlea is completely impaired and is unaidable with a traditional hearing aid, but there is still one functioning cochlea on the opposite side. The bone-anchored device stimulates both cochleae, so the normally-functioning cochlea on the contralateral side will be able to pick up the sound from the impaired side. In this way, it works like a CROS hearing aid, except sound is delivered directly to the good cochlea via bone conduction.
The advantages of the bone-anchored sound processor for people with SSD are that it reduces the head-shadow effect and increases speech understanding in noise. Compared to an air-conduction CROS hearing aid, again, the ear canal now remains open. Some of the older CROS hearing aids had cords connecting the two sides, and they were cumbersome. Today, we have very advanced CROS hearing systems that are wireless, so that is not as much of an issue, but there are still two devices that are worn on or in the ears.
What is the benefit of using the BAHS to those patients going unaided with SSD? A recent study (Ortega, Draper, Wazen, Thompson, & Daugherty, 2011) compared the Cochlear BP100 and the Oticon Medical Ponto Pro to an unaided condition using consonant-nucleus-consonant (CNC) monosyllables and the Hearing in Noise Test (HINT). Speech discrimination scores were higher using the bone-anchored devices than without, no matter which device was used, the BP100 or the Ponto Pro. Patients are going to be compromised on speech discrimination in the unaided condition, whether they are in quiet or in noise.
Design Features of the Ponto BAHS
Design is not just how the device looks. It is how it works. Design is an important aspect of the performance of the device. We do have to look at the cosmetics of the device, but more importantly, we have to look at how it is going to function to serve these patients. There are so many features that have to be considered (Figure 4): the microphones, the volume control, the push button, mute button, safety line attachment, battery drawer, and the coupling system. They are all very important.
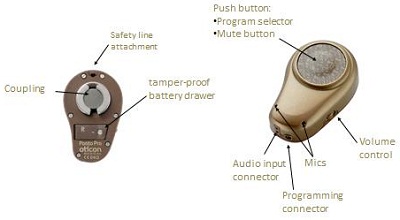
Figure 4. System components of the Ponto processor.
The Ponto system is designed with the users in mind. For one thing, it is shaped to blend in. It looks like the ear. We even have left and right dedicated versions. It is not a simple box that is the same for the left or right side. It is very easy to use because you the program buttons are big and easy to find, and the features are fully automatic. The Ponto even has a self-learning volume control. After using the device for a week, the device will memorize the volume settings in different environments. When the user moves into an environment that the device recognizes, it automatically changes the volume to a learned level set by the user that is appropriate for the patient in that situation. It also has a low battery warning system.
Nano coating is also available in the Ponto Pro and the Pronto Pro Power processors. It protects the processor from moisture and dirt. More importantly, it seals all the narrow slits between the plastic parts in the product. That protects the outside and the inside of the sound processor, making it more durable and reliable. It enables reliability for a longer time, especially in humid climates and environments where there is going to be a lot of dirt.
The Ponto bone-anchored sound processors are optimized for cell phone use. There is no interference whatsoever. They are fully compatible with cell phones and other wireless phones, so people using this device can enjoy all the wireless communication they need. There is hardly any distortion or noise when using the device with wireless phones.
It can also include a tamper-resistant battery door. That is an important feature because there were reports of another model's battery doors being broken regularly, which can be hazardous, especially in the pediatric population. Ours is tamper resistant, vigorously tested and used by Oticon. We have been getting a lot of positive customer feedback on this feature.
The transducer impact protection is another good feature to have (Figure 5). That boils down to the design of the coupling system. When the device is attached to the abutment, we tend to exert quite a bit of force on it. The coupling system itself is designed in such a way that it can take quite a significant load, without affecting the vital parts inside the sound processor. The coupling protects the vibrator from strong forces when connecting the processor.

Figure 5. Ponto coupling system to avoid excess force on the processor.
As I mentioned, there were pediatric considerations made in the design of the Ponto device also. We have a dedicated softband fitting mode in the fitting software. When clinicians go into Genie Medical software and check softband, the different settings in the device will be programmed automatically. We have adapted Ponto to fit children's needs. We have special programs for them, and we have the flexibility to deactivate some of the controls. They are also designed with school in mind, and they are fully compatible with the Oticon Amigo FM system or other systems through the audio input connector. We included safety features in the Ponto such as the tamper-resistant battery door and the safety line so that if it comes off the head, it is still attached to the child's clothing. We also provide stickers so the children can personalize their processors (Figure 6).

