Editor's Note: This is a transcript of the live seminar presented on October 17, 2011. To view the course recording, register here.
A pdf of Dr. Bankaitis' slide presentation from the live seminar is available for download here (PDF).
Welcome to today's presentation, Infection Control Part II: What Audiologists Need to Do. As indicated by the title, this is the second of a two-part infection control talk. The first course, Infection Control Part I: Why Audiologists Need to Do It, is available via the AudiologyOnline library as a recorded course and as a text course. If you have not yet viewed Part I, you are still going to benefit from the information presented in today's course. Because today's presentation builds upon what was reviewed in Part I, I do encourage you to take the time to view it in the future. My contact information will also be posted, so please feel free to contact me at any time with any questions or concerns that may develop in the future as it pertains to infection control.
The specific objectives of this presentation are as follows: first and foremost to review the required components of a written infection control plan; secondly, to build upon this concept of work practice controls that were initially introduced in the infection control Part I; third, to discuss key points associated with selecting appropriate products to use in the clinic for the purposes of minimizing the spread of disease; and finally, I want to put it all together so you are aware of resources readily available to you and help you implement a comprehensive written infection control plan in your clinic.
Is a Written Plan Necessary?
Let's start with our first objective which is reviewing written requirements. A fair question to ask would be if a written infection control plan is really necessary. The short and very succinct answer to that question is, yes. Infection control is a federal mandate that is overseen by the Occupational Safety and Health Administration (OSHA), which is the federal regulatory agency responsible for overseeing safety in the workplace. Infection control does fall under this umbrella of safety as dictated by OSHA, who has specific guidelines as to how to minimize the potential spread of disease in the workplace. So audiologists, audiology technicians, hearing instrument specialists, students and the like are obligated to legally and ethically uphold the standards that have been outlined by OSHA.
Despite the fact that a written infection control plan is the law, it is also a federal requirement. I often wonder how many of us have a written infection control plan that is readily accessible in our clinics. Amlani conducted a survey in 1999 and asked practicing audiologists whether or not a written infection control plan was in place in their clinic. A decade ago, the majority of clinicians responded "no" or "I don't know" with another 44% indicating their clinic did have a written infection control plan in place (Figure 1).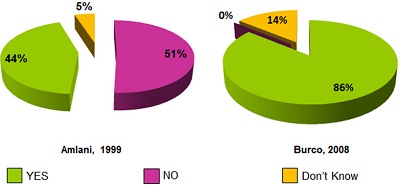
Figure 1. Comparison of survey data collected in 1999 versus 2008 in answer to the question, "Is there a written infection control plan in place in your clinic?"
If we fast forward about 10 years, Burco (2007) essentially repeated Amlani's survey (1999) as an Au.D. student at Central Institute for the Deaf within Washington University School of Medicine in St. Louis. When she asked practicing audiologists the very same question, an overwhelming majority of 86% reported that they were, indeed, aware of a written infection control plan for their clinic. So in contrast to what Amlani (1999) found, none of Burco's (2007) respondents indicated they did not have a plan, and only 14% indicated they did not know (Figure 1). This is a huge change from just 10 years ago. In my mind, this suggests one of two things: either we have made a lot of improvement in educating audiologists about infection control requirements and they are being written and implemented, or we are making a lot of improvements in educating audiologists about infection control requirements, and we are simply getting better at knowing how to answer the question correctly.
The reason that I say this is because about a year ago I personally received a call from a private practice that communicated to me they had heard an infection control talk that I conducted. They were aware of the requirement of needing to have a written infection control plan in place and they wished they had acted upon it. To make a long story short, OSHA showed up on their doorstep one day unannounced and asked the owners to see their written infection control plan. When the written plan could not be produced because it did not exist, two immediate things happened. Number one, the private practice was audited by OSHA to assess global infection control standards, and number two, the practice was initially fined $5000 for not having a written plan in place. The fine was actually reduced to $500 under the contingency that the clinic would produce a written document within two weeks. The amount of anxiety and stress that occurred during this process was immeasurable. This story illustrates that it is not enough to know what the requirements are; you need to act upon them.
