Learning Outcomes
As a result of this Continuing Education Activity, participants will be able to:
- Identify 5 evidence-based stimulus parameters for ABR measurement in infants.
- Identify 5 evidence-based acquisition parameters for ABR measurement in infants.
- List 3 practical steps for minimizing ABR test time.
Introduction
Dr. Jay Hall: Today’s course will focus on an evidence-based protocol for infant hearing assessment that has evolved over the past 45 years. This webinar is part of a 2-part series on infant hearing assessment using electrophysiological measures. The second part is entitled Value of Chirp Stimuli in ABR/ASSR Measurement, and you can find the course in the AudiologyOnline library in both recorded and text formats: access it here.
Although the protocol I’m discussing today about would work with adults, it is specifically designed for infants. I’ll start with a review of what is meant by the term “evidence-based”. This is a term you hear very often in audiology and it is important to understand what it means. In addition to covering evidence-based acquisition parameters, how we acquire the response, and specifics about electrodes and filter settings, I will spend some time toward the end of the presentation on how to increase the efficiency of ABR assessment in infants. Efficiency is essential; the quicker we can conduct the ABR, the more information we collect for every minute of time, then the more likely we are to get all of the information needed for audiological diagnosis and management. ABR on an infant is often our only opportunity to define an infant’s hearing status, which of course is necessary for implementing management and intervention. Efficiency is a very important part of this process.
Evidence-Based Practice
This is a quote from Leonardo Da Vinci that is very appropriate when we talk about evidence-based practice: "Those who fall in love with practice without science are like the sailor who steers a ship without a rudder or compass; you never know where you'll end up."
We must use evidence from research, primarily clinical research but also basic research, in order to guide what we do in the clinic. Using evidence from research will lead us to the best diagnosis and the most effective management. There are great resources on the topic of evidence-based research. An often-cited textbook on evidence-based medicine is Evidence-Based Medicine: How to Practice and Teach EBM (Sackett, Straus, Richardson, Rosenberg, & Haynes, 2000). These principles certainly apply to audiology as well as medicine.
With evidence-based practice, we take all the research knowledge that's appropriate for the particular patient we're providing services to, and then apply that knowledge along with our clinical expertise. One important point is that evidence-based practice does not mean simply following protocols. It does not mean that even an incompetent person can get good results with these protocols. Evidence-based practice means combining the best evidence with our clinical skills and our clinical experience to answer a question. The question might be, "Does this child have a hearing loss, and if so, exactly what is type and degree of loss in each year?” The evidence should lead to logical and rational decisions about management.
Not all evidence is created equally; some evidence is better than others. There are four or five different grading systems for evidence. One of them is from the US Preventative Services Task Force, which has five levels of evidence, with Level I being the strongest:
Level I: Randomized control trial
Level II: Non-randomized control trial
Level III: Cohort or case-control study
Level IV: Ecological or descriptive studies
Level V: Opinions of respected authorities based on: clinical experience, descriptive studies, or reports of expert committees
There are some expert committees that develop guidelines like the Joint Committee on Infant Hearing (JCIH), and they don’t really fall into this category. Committees that develop guidelines review evidence as part of their processes so those guidelines have a higher grade of evidence. The best evidence is always from a randomized control trial where there are normal subjects, controlled subjects, and all the variables are controlled. An important point to remember when discussing evidence is that just because something has been published does not mean it is the very best evidence.
The JCIH position statement on a protocol for evaluating hearing loss in infants and toddlers is an excellent example of an evidence-based document that describes evidence-based practice. The most current JCIH position statement was published in 2007 but a newer one is due out soon. You can access the 2007 position statement here.

Figure 1. Summary JCIH (2007) protocol for infant/toddler hearing evaluation.
In Figure 1, you can see that the auditory brainstem response is a very important part of this protocol. In fact, it is the most important part of the protocol to define hearing status in children from birth to age 6 months, and that is why we are focusing so much time on it. Without the ABR, you really cannot define hearing status in infants of this age. With the ABR, you should be able to define hearing sensitivity adequately in order to provide the needed management, if indicated.
Historical Perspective
The ABR was discovered in 1970 by Don Jewett. He would not have done it without his mentor, Robert Galambos, guiding his research. In 1971, Jewett published a definitive article on what we now call the ABR (Jewett & Williston, 1971). The ABR went by many names back then. We are now celebrating the 45th anniversary of the ABR; it has been around that long.