Figure 6. Pediatric stickers on the Ponto device.
The ease of use of the device, how it device looks, how easily it can be fitted by the clinician, and how it is being used by the user are very important considerations. An independent study out of Denmark (Olsen, Glad, & Holme-Nielsen, 2011) looked at the ease of device handling of the Ponto and the BP100. They examined four parameters: changing the battery, putting the device on, adjusting the controls, and appearances. They found that the user ratings for the Ponto Pro came out higher compared to the BP100 on all four parameters. As far as changing the battery, adjusting the controls and appearance, the Ponto Pro came out statistically better in this study.
Technology
The Oticon Ponto, Ponto Pro and Ponto Pro Power are all three based on the most advanced platform we have today for hearing instruments, which is the RISE platform. All these devices can be fitted very easily using the Genie Medical software. If you can fit one patient, you can fit them all. The three devices represent a very powerful family. All of them have advanced signal processing features in them. The three features on which I would like to focus are advanced directionality, the Speech Guard amplification system and two-stage feedback management. Some of you who are familiar with Oticon hearing instruments may be familiar with Speech Guard amplification, and the principles here are the same. We were able to incorporate the advanced features of the Oticon hearing aid family, such as Speech Guard, into our Ponto processors.
The directional system we have in these processors is automatic and adaptive. It is available in three modes and across four channels. The patient does not have to manually select the directional mode; the device does it automatically. Additionally, within the directional mode is a split-directional mode. The split mode keeps the low-frequency bands in omni-directional mode while the high-frequency bands are in directional mode. This is useful in situations where you do not need to use full directionality. When you use full-directional mode, many of the low frequencies are lost. We can keep some of the low frequencies to make it sound more natural. For example, if you are in a café with three or four people and there is not a lot of noise, you can benefit from directionality by being in the split mode. If you are at a cocktail party, on the other hand, you really need a full-directional system in order to understand speech to the fullest extent.
With the automatic multiband adaptive directionality, you can automatically change between the programs without having to manually manipulate the push button. There is more directionality available to you, and you would have better speech understanding in difficult listening situations.
The Speech Guard amplification system does exactly what the name says: it guards the speech. It it designed to protect and preserve the intricate details and nuances of speech, making speech very natural. It will preserve the speech integrity by being mostly in a linear processing mode. We know, historically, that we can preserve speech integrity better in a linear mode as opposed to applying traditional compression. The effectiveness of this signal-processing feature has been very well studied in research and published.
I would like to move on to the two-stage feedback management system we have. Feedback can occur due to two reasons in bone-anchored sound processors. First, there is feedback due to static conditions, like the vibrations radiating from the skull back into the processor. That is something very unique to bone-anchored processors. There can also be feedback due to dynamic conditions, and that is also applicable to hearing aids when people move their head, hug another person or change to a listening environment that requires more power. Dynamic conditions change often or occur more suddenly.
We have to address feedback due to static conditions before we can even talk about dynamic feedback. We have a way of addressing static feedback. First, we have to measure individual feedback limits. For every patient that we see, we measure his or her own feedback limit, as these limits vary from person to person. If we measure these individual feedback limits within the software, we can enhance the capacity of the dynamic feedback cancellation system. The dynamic feedback cancellation system in bone-anchored processors will only work if we have taken care of the static feedback first. We cannot have a good dynamic feedback cancellation system if we have not addressed static feedback first.
When we measure individual static feedback limits, we can set the maximum gain across all the frequencies, and then we will not exceed that static feedback limit. The maximum gain will be set at that feedback limit or lower. That way, we can allow the dynamic feedback cancellation system to work well. Figure 7 is a screen shot of the Genie software. The black curve is the static feedback limit, and the red or blue line is the gain. The gain will never exceed the black line. You can go up and up and it, but it will not exceed that black line.
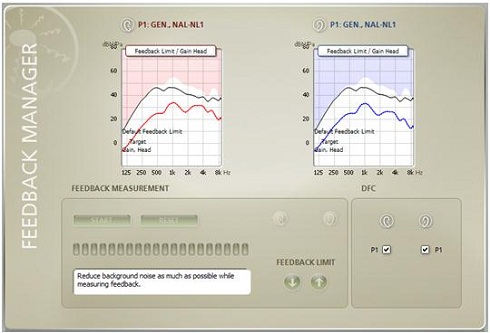
Figure 7. Fitting screen showing the feedback management settings. The black curve indicates the static feedback measurement, and the red and blue fitting curves indicates the gain.