Written Infection Control Plan
As previously mentioned, OSHA requires each facility to have a written infection control plan. This written infection control plan is the cornerstone of all infection control programs, and it contains the following requirements:
- Have an employee exposure classification
- Hepatitis B vaccination plan
- Plan for annual training and recordkeeping
- Plan for accidents and accidental exposure follow up
- Implementation protocols
- Post exposure plans and records
Of the six required elements of the written infection control plan, five are generic. Section 5, implementation protocols, is that section of the infection control plan that outlines work practice controls specific to each clinic. This section will differ from infection control plan to infection control plan because it contains written procedures that reflect not only the scope of services that are provided by a particular audiology or hearing aid clinic, but also the procedures that are consistent with the adoptive infection control philosophies of that clinic. These dictate how a clinic will be performing clinical procedures based on their specific approach to minimizing the spread of disease. This will become clearer once we start reviewing examples of different work practice controls later in the presentation.
First, let's review the basic infection control requirements that are universally similar across all infection control plans. Once we review sections 1, 2, 3, 4 and 6, we will go back to review the written requirements falling under the category 5 of implementation protocols.
With regard to employee classification, each employee must be classified into a category on the basis of their potential risk of exposure to blood and other infectious substances. A classification category must be assigned for each employee and documented in writing within the infection control plan. The three classification categories to which employees may be assigned to are as follows: category No. 1 is when the primary job assignment involves exposure to blood or other bodily fluids on a regular basis; No. 2 when the personnel has a secondary job assignment that involves exposures to blood or other bodily fluids on a regular basis; and No. 3 for those personnel whose job assignment does not involve any exposure to blood or bodily fluids.
With regard to audiology, those audiologists who are actively involved in intraoperative monitoring or post surgical audiological assessments as their mainstay of employment meet the classification category of 1. Most audiologists and hearing instrument dispensers involved in handling hearing aids, earmold impressions, cerumen removal, and cleaning, disinfecting and sterilizing instruments meet the employee classification category of 2. Finally, any audiologist who is an administrator or any front office staff who does not have patient contact will be classified as a category 3.
Now that we understand the employee classifications we will take a look at the second requirement of a written infection control plan which is the Hepatitis B vaccination plan. Employees who have the potential for encountering blood or other infectious substances must be offered the opportunity to receive a Hepatitis B vaccination (HBV). The HBV must be made available to all category 1 and category 2 workers free of charge. The employee is not required to accept the offer of the vaccination; however, it would be a requirement for the employee to sign a waiver noting the refusal of the offered vaccine, and that refusal has to be placed in the infection control plan records. The vaccination obviously needs to be administered by trained medical professionals and given according to pursuant medical standards.
With regard to training, each office is responsible for conducting and documenting the completion of annual training in infection control. Training must be provided at the time of initial assignment and take place at least annually thereafter. While the standard does not specify the length of training, OSHA standards do list elements that must be incorporated in the training program including modes of transmission, information on the HBV vaccine, use of protective equipment and the like. Anyone providing audiological services must have initial training within 90 days of hire. It is preferred that that training occur prior to the provider ever seeing a patient to ensure that they are practicing in a manner consistent with your infection control standards.
As mentioned, OSHA requires annual training. Annual training can be as short as a 10-minute update. The initial training is probably more important and can last as long as it needs to fully train the individual. Additional training also has to be conducted in a timely manner. Any time an audiologist is reclassified, for example from a category 2 to a category 1, they have to undergo infection control training within 90 days of that reassignment. Then finally, any time there is a new or updated procedure that is going to be implemented in the clinic, it is necessary for everyone to undergo training so that everybody knows how to conduct that procedure in a manner consistent with the established infection control guidelines. It must also map out a plan for what to do in the event of an accident. This includes steps that will be taken when accidents occur which can expose individuals to bloodborne pathogens or other infectious agents. Accidents include a patient falling, getting a nosebleed, somebody who vomits, et cetera. Every member of the office staff needs to know what to do and how to do it.
The final generic written infection control plan requirement addresses post exposure evaluation and follow up. Accidental exposures to bloodborne pathogens do require follow up. While these may be relatively rare in the audiology clinic, an emergency plan should be created. As dictated by OSHA, if the exposure involves a percutaneous or mucous membrane exposure to blood or other bodily fluids or cutaneous exposure to blood when the worker's skin is chapped, abraded or otherwise broken, a specific follow up evaluation must occur. The goal of this follow up is to confirm that a disease has or has not been transferred, and in the event of a transfer to treat the disease more effectively and efficiently.