Almost immediately after the ABR was discovered, it was applied clinically by Robert Galambos and a neurologist, Kurt Hecox (Hecox & Galambos, 1974). They described the ABR in infants and children, and also in adults. It was quite clear at this point that the ABR would be revolutionizing hearing assessment. For the first time ever, it was possible to evaluate hearing in children within hours after birth. My career started in audiology in 1974, when I got my certification in audiology (CCC). I'd already had my CCC in speech pathology and I made my conversion from speech pathology to audiology. Fortunately, I was at Baylor College of Medicine in Methodist Hospital with Jim Jerger, Deborah Hayes and others. Jim Jerger was good friends with Bob Galambos. They talked all the time on the phone and Bob Galambos would come by the clinic every now and then when he was visiting from California.
We were doing ABR's in 1974 in infants and young children and we have never looked back. The technique is still very trustworthy as the best way to define hearing status in infants.
In the early years, the equipment was very large. Some of you may remember the Nicolet CA 1000 device that was introduced in the early 1980's. Many of us used it – there were a lot of buttons and knobs that had to be pressed in order to operate the device and yet it did a good job. These devices are still in use, by the way. When I was recently lecturing in Mexico, I met a few audiologists who are still using it.
Cross-Check Principle
In discussing ABR in infants and young children, it is important to at least mention the Cross-Check Principle. I recommend that everyone read the seminal article on the Cross-Check Principle (Jerger & Hayes, 1976), if you haven’t already read that at some point in your career. I invited Dr. Hayes to present an AudiologyOnline webinar on the Cross-Check Principle, which is just as valid today as it was when the article was published 40 years ago. You can find her course in the AudiologyOnline course library - access it here.
The main point of the article is that we need to cross check behavioral audiometry results to ensure an accurate diagnosis. We need independent objective tests that do not depend on a behavioral response, such as immittance measurements like tympanometry and acoustic reflexes. Objective tests also include ABR. If we use these independent tests, we can cross check, or validate, the results of behavioral audiometry. By doing so, the result will be an accurate diagnosis that will lead to proper management. If we instead rely exclusively on behavioral audiometry, we will make mistakes, and some of the mistakes will have a lifelong negative impact on the children we are evaluating. We must cross check behavioral measures with independent objective tests whenever possible, and it is almost always possible.
In many cases, particularly in infants, we can always perform the objective tests such as immittance measurements, ABR, ASSR, and electrocochleography (ECochG) even though we may not always be able to administer behavioral tests.
Evidence-Based Protocol
Today, there are many options for conducting an ABR. When the 1976 paper by Jerger and Hayes came out, the only stimuli available for air and bone conduction ABR were click stimulation. Today, 40 years later, we have clicks, traditional tone burst, and chirp stimulation. In 1976, we didn’t have ASSR. We had EcochG and cortical measures, but they weren't being used clinically at that time.
Figure 2 is an image from the e-handbook of Auditory Evoked Responses (Hall, 2015).
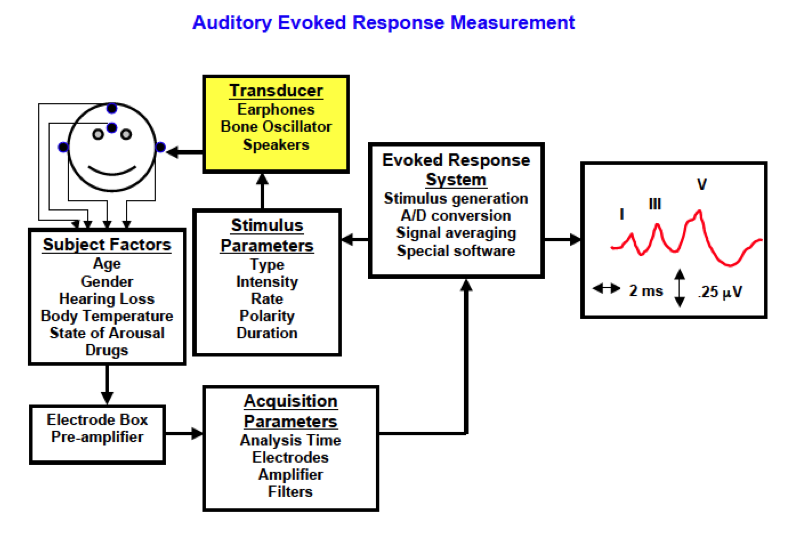
Figure 2. Auditory evoked response measurement. Source: e-handbook of Auditory Evoked Responses (Hall, 2015).