Another feature in all the Ponto sound processors is wind noise reduction. This is useful when using the device in windy places like Chicago, for example. When wind crosses the microphone it creates a lot of noise, and patients find that very annoying and will take the processors off. With the wind noise feature, they do not have to take the processors off. The device will detect wind by virtue of the input into the two microphones. It will do a correlation analysis to see what the inputs are like, and will adjust the gain accordingly. The wind noise feature will go into omni-directional mode and turn the gain down. The more wind that comes into the device, the greater the gain reduction will be.
Current Challenges & Future Directions
One of the current challenges we have is the lack of understanding of SSD. Patients with mixed and conductive hearing losses benefit readily from this device, and so can those with SSD. However, SSD cases are often more challenging than the mixed or conductive hearing loss groups. They still come back and report that they do not understand speech the way they would like. It might have to do with expectations, but I think there is a general lack of understanding of SSD. There needs to be more systematic research to identify what the needs and challenges of this group are and what we can do to best address their needs.
There is also a lack of standardized evaluation tools for the bone-anchored world. That is true for all hearing loss categories. We have been taking tools from the hearing aid world and transplanting them into the bone-anchored world. We are not sure if that is the right way to do it, but we need more evidence-based studies to look at what tools might be appropriate for mixed hearing loss, conductive hearing loss, and SSD for adults versus children. Clinicians depend on evidence, and this is one area that needs more work.
We currently have a lack of appropriate objective methods to demonstrate clinical effectiveness and to verify output. Some of the research projects in which I have been involved show a lack of agreement between the objective test data and the subjective test data. One example is the localization ability in SSD. Objective methods that have been used to study localization have shown that this device does not really enhance localization ability. On the other hand, subjective tools such as the Abbreviated Profile of Hearing Aid Benefit (APHAB) and the Speech, Spatial and Qualities of Hearing Questionnaire (SSQ), in particular, are showing us that these patients are reporting better localization skills with the bone-anchored device. There is a disconnect between the objective and subjective methods. While many papers are published in this area, there is generally a lack of good quality research. Sample sizes have been very small, and there have not been good controls in the studies. Better quality studies in this area will give us better evidence on which we can base our clinical judgments.
What Can We Do?
I think we need to try and establish best-practice guidelines. That is obviously not something we can do overnight. It is a process. It is going to take months, if not years. However, most clinicians today agree that there is a lack of standard procedures. How do we counsel these patients? How do we evaluate them? How do we predict post-surgical outcomes, both in audiological and non-audiological outcomes? There is a consensus that we need better testing methods, better evaluation tools, more research and better evidence so that patients are served optimally.
In February 2012, Oticon Medical gathered a dozen audiologists from across the country to meet in New Orleans and discuss issues pertaining to counseling, pre-surgical evaluation and post-surgical verification. All the clinicians in attendance agreed that there is lack of a standard of care. Everyone is doing things differently. There is a lot of variation between clinics. They all agreed that we need to work on some sort of uniform protocol. As a result of this meeting, the American Academy of Audiology (AAA) will be driving a taskforce and establish best-practice guidelines down the road. This is an initiative that has to come not from the industry, but from the audiological community and the professional association working with the audiological community. Michael Valente from Washington University School of Medicine is initiating this by creating the taskforce and that will work through AAA in the process.
The Future
We need better clinical tools to evaluate the patients. Many clinicians working in private practice are pressed for time, and they want tools that they can use quickly and easily. We need more data, not just on audiological outcomes, but also non-audiological and non-acoustic outcomes like quality-of-life data. That is missing from all the studies that have been done in this area. Few studies that looked at quality-of-life measures, and almost all of those were conducted in Great Britain.
There should be more studies reporting quality-of-life changes in these patients not just immediately after surgery, but six months, twelve months, and even a couple of years post-surgery. We need better controlled systematic studies to demonstrate clinical effectiveness. All of that ties in with the other points that I mentioned, namely establishing best-practice guidelines for clinical practice.
Thank you very much for listening in. Should you have any questions, please feel free to email me at [email protected]
Question & Answer
What are your thoughts on fitting infants with SSD with bone-anchored devices?