We have quickly reviewed five of the six requirements of a written infection control plan that, for the most part, are going to be identical across different clinics. Since section 5 includes the written procedures that outline how clinical services will be executed in a manner consistent with controlling the spread of potential disease, the content appearing in this section will differ from one clinic to the next and, therefore, from one infection control plan to another. Because the content is going to be unique to your specific work environment, we need to spend more time on dissecting implementation protocols.
Implementation Protocols
Content appearing in the implementation protocol section of your infection control plan will be dictated by the scope of services provided by your clinic. For example, if the clinic where you work dispenses hearing instruments, the implementation protocol section of your infection control plan must encompass any and all services that are related to hearing aid dispensing that your clinic provides. This includes, but is not limited to, making earmold impressions, modification of earmolds or hearing instruments, listening checks and the like. If the scope of your clinic's practice includes routine diagnostic audiometry, such as otoacoustic emissions, vestibular testing, intraoperative monitoring, or cerumen removal, your infection control plan must include written procedures that outline how all those specific tests or procedures will be performed in a manner consistent with controlling the potential spread of disease.
This brings us to the second objective of this presentation, which is a review of work practice controls. As part of an exercise assigned during my Infection Control Part I webinar, viewers were asked to make an exhaustive laundry list of what procedures they performed for their patients. The reason the assignment is critical is that it will identify the specific work practice controls that you will need to develop and include in the implementation protocol section of your clinic's specific written infection control plan. Recall that a work practice control is a profession-specific procedure that will outline how we as audiologists will perform that procedure with the specific goal of eliminating or minimizing the spread of disease. For those audiology clinics located within larger healthcare institutions, I will tell you a written infection control plan containing the majority of the required written elements is most likely already filed with the specific regulatory division in the hospital that oversees infection control; however, that filed plan will not cover audiology-specific procedures. In other words, the general hospital plan will contain parts 1, 2, 3, 4 and 6 but they are not going to contain the work practice controls that fall under the implementation protocol section. According to OSHA, each plan must include these work practice controls. Audiologists are going to be required to develop these protocols since we are the professionals who actually provide these specific services.
Standard Precautions
These work practice controls are written procedures that are based on standard precautions. Standard precautions refer to a list of five guidelines that have been issued by the CDC that are intended to minimize the spread of potential disease in healthcare settings. We will address each one individually and how they apply to the audiology clinic.
Personal Barriers
When we talk about personal barriers we are referring to gloves, safety glasses, disposable masks, as well as disposable gowns. Within the confines of the audiology environment, gloves must be worn when you are immersing or removing instrument from a cold sterilant. In the event instruments are not clean and disinfected first, gloves should be worn during modification procedures. Gloves should also be worn when handling hearing instruments received from patients, during removal of earmold impressions and handling of earmold impressions that have been removed from the ear as well as any time you or your patient exhibits visible wounds in areas that may make direct or indirect contact with another person.
Safety glasses, as well as disposable masks, should be worn when using the buffing wheel during hearing aid modification procedures, even if the hearing instrument or earmold surfaces have already been cleaned and disinfected. This not only keeps you from breathing in or making contact with any microbes that potentially reside in the hearing instrument surfaces, but it also protects you from breathing in or being exposed to particles that already reside on the buffing wheel or grinding stone. Those things are almost impossible to clean and disinfect.
Hand Washing
The second guideline encompasses hand washing, also referred to as hand hygiene. Hand washing represents the single most important procedure for effectively limiting the spread of disease. It is one of the most critical components of a basic infection control plan. Hands need to be washed before and after every patient contact, after glove removal and as needed. The number of times hands must be washed during patient appointments is going to depend on what you are doing during that appointment. In terms of patient appointments, hands must be washed immediately prior to the patient appointment, preferably in front of the patient, or they should be washed at any time during the same patient appointment that you feel it is necessary. Once the appointment is complete, hands must be washed as well. In the event you will be seeing another patient immediately following the previous patient appointment, washing your hands before the next patient will actually fulfill the requirement of washing your hands after the last patient.