In the upper left of the figure is the patient. We present the stimuli through transducers – either some type of earphone, or a bone oscillator or even a loud speaker. A loudspeaker is typically not used for ABR, but might be used for the cortical responses.
There are electrodes on the patient’s scalp and we have to consider other acquisition parameters - filter settings, analysis time, and the amplifier. While we can control the stimulus and acquisition parameters, we cannot control the subject factors. Subject factors include the patient's age and gender, which both play a role in the ABR. They also include hearing loss and even physiologic parameters such as body temperature, state of arousal or any drugs like sedatives or anesthesia that may be used during testing. These are all the different factors – some of which we can control and some that we cannot - that we need to consider. Today we will focus on the ABR stimulus and acquisition parameters.
Stimulus Parameters
I'm going to summarize an evidence-based protocol beginning with stimulus parameters outlined in Figure 3.
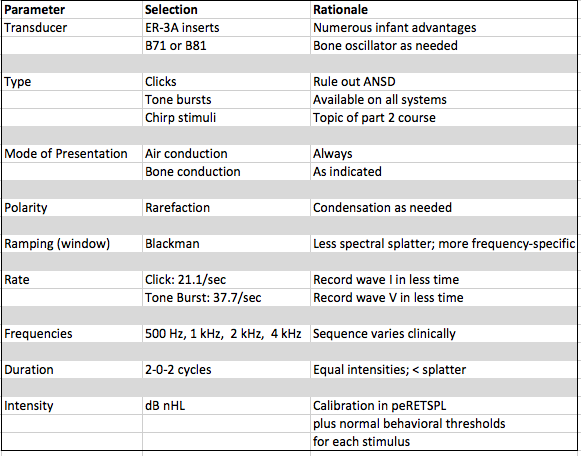
Figure 3. Evidence-based protocol: Stimulus parameters.
Each and every one of the parameters in this protocol has been proven to work, according to the research. In other words, someone studied the benefits of insert earphones, someone studied the different types of bone oscillators, someone has studied the effect of polarity on the ABR, etc. When we use these parameters in our test battery, we are not just selecting them randomly to see if they will work. They are selected based on research findings so we know they are going to work.
If you follow the evidence-based protocol presented here to record ABRs in infants, and your patient is sleeping quietly with very little muscle artifact and very little electrical artifact, I can almost guarantee you will get the very best results possible, even if you're a beginner. I've seen students who have never done an ABR before use this protocol to perform an ABR on a patient who was sleeping quietly. They obtained the same quality of ABR that I would have obtained with my 40 years of experience recording ABR's.
I cannot emphasize enough the importance of a protocol. It is the most important thing that you can control in recording an ABR.
Transducer
You should always use insert earphones for an infant ABR unless the child has aural atresia and has no ear canal. In that case, you're going to need a supra-aural earphone. Otherwise, there is no case of a child who will be better evaluated with supra-aural earphones. Insert earphones are by far the best option, and that is universally agreed upon.
The general advantages of insert earphones apply to their use in pure-tone audiometry and speech audiometry, as well as in ABR (Figure 4). I cannot emphasize enough how important these advantages are to audiology best practices when working with infants and children.

Figure 4. The advantages of insert (ER-3A) earphones versus supra-aural earphones.
With insert earphones, you have the option of using an adaptor. You can get them from any of the manufacturers. They're not expensive, so buy four or five of them in case they get lost. They slip over the plastic tubing or the rubber tubing and allow you to use any of the immittance tips. You can fit insert earphones properly to just about any ear. Again, for most children, you do not want to use supra-aural earphones as there are too many disadvantages.
For bone conduction ABR, which I’ll just mention briefly today, the B-71 transducer or oscillator is the one of choice, although there is a new B-81. They are essentially the same except you can handhold the B-81 better. It fits against the baby's head better.
Types of Stimuli
There are three major types of stimuli: click stimuli, tone burst, and chirps. Click ABR still plays a role in infant assessment. The click ABR produces energy around 2000 - 4000 Hz. Because of cochlear physiology and mechanics, the most energy that activates the cochlea and produces the ABR is around 3000 Hz. We are not evaluating low frequency or higher frequency hearing sensitivity using the click ABR. Click stimuli still have a role in testing, for example, to rule out Auditory Neuropathy Spectrum Disorder. Tone bursts are an essential stimuli in ABR testing. Chirps are a variation of clicks and tone burst. For more information on chirps, refer to the next course in this series - you can access it here.