The FDA regulations stipulate that only children five years or older can be implanted, but there have been cases where children as young as two years of age and even younger have been implanted. Children have thin bone, and often the surgery is done in two stages to maximize osseointegration. The loading of the device onto the abutment typically would not take place until five or six months after implantation. There is more research being performed now on children, but there is not enough evidence yet for us to say that this is a safe and effective method of amplification for infants and children with SSD.
What do you see to be the direction of technology for bone-anchored devices?
I think there are still things that could be done to improve bone-anchored technology. We have very advanced features now, but I think we can still look at wireless capabilities in these devices. We could also look at partially-implantable technologies, moving away from percutaneous to a transcutaneous solution. We will see various technological advances coming through in the next few years or so. There is always room for advancement.
Are the wireless technologies difficult to implement?
Yes, they are difficult. We are dealing with a vibrational technology here. Anything that we bring from the hearing aid world to the bone-anchored world is not as straight forward as many people would like to think. Bone anchored processors are not actually hearing aids as we think of them, even though the name implies it. However, the algorithms are much different and how acoustics translate into vibration is a challenge and requires a lot of work. So I think wireless will happen, but it is not as easy as taking the technology from the hearing aids and putting it in the bone-anchored device.
What about patient indications for bone-anchored systems?
This is a very good question and something that we are looking at from a research perspective that will have huge clinical implications. For SSD, the FDA stipulates that one ear has to have normal hearing, defined as 20 dB or better air conduction thresholds. The impaired ear may not have any hearing or at least no aidable hearing. However, increasingly more patients with some degree of hearing loss, mild hearing loss, mild to moderate hearing loss or even moderate hearing loss in the better ear, are being fitted with this device, and they have been benefiting from it. We need more research, though. If evidence shows that more and more patients are going to benefit from the device, then we would like to see the FDA indications changed to be more inclusive.
We also have a power device that will provide enough amplification for average bone conduction thresholds of 55 dB. That is moderate hearing loss. If patients have 55 dB bone conduction thresholds in the contralateral ear and the other ear is completely impaired, they can still benefit from a bone-anchored device. We need to collect more evidence from those patients as well.
We have the ability to provide amplification to many patients, and there are many who are going to be in their 60s and even late 50s and will experience a hearing decline due to presbycusis. They may have normal hearing today, but two years down the road they may have a mild to moderate sensorineural hearing loss in the better ear. So would they not benefit from this device? Yes, they would. As I said, even if the hearing loss dips down to 55 dB, they might still benefit from it. We simply need more evidence in this area for several patient subsets so that we can help educate the FDA on possible revision of the fitting indications and criteria. We simply need more evidence in this area for several patient subsets so that we can revise the FDA fitting indications and criteria in the future.
References
Niparko, J.K., Cox, K.M., & Lustig, L.R. (2003). Comparison of the bone anchored hearing aid implantable hearing device with contralateral routing of offside signal amplification in the rehabilitation of unilateral deafness. Otology & Neurotology, 24(1), 73-78.
Olsen, S., Glad, H., & Holme Nielsen, L. (2011). Comparison of two bone anchored hearing instruments: BP100 and Ponto Pro. International Journal of Audiology, 50, 920-928.
Ortega, C., Draper, K., Wazen, J., Thompson, J., Daugherty, J. (2011, March). Performance of the Ponto Pro and BP 100 processors in patients with single sided deafness (SSD). Presentation at the Third International Symposium on Bone Conduction Hearing - Craniofacial Osseointegration. Sarasota, Florida, USA.
Vaneecloo, F.M., Hanson, J.N., Laroche, C., Vincent, C., & Dehaussy, J. (2000). Prosthetic rehabilitation of unilateral anakusis: study with sereoaudiometry. Annales d'Oto-Laryngologie et de Chirurgie Cervico Faciale, 117(6), 410-717.
Vaneecloo, F.M., Ruzza, I., Hanson, J.N., Gerard, T., Dehaussy, J., Cory, M., Arrouet, C., & Vincent, C. (2001). The monaural pseudo-stereophonic hearing aid (BAHA) in unilateral total deafness: a study of 29 patients. Revue de Laryngologie-Otologie-Rhinologie, 122(5), 343-350.
Wazen, J.J., Spitzer, J.B., Ghossaini, S.N., Fayad, J.N., Niparko, J.K., Cox, K.M., Brackmann, D.E., & Soli, S.D. (2003). Transcranial contralateral cochlear stimulation in unilateral deafness. Otolaryngology Head & Neck Surgery, 129(3), 248-254.