Since the use of gloves indicates that you are likely involved in a procedure that may potentially expose the hands to microbial contamination, bodily fluids or secretions, it is possible to knowingly or unknowingly come in contact with contaminants during glove removal. This is why it is a requirement to wash your hands immediately following glove removal. You can wash your hands any other time as you feel is necessary. One of the interesting questions to ask is, "Do we as audiologists actually wash our hands when we should?" According to Amlani (1999), when asked if hands were washed after each patient as well as after the use of the bathroom, which are both CDC requirements, only 26% of practicing audiologists admitted to washing their hands after each patient, and only 50% washed their hands after using the restroom facilities. Burco posed the same question in 2007 and found that 82% of audiologists washed their hands after each patient, and 87% reportedly washed their hands after using the bathroom. While the numbers have improved over the last 10 years, we have yet to reach a point where we are in full compliance with the current CDC requirements when it comes to washing our hands.
Touch and Splash Surfaces
The third standard precaution says that touch and splash surfaces must be pre-cleaned and disinfected. There are several different terms we need to define here. A touch surface refers to any surface area that comes in regular contact with hands, mainly those horizontal surfaces such as countertops, tables or the armrest of a chair. Splash surfaces refer to any surface area that may be exposed to secretions when, for example, a patient sneezes. So it can be the same types of surfaces. Prior to providing a definition of what the terms clean and disinfect mean, Amlani (1999) surveyed practicing audiologists to determine whether or not they knew the difference between the two terms. Interestingly, 74% of the respondents indicated that they did know the difference between the term clean and the term disinfect; however, when follow-up questions specifically asking the same audiologists to identify the correct definition were administered, 73% correctly identified the term clean correctly, but only about 55% correctly identified the term disinfect. If I were to put this on a straight grading scale, we are performing at a level of C to F level in terms of identifying the definition of clean versus disinfect.
Eight years later, Burco (2007) posted the same question to practicing audiologists, and although improvements are seen with clinicians correctly defining both terms nearly 80% of the time, we are still only performing at essentially a C+ level when it comes to differentiating basic infection control terminology. So, then, what exactly do we mean by these terms? The term clean refers to removal of gross contamination. It does not necessarily involve the killing of germs. Cleaning is an extremely important precursor to disinfecting as well as to the process of sterilizing. In contrast, disinfect or disinfecting is a process whereby germs are killed. Now, the spectrum of kill basically is going to depend on what kind of product that you are using.
Disinfection must be performed on touch and splash surfaces or on individual patient items that are not transferable to others. For example, when it comes to knowing what to do in terms of touch surfaces, we are not doing a very good practical job. When Burco (2008) asked audiologists whether or not they properly clean and disinfect touch and splash surfaces after each patient appointment, only 55% reportedly are doing so. So on the Bankaitis grading scale, we are essentially performing at a solid F when it comes to executing required infection control standards.
Sterilization
The fourth standard precaution refers to the sterilization of critical instruments. Again, there are a couple of terms that we need to define. Critical instruments refers to any instruments that are introduced directly into the bloodstream that you intend to reuse, or any non invasive instrument that comes in contact with mucus membranes or bodily substances, or any instrument that can potentially penetrate the skin from use or misuse. There are a lot of different critical instruments throughout the audiology environment including reusable immittance and OAE tips and specula as well as every cerumen management instrumentation including curettes, loops, forceps, and the tips on penlights used for earmold impressions. By way of terminology, we previously indicated disinfect refers to a process of killing germs, whereby the spectrum of killing germs is dependent on the product that you use. In contrast, the term sterilization is a process whereby all germs are killed each and every time. As such, sterilization requires a product to meet specific requirements as outlined by the Environmental Protection Agency (EPA). Sterilization must be performed on any and all reusable critical instruments that you have in your environment.
When it comes to identifying the appropriate definition of sterilize versus disinfect both Amlani (1999) and Burco (2007) felt that audiologists were correctly identifying the definition of sterilize when they are provided with closed-set choices. While correct identification of the terms is evident by an A to B+ performance, does this knowledge translate to the clinical environment? The short answer to that is no.