Mode of Presentation
Air conduction stimuli are always used in the ABR evaluation of a child. We always use air conduction and very often, we also need bone conduction. You should think of using bone conduction ABR as often as you would think of using bone conduction in pure-tone audiometry. Whenever there is a clinical indication, you must test bone conduction. In my experience, depending on the population you are testing, anywhere from 20 to 30% of the children you are evaluating with ABR will need bone conduction ABR testing. Susan Small has a course exclusively on bone conduction ABR on AudiologyOnline – you can view it here. I will touch on bone conduction ABR briefly, but I encourage you to view Susan Small's course for more details.
When you're performing a bone conduction ABR with a baby, you can stimulate any part of the temporal bone. Usually, we're stimulating the mastoid, and obviously we're doing this on the stimulus ear. Stimulating the forehead or some other part of the head is not as effective as stimulating the temporal bone. In infants under the age of 6 months, the temporal bone has not yet fused with the occipital bone, the parietal bone, the frontal bone or any of the other bones. Therefore, when we stimulate even at high intensity levels by bone conduction, the energy almost all goes to the stimulus ear. It doesn't crossover to the other ear.
In the nursery, if you see a baby who has just been born with aural atresia you can perform the ABR as soon as possible. If you test the child within the first month or two after they were born, you don't have to worry about masking at all as you don't have to worry about crossover of the stimulus. Bone conduction ABR is indicated whenever there's a risk factor for conductive loss such as aural atresia or any other medical finding consistent with conductive loss. With a flat tympanogram in a child, or when there is an ABR with a nice waveform but delayed wave I, wave III, wave V, then bone conduction is indicated.
If we conduct bone conduction ABR, for example by stimulating the right ear, we're recording the response from the right earlobe all the way up to the brain. We're using an electrode on the forehead to do that. If we stimulate the right ear, and get a wave I in the channel that's on that side, but we don't get a wave I when we're looking at the opposite channel (forehead to contralateral ear), then we are certain that this response is from the right side (test ear).
That's the EcochG principle: the wave I comes from the auditory nerve on the side we stimulate. If we get a wave I from the same side electrode and we do not get a wave I from the other side, we know for sure that we are recording the test ear and we do not have to worry about masking and crossover. That is a very important point.
Bone conduction ABR not only helps us to evaluate sensory function, identify air-bone gaps, and diagnose conductive loss, but it also can be done without the need for masking of the contralateral ear. We can get ear specific results because we have a biological marker, wave I, for the ear that we are testing. That is very different than behavioral audiometry.
Polarity
Rarefaction stimuli are the best option from an electrophysiological point of view to see what is happening in the cochlea. They'll produce a bigger response with a shorter latency.
When you are trying to record an ABR, and it is not coming out the way you would have hoped, start changing things in a logical and step-by-step manner. For example, if the ABR waveform in an infant who should have a really good ABR looks pretty poor using rarefaction, then change to condensation polarity. Sometimes that will produce a better quality ABR. Better yet, start your ABR with both rarefaction and then condensation. That is a great way to rule out auditory neuropathy. If your results show only a cochlear microphonic present and no ABR, then you would likely want to evaluate that child for auditory neuropathy. If the ABR looks about the same for rarefaction and condensation clicks, then it's really an ABR. At that point you can then decide which polarity is producing the better ABR waveform, and go with that type of polarity.
What is the rationale for using rarefaction stimulus polarity? When a negative polarity, electrical stimulus reaches the insert earphone, it produces a movement of the diaphragm of the earphone away from the ear. That movement will pull the eardrum outward, pull the stapes' footplate outward, and move the basilar membrane upward. That is what happens physiologically. The upward movement of basilar membrane is most effective in eliciting hair cell activation, so the rarefaction click directly activates the cochlea in most patients. With condensation polarity you can also get an ABR, because after you get the negative, you will get the positive polarity. With condensation, it is not the negative direction of the basilar membrane that actually produces the ABR; rather, it is the rarefaction part of the stimulus that follows the condensation that produces the ABR. Therefore, a condensation ABR tends to be a little later and a little smaller than that produced by rarefaction.
Ramping
Modern ABR systems have frequency specific stimuli. We know that we need to somehow present the stimulus at maximum amplitude from the beginning, and there are linear ramping or other mathematical equations to do that.
An electrical engineer who worked at MIT named Ralph Blackman developed an equation to go from no stimulus to maximum amplitude for the stimulus in a matter of a few milliseconds, with almost no excess energy except at the frequencies where we want it. Most tone burst stimuli are not a single frequency; that would be impossible. Tone bursts have a very brief duration, so they will be more than a single frequency, but only an octave band. I will go into more detail in the next course on chirps. If we get an octave band around a certain frequency, for example 2000 Hz, we have enough frequency specificity to adequately manage that patient.
Rate
Rate refers to how many stimuli we present per second. As a rule, the faster the rate, the better. However, we need to keep the rate within a range where we obtain optimal results; I usually use 21.1 per second for the click. I want to get a clear wave I, and at faster rates you might not get clear waves. At the same time, you do not want too slow a rate such as 11 per second. As we increase the rate from 11 per second to a very fast rate like 91 per second, the wave I usually almost disappears and the wave 5 will shift out. You can use very fast rates, at least with a perfectly normal adult, and still get a very good ABR.
With tone burst ABR, we are looking for wave V and tracking it down to threshold. We are not that interested in wave I. We have already determined that we are testing the test ear - the wave I on the test ear indicates that it is indeed the test ear. We have already evaluated the interwave latencies. So for tone burst ABR, a faster rate is adequate - I usually use 37.7 per second and you could even use a little faster rate. The faster the rate, assuming the response is still good, the quicker the test time.
Frequencies
In terms of frequencies, we need to test at least 500 Hz, 1000 Hz, 2000 Hz, and 4000 Hz. That is usually adequate, because then we're evaluating hearing within the speech frequency region.
Duration
Most stimuli have an onset, a plateau, and an offset. Pure tones, for example, have a very long plateau. For ABR, no plateau is the best option. Many years ago, Mike Gorga, Aaron Thornton and others showed that by eliminating a plateau and using a very brief onset and offset of the stimulus, we can get the same amount of energy for all of the frequencies and minimize spectral splatter. A duration of 2-0-2, with 2 cycles of rise, 2 cycles of fall, and no plateau means the stimulus is very frequency specific – it’s one octave band. With a plateau, you may introduce a few extra frequencies. The ABR is an onset response, so a plateau doesn't help generate the response.
Intensity
We do not have time today for a total review of intensity in ABR – it could be the subject of an entire webinar. We'll cover some of the main points today.
First of all, all equipment should be calibrated physically, using peak equivalent, reference equivalent thresholds in SPL. We also need to double check our stimulus intensity using behavioral assessments on a small group of normal hearing adults where we perform the ABR in the clinic.
I want to go into a little more detail about some of these parameters.
Tone Burst Stimuli Acoustic Spectrum
Keep in mind that tone burst stimuli are frequency-specific but they are not pure tones. We are not using pure-tones to generate ABR; we're not really getting pure-tone information even with an ASSR. We're getting information on octave bands. An octave band of energy is centered around a certain frequency. For example, an octave band centered around 2000 Hz will have a little energy below it and a little energy above it and that is fine for fitting amplification. Hearing aids don't amplify individual frequencies, they amplify bands of frequencies. As long as we estimate hearing in different regions including 500 Hz, 1000 Hz, 2000 Hz and 4000 Hz, and are using octave band stimuli, we will have the frequency specificity needed for fitting amplification.
Effect of Stimulus Intensity on ABR Wave V (Latency/Intensity Function)
As we decrease the stimulus intensity, we know that the latency of the ABR is going to increase. As we increase the stimulus intensity, we know the latency will decrease. That's a very important principle and we can determine whether the response is normal or abnormal by comparing a child's latencies to normative data. This is a basic physiological function and our goal is to get to the lowest intensities, as low as 20 dB and sometimes lower, and then determine whether the response the child produces is still within the normal range.
Figure 5 shows waveforms from an ABR recorded at gradually decreasing intensities. You can see the wave V latency increases as the stimulus intensity decreases. There are two other changes to note about the ABR as the intensity decreases – Wave I disappears, and the morphology changes.
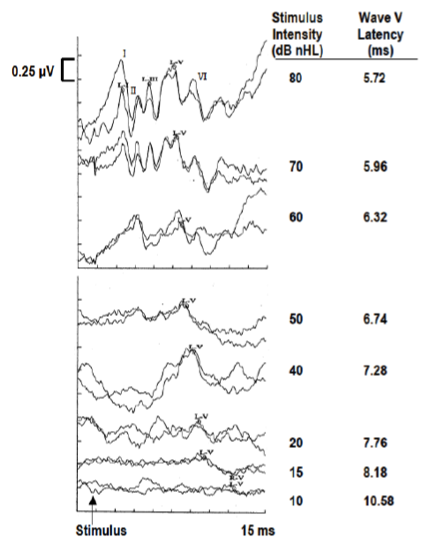
Figure 5. Effects of stimulus intensity on ABR - changes in morphology.