For example, when the same audiologists who correctly defined the term sterilize were asked whether they actually sterilized critical instruments such as reusable cerumen removal instruments, Burco (2007) found that less than half (45%) sterilized the instruments prior to reuse. So, despite doing a great job defining the terms, this suggests that we are doing a poor job, about an F, when it comes to executing some basic infection control procedures.
Infectious Waste
The final standard precaution refers to the disposal of infectious waste. Infectious waste refers to any sharp instruments such as a blade, needle or scissors that are used for hearing aid modifications or tubing cutting, as well as any kind of waste contaminated by cerumen and blood. Now that we are familiar with these standard precautions and how they relate to audiology, I want to demonstrate the application of this knowledge by taking you through my thought process to create a written work practice control.
Let's specifically take a look at how we should be performing a hearing aid listening check that is in a manner consistent with minimizing the potential spread of disease and create a work practice control. The first thing we need to figure out is which of the five guidelines must be accounted for in our work practice control (Figure 2). To make things easier, we are going to approach this work practice control as if we have already greeted the patient. We have already washed our hands and we are in the middle of an appointment and need to perform a listening check. In terms of personal barriers, since we are handling a hearing aid that may or may not be contaminated we need to account for this precaution in the work practice control. I am thinking specifically that I am going to use either gloves or disinfectant towelettes as a personal barrier so that I avoid making contact with the hearing instrument directly.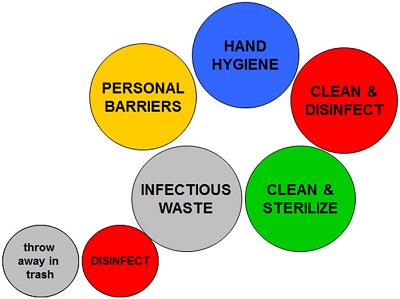
Figure 2. The five main areas, represented in large circles, for which to account when developing a work practice control.
When it comes to cleaning and disinfecting, I prefer to clean and disinfect the hearing aid so I can handle it with my bare hands. I want to make sure I account for that. Clean and sterilize does not apply in this situation, but infectious waste does. I am going to have contaminated towelettes that I have to dispose of. With this line of thinking, I need to write a work practice control that is designed to minimize the spread of disease by accounting for these three standard precautions: personal barrier, clean and disinfect and infectious waste.
Figure 3 illustrates one example of a perfectly acceptable work practice control for performing a hearing aid listening check without cross contaminating hearing instruments and/or the actual listening stethoscope. The standard precautions that we need to integrate into our work practice control are color coded by the bubble and the written portion of the work practice control.
Figure 3. Example of work practice control for hearing aid listening check.
In this situation, we start with a personal barrier by accepting the hearing instrument with a disinfectant towelette. We are not going to make direct contact with it. We are going to then clean the hearing aid surface with a portion of the towelette and then disinfect the same surface with an unused portion of the towelette. We are going to discard the towelette in the trash. We are going to perform the hearing aid listening check. Then, once we are done performing the listening check, we are going to use a fresh disinfectant towelette to clean the listening bell and both ear pieces. Then you will return the stethoscope to its appropriate resting location and discard the other towelette in the trash.
This could be an example of just one correct hearing aid listening check procedure. There might be other clinics that want to be a little more conservative and make sure that gloves are being used, so that procedure can be easily modified to account for all the things needed to ensure that the work practice control is consistent with the guidelines.
Infection Control Product Selection
Next we will talk about selecting products. I am not going to endorse a one brand over the other. The point of this is to take those different standard precautions and talk about some of the things to keep in mind when making informed decisions about what products you purchase.
Personal Barriers
Let's start with personal barriers. The biggest question that we get pertains to gloves. There are a variety of gloves available in both latex and non latex versions. This is a matter of preference, but keep in mind there are a fair number of clinicians and patients who have a sensitivity to latex. In addition many of the gloves provide you the option of powdered or powderless. This is not magic powder, nor does it make it easier to do your job. The powder simply helps with putting the gloves on. Again, it is a matter of preference.
Finally, when it comes into gloves, size does matter. There is no such thing as one size fits all. We do not work at Subway where we assemble sandwiches. We are hearing healthcare professionals that provide audiological services to our patients. It is important to ensure that the glove that you choose as fit like a second skin. When you are wearing an inappropriately sized glove it is going to compromise your manual dexterity ability and you are going to get extremely frustrated. In some clinics you have to provide three or four different sized gloves because everyone's hands are sized differently.