Wave I usually disappears around 40 – 45 dB nHL (nHL = normal hearing level). Normal hearing level refers to how much above a perfectly normal hearing person's threshold for that stimulus you're presenting. For example, 20 dB nHL means the stimulus is 20 dB above the threshold of a group of normal hearing adults for that stimulus. In terms of morphology, higher intensities produce very sharp waves, and as intensity decreases, the waves become more rounded and may lose their peaks. As we get near lower intensities, we may just have a trough.
ABR Thresholds and Estimated Behavioral Thresholds
We know there are very predictable relationships between ABR thresholds and estimated behavioral thresholds, and it varies with degree of hearing loss (McCreery et al., 2014). We can usually get within 5 to 10 dB of a person's behavioral threshold using ABR, but it's a little more complicated than that.
Let's say you have a baby who actually has normal hearing. In this case, we tend to overestimate hearing sensitivity a little bit using ABR. As we get into children with greater degrees of hearing loss, we may underestimate their hearing with ABR, even with tone burst stimuli.
It is beyond the scope of today’s course to cover calculating estimated hearing level (eHL) from the ABR using correction factors, but the point I want to make is that ABR is a valid and reliable predictor of behavioral thresholds.
ABR Acquisition Parameters
What do we mean by acquisition parameters? When we acquire an ABR, we have electrodes on the patient, and we run the output from those electrodes to a filter to get rid of some of the frequencies that don't contribute to the response. We need to pre-amplify the response because the ABR is tiny; it is less than one half of 1 microvolt.
Recommendations for acquisition parameters along with the rationale for each is shown in Figure 6.
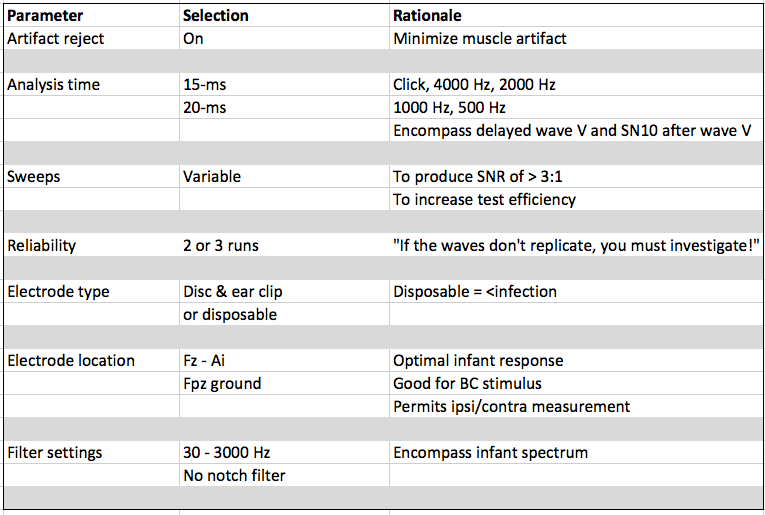
Figure 6. Evidence-based protocol: Acquisition parameters.
Artifact Reject
With most devices, we want to reject any big electrical or muscle activity coming into the electrodes or into the system through the electrodes. So the recommendation is to keep the artifact reject “On” in most cases. There are some clever ways of selecting and recording a response that has some background noise, and then amplifying or emphasizing responses that are recorded when that noise is very low. It is an advanced technique for artifact rejection that is beyond the scope of today’s talk. However, most of the time, we keep the artifact reject on.
Analysis Time
I recommend using an analysis time of 15 milliseconds after the stimulus for the click and for high frequency tone bursts (2000 Hz and 4000 Hz). I recommend using 20 milliseconds for 1000 Hz and 500 Hz tone bursts. I always use a pre-stimulus baseline of 1 millisecond so you can see what is happening before the stimulus is presented, which will give you a measure of how quiet the patient is during your test.