Hand Hygiene
Until recently, the only way to wash your hands was with traditional soap and water. Then the CDC and the American Medical Association (AMA) approved the use of no-rinse hand degermers as an alternative to hand washing, particularly when a clinician does not have access to a sink with running water. Keep in mind that really nothing takes the place of traditional hand washing. The no-rinse degermers were most likely approved because healthcare professionals were reportedly doing such a poor job washing their hands in the first place. Additionally, these foams and gels are alcohol based, which will dry out hands more readily, creating some potential issues in terms dry chapped skin. In the event you come in direct contact with any form of contamination such as blood or saliva, you should wash your hands with soap and water and avoid or refrain from performing hand hygiene procedures with these degermers.
You hear the term hand hygiene used to refer to either traditional use of soap and water or hand degermers. When you are investing in soap, it has to be liquid soap. Bar soap is a breeding ground for germs and is not appropriate in the clinical environment. In addition, make sure that the liquid soap is medical grade because it will contain special emollients that help keep your hands from drying out. In addition, using antimicrobial soap does not matter. Finally, remember that these no-rinse hand degermers should be treated as an alternative to hand washing, not a replacement.
Disinfectants
Disinfectants come in a variety of forms. You can get them in sprays, towelettes and liquids; it ends up being a matter of preference. You want to make sure that the disinfectant that you use is hospital grade because they do have a broader kill spectrum than some household disinfectants. In the audiology environment, you should consider using non alcohol based disinfectants, particularly on hearing instruments and earmolds because alcohol will quickly dry out acrylic plastic, rubber and silicon. Use something such as Audiologist's Wipes, Cavicide, or other non alcohol based product to help preserve those items. The other thing to keep in mind is disinfectants do not sterilize. Now you know the difference between disinfect and sterilize as we previously indicated.
Sterilants
Sterilants have specific product requirements. There are a variety of ways that an audiologist can sterilize something, but the most common procedure is referred to as cold sterilization. Cold sterilization involves sterilizing instruments by immersing them in a chemical for a specific amount of time. This requires investing in specific equipment including a tray as well as the chemicals. There are only two ingredients to date that have been approved by the EPA to work as cold sterilants. Any product that contains 2% or higher concentration of the chemical glutaraldehyde, such as seen in the brand Wavecide, or 7.5% hydrogen peroxide or higher concentration, as seen in Sporox, are approved sterilants that can be used. Do not confuse this hydrogen peroxide with the hydrogen peroxide that you can buy over the counter at a drug store. It is not the same thing. Regardless of which product you use, it is critical to follow the instructions because they differ across several factors.
First and foremost, you have to soak critical instruments for a specific amount of time to achieve sterilization. If you remove the instruments prior to the minimum amount of time, you have not achieved sterilization. The only thing that you have done is highly disinfected that item. For example, the minimum soak time for glutaraldehyde-based solutions in general is 10 hours with a maximum use in the tray of 28 days. In contrast, hydrogen-peroxide based sterilants must soak for at least 6 hours and have a maximum use in the tray of 21 days. You have to be familiar with the use and reuse of the liquid that you have poured in the tray. Each manufacturer will also have different guidelines of how to dispose of the liquids properly. When you invest in these types of products, you are responsible for reading all the instructions so you are using them correctly.
Furthermore, you should be aware and familiar with the Material Safety Data Sheet or MSDS. This is a document that outlines the hazards associated with chemical products. If you are going to be investing in chemicals for cold sterilization it is important for you to ensure that you have an MSDS on file. The MSDS outlines the chemical composition, as well as the physical and chemical characteristics of the product, exposure limits and any kind of precautionary measures that need to be taken in the event of an exposure. They are not necessarily included in the packaging, so you should be able to ask the distributer or manufacturer from which you are purchasing these products to provide you with the MSDS sheet. You have to have that on file as part of JCAHO certification. OSHA also requires access to the MSDSs as well. I know for every product that Oaktree sells, there is an electronic version of the MSDS to make your lives easier.
Some people would rather be doing something different when it comes to chemicals and sterilants, and some clinics have decided to approach infection control from the perspective of disposable products. Disposable products are one-time-use only products that you throw out after use on one patient, so there is no need to clean or sterilize the item. The nice thing about disposable audiology supplies is that it does decrease potential infection control errors by eliminating the human error that can occur when cleaning or sterilizing equipment. There are quite a few different disposable products that are available.
Disposable Supplies
In terms of disposable diagnostic tools and supplies, we have the traditional disposable otoscope specula, insert earphones, headphone covers, immittance tips, OAE tips and electrodes that are both generic and manufacturer specific. These are the most popular items. There are also disposable a variety of disposable probe tubes for real-ear measurements as well as temporary earmolds that are disposable. Grinding-wheel caps are also considered disposable, as well as mini muslin buffs, suction needles and a variety of cleaning tools, and bit blocks for earmold impressions.
There have been some new developments in cerumen management, as well. Not only do we have access to disposable curettes and loops, but also to the day hook, which is one of my favorite tools, as well as disposable suction tubes for suctioning cerumen out of the external auditory canal and disposable forceps that are relatively affordable. These are items that many clinics are investigating. Now, whether or not you should or should not take the disposable supply approach is likely an executive management decision coupled with personal preference, but it is important to at least look at what may make the most financial sense for your clinical practice. Products cost money and good decisions need to be made by each clinic independently.
For example, say your clinic removes cerumen mechanically on average of twice a day. If you assume there are 250 business days a year, the total number of patients seen for cerumen removal is going to be 500 each year. If you are going to take the reusable approach, you might minimally have to invest in two buck curettes, two day hooks and two forceps, and invest in a soaking tray as well. Over a one-year period of time you will use about three gallons of Sporox in order to fulfill this removal of cerumen on a twice-a-day basis. Your total investment for one year is going to be about $350. If you take the disposable approach, you will buy a box of disposable curettes with 100 to a box and day hooks that are about 100 to a box and some disposable forceps. You are not going to have to invest in a tray or any type of chemicals. You might spend $500. However, bear in mind if you are going to do the reusable approach you are only going to be spending $100 the second because you have already invested in the reusable tools and only need chemicals, whereas in year two of disposables you are going to spend another $500. If you are going to remove it fairly often, it makes sense to go the reusable route. In contrast, if you only remove cerumen once a week or 50 patients annually, it may make more sense for you to go the disposable route.
Infectious Waste
Most infectious waste can be disposed of in regular receptacles and discarded through regular disposable procedures. Disposables that can injure, such as blades, must be placed in puncture-resistant disposable containers such as the sharps container. These are available in many different sizes. In certain events, when waste is contaminated with cerumen or blood, the material is placed in a separate impermeable bag and discarded in the trash. There are also spill kits available that help you safely clean up blood, urine or other infectious waste. The kit contains absorbent beads that turn into gel when poured on to a spill. The spill kit has everything you need to disinfect and dispose of two gallons of spill fluid.
Resources
We reviewed a lot of information today. Rest assured, there are resources readily available that will guide you. There are three different infection control books that are specific to the dispensing environment (Bankaitis & Kemp, 2003), the audiology clinic (Bankaitis & Kemp, 2005) as well as for speech language pathologists. These books cover everything that we reviewed in the past two presentations, as well as provide you with a template of the written infection control plan. I have provided my contact information at the end of this presentation to anyone who is interested in receiving a copy of the template. I do not see why you have to reinvent the wheel.
In addition, there are free patient resources in the form of infection control counseling cards that can be used to educate our patients on properly cleaning and disinfecting their hearing instruments. As an audiologist I remember spending about 30 seconds instructing patient on how to use the little brush, clean it like this, you are fine. But it is important for us to start treating hearing aids as if they were dentures and provide counseling on how to clean and disinfect them. Furthermore, my blog, which can be accessed through Word Press at aubankaitis.wordpress.com, has an infection control section which provides information on infection control. You can subscribe to it by entering your e mail address and you will get an automated update every week, or you can just access it on an as needed basis.
Summary
The take home message is that infection control is a required element of our clinical practice. We are ethically and legally obligated to do it. Use your standard precautions as your guide to create those ever-important work practice controls which will dictate how you and your staff will be performing clinical procedures in a manner that is consistent with minimizing the spread of disease.