Why do we choose an analysis time of 15 milliseconds for click stimuli and high frequency tone bursts? We need to take into account the fact that a typical high intensity ABR for most adults is somewhere between 5.5 – 6 ms, and decreasing the intensity to 20 dB nHL will add on about 3 ms. Then, we account for all the factors that might influence the latency of the response in the worse case scenario. These factors include: a maximum conductive component (adds 2 ms to the latency), age-related latency for term infant (1 ms), hypothermia (1 ms), and a pre-stimulus baseline (1 ms). If you take all of that into account it adds up to 14 or 15 ms; with this analysis time, we still can see the response. As a quick rule, take the typical latency for wave V for a high intensity ABR for an adult, and double that latency to figure out a ballpark analysis time.
Sweeps
How many stimuli do you need to present, or how many times do you present a stimulus and sweep through the analysis time? Some audiologists may say you need 1500 sweeps, while others might say you need 2000 sweeps, however both of these answers are wrong. There is no right answer, as the number of sweeps will vary depending on your testing conditions.
When recording an ABR, our goal is to detect the signal, which is the ABR, in the presence of everything that's not the ABR such as the ongoing brain EEG, muscle artifact, electrical artifact from a fluorescent light or some other source, etc. Whatever is not the response is considered noise. We want to produce a response that's twice as big as background noise; we want a 3:1 signal-to-noise ratio (SNR) or better.
When testing a quiet child using a high intensity click stimulus, you might reach a 3:1 SNR after 500 stimuli. When you are testing a child with a hearing loss near threshold using a tone burst stimulus, it might take a lot more stimuli before you reach that SNR.
We can get a better signal-to-noise ratio by presenting more stimuli, and also by reducing the noise. We must always try to reduce the noise to the absolute minimum, which is why we sometimes have to use sedation or anesthesia to do an ABR. Once we've recorded an ABR that is 3 times bigger than all background noise or all residual noise, we can stop averaging.
Factors contributing to the signal include the intensity, the patient’s hearing status, and the type of stimulus (such as use of a chirp, which we cover in part two of this course). Factors contributing to the noise include muscle and electrical activity.
One way we can improve test efficiency is by only presenting as many stimuli as we need depending the testing conditions.
Reliability
When we're making our final measurements at low intensities that are going to be used to estimate the child's behavioral threshold, we need more than one run. If the response repeats, there's really a response there. If the waves do not repeat, your test is not complete. As we like to say, “If the waves do not replicate, you must investigate”.
Electrode Type
Some electrodes are reusable and some are disposable, but either work well. With infants and young children, disposable are often better because they minimize the chance of infection.
Electrode Location
For ABR, electrode locations are quite simple. We need an electrode on the high forehead, that's the non-inverting electrode that picks up the ABR wave III and wave V, and an inverting electrode on the ipsilateral earlobe that picks up wave I.
The mastoid will work, but there are many disadvantages to a mastoid placement for the inverting electrode. The wave I is smaller when the mastoid is used. Also, an electrode on the mastoid will be in the way when you go to do bone conduction. I would encourage you whenever possible to use an earlobe electrode rather than a mastoid electrode.
The ground electrode can be anywhere on the body such as on the shoulder or on the cheek, but we typically put it on the low forehead because we've already scrubbed that area.
Figure 7 shows the typical setup for an ABR. In this figure, disposable electrodes are used. The gray electrode wires are reusable, and they snap on to the white and gray ovals, which are single-use electrodes. The electrodes are pre-gelled and have a self-adhesive to stick on to the patient’s skin. In terms of electrode location, this photo shows the non-inverting electrode on the high forehead, the ground electrode on the low forehead, and an inverting electrode on each earlobe. Notice the distance between the electrode wires and the transducers, which are clipped on the patient’s shirt in this figure. Electrical artifact is produced at the transducers, so it is important to keep them away from the electrodes and minimize any possible stimulus artifact in the ABR.
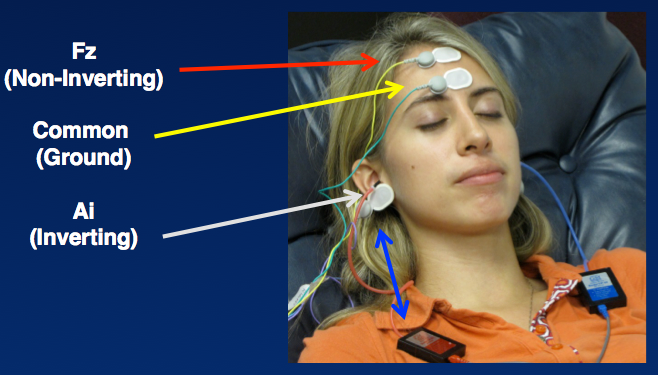
Figure 7. ABR: Electrode locations.