Finally, make sure that you take various factors into consideration when selecting products for your clinic. Make sure you implement your written infection control plan by ensuring appropriate training, and then be sure to rely on resources, one of which is me. I can be easily reached at [email protected]. The customer service staff at Oaktree Products is also extremely knowledgeable about infection control and can certainly guide you, and you can contact us at (800) 347-1960. Thank you so much for your attention.
Questions and Answers
Do you need to dispose of the used towelette in the biohazard rather than the trash?
That is a good question and a discretionary call depending on if there is a lot on the hearing instrument. In general, no, you can throw them in the trash. The biohazard bag is typically more for those instances when you are doing cerumen removal and you have copious amounts of cerumen on the disposal curette or for when you remove insert ear tips and there is a significant amount of residual earwax or blood. Again, it is somewhat of a discretionary thing. Some clinics like to be more conservative and say that everything goes in a biohazard bag, and other places use discretion depending on what can be visibly identified on the disposable products.
Doesn't the stethoscope bell for listening aids also get contaminated? I did not see any mention of cleaning, disinfecting or sterilizing these. Additionally, are there disposable bell tips for standard listening scopes available?
Let me answer the second part of that question first. Yes, there are disposable bells available, but you are going to have to look at how often you are considering of disposing them to decide if it is cost effective for you. If you first clean and disinfect the hearing instrument surface and then place the bell on it, and then once you remove the bell clean and disinfect the bell, you are pretty much okay in terms of moving forward and doing the next listening check. Some clinics prefer to remove the bell and sterilize it. But since you have already essentially disinfected the hearing instrument, you have eliminated 99% of the germs that may reside on that surface. This is where infection control is also an art. It is going to boil down to the philosophy of the clinic. Some people like to be more liberal and some people like to be more conservative.
I have a question about the precautions for Methicillin-resistant Staphylococcus aureus (MRSA) positive patients in audiology testing. I think some individuals think that all is well because they are wearing gloves, forgetting that they are infecting the surfaces that they are touching if they are not changing the gloves.
Correct. That is a perfect point. One of the things that often occurs when something like MRSA pops up, like HIV in the 1980s, is that people start freaking out. Not to be a smart aleck, but my response is that you should be doing what you typically do with every one of your patients, because you have no idea who does or does not have MRSA. It is extremely important that when you are manipulating or touching somebody with a gloved hand that you are also cognizant of the areas you may or may not be touching in the clinic. Once you take those gloves off, you should commence with hand washing and proceed to the next step. Those are very good points.
What is your recommendation for cleaning toys used in visual and play audiometry which children of these young ages usually put in their mouths?
Great question. First, we need to differentiate a motivational toy that is used for diagnostic purposes versus a waiting room toy. A waiting room toy is going to refer to a toy or an object that resides in the waiting room and, therefore, is not used for diagnostic testing. You are asking about a motivational toy, which refers to a toy or object that is going to be used specifically during, behavioral audiometric testing or any diagnostic procedures. Toys and motivational devices that are used for audiological assessment should be cleaned and disinfected after each use. It is considered a touch surface. So in between each patient, those items need to be cleaned and disinfected minimally. Toys really need to be non porous or plastic so you can easily disinfect them. This is going to sounds like the no fun zone, but plastic materials are easier to clean and maintain than wood or fuzzy toys. Children place toys in their mouth so great care should be taken when handling toys that come into contact with saliva. You need to make a decision whether to dispose of it or sterilize it. In terms of waiting room toys, disinfecting them once a day is sufficient.
References
Amlani, A.M. (1999). Current trends and future needs for practices in audiologic infection control. Journal of the American Academy of Audiology, 10, 151-159.
Bankaitis, A. U. & Kemp, R. J. (2003). Infection control in the hearing aid clinic. Boulder, CO: Auban.
Bankaitis, A. U. & Kemp, R. J. (2005). Infection control in the audiology clinic (2nd Edition). Boulder, CO: Auban
Burco, A. (2007). Current infection control trends in audiology. Independent Studies and Capstones. Paper 287. Program in Audiology and Communication Sciences, Washington University School of Medicine, St. Louis, MO. Retrieved from: digitalcommons.wustl.edu/pacs_capstones/287