Filter Settings
You have to filter out unwanted electrical or muscle activity but you do not want to filter out the actual response. With infants, the ABR is dominated by low frequencies as their brains are immature, their neurons are functioning a little slower than adults, and there is not as much myelin. We must include low frequencies when we filter an ABR for an infant; we do not want to filter up around 200 – 300 Hz as then we’ll filter out the response itself. We also do not want to eliminate filtering altogether or we will get too much noise. I recommend filtering of 30 – 3000 Hz for an infant ABR, as that range encompasses the infant ABR spectrum.
Saving Test Time
Let’s talk about how we save precious test time. I actually did record an ABR once in 16 minutes, which I think is the fastest time possible for conducting an ABR. I was in the operating room and the child was anesthetized and ended up having normal hearing. While that is not typical test time, we can almost always record an ABR even with traditional stimuli in a half an hour or less. I often hear audiologists remark that ABRs are time consuming and “take at least an hour and a half." That is not true - an experienced clinician using an effective protocol with a quiet child can get all the frequency information they need in both ears and record a quick click ABR in just a matter of 30 minutes at most.
How do you do that? Here are some suggestions for conducting an ABR as efficiently as possible.
Preparation
Be prepared to begin an ABR as soon as the child is asleep. Set up your equipment ahead of time and input the child’s information. Do not waste time setting up the equipment when the child is sleeping. Have the electrodes handy with electrode gel or paste. Get the insert earphones ready with the correct size tips.
Optimize the SNR
Do everything possible to optimize the signal-to-noise ratio. Do anything necessary to make the signal larger and the noise lower. This includes ensuring the child is as quiet as possible – the child may be sleeping, sedated or anesthetized. Keep electrode impedance low and balanced, with little or no electrical artifact. Use deep fitting insert earphones to minimize acoustic noise. Use chirp stimuli to increase the SNR; in fact, simply using chirp stimuli alone can almost double the SNR. I will discuss chirps at length in the next course.
Use a Stimulus Presentation Rate of >37.7/sec
Save time by using the fastest stimulus presentation rate that still produces a high quality response.
Immediately Troubleshoot Questionable Findings
Anytime you get findings that don't match up with your expectations, or whenever you do not have a quality recording, start troubleshooting. Don't waste time. Don't keep going and think that things might improve. Stop right away, figure out what is going on, correct the problem, and then resume testing.
Think Ahead
ABR requires a tremendous amount of concentration. It's like driving down a winding road at a very fast speed - you can't take your mind off what you are doing for a second. Do not talk to people or let your mind wander when you are conducting an ABR; stay one or two steps ahead of yourself.
Think about the next step in the protocol. After you get a response at the current intensity, what are you going to do next? As you get near threshold, will you test another frequency, switch to the other ear, or move to bone conduction? Thinking ahead will help you to move quickly and get as much information as possible.
Work Smarter at High Stimulus Intensities
Particularly in higher intensities, discontinue the averaging as soon as you see a clear response that's three times bigger than the background noise. Any audiologist who routinely records an ABR to a fix number of stimuli, like 2,000 for every frequency no matter what the intensity level, is wasting time. Instead, immediately replicate with even fewer averages. Calculate latencies and amplitudes while also collecting data at the next intensity level.
Drop the Stimulus Level Quickly
Do not drop stimulus intensity levels down by 5 dB steps or 10 dB steps. Drop the stimulus intensity level as quickly as possible to near threshold. If you get a clear, beautiful ABR at 80 dB with a nice wave I and wave V, it repeats, and all latencies are good, drop down to 40 dB. The likelihood of normal hearing or pretty good hearing is very high and you can save yourself a lot of time.
Move from Click to Tone Burst
After hearing thresholds are estimated with click stimuli, begin presenting subsequent tone burst stimuli at intensity levels 20 to 30 dB above the anticipated ABR threshold. For example, if you get a good ABR with clicks at 20 dB, you could go to tone burst at a level not too far above that, and likely get a response.
Don’t Replicate “Flat” ABR Tracings
When you don't have a response, then don't bother replicating. There's no value in that.
Resource and Attribution
All of the information in this course, including images and figures, came from my book, eHandbook of Auditory Evoked Responses.
Questions and Answers
Do you prefer the plastic insert immittance tips for inserts versus the foam tips cut down with scissors?
When a parent brings a child in for an ABR, they are usually worried that their child may have hearing loss. They expect you to have the latest technology, and they’re hoping to get good results. When they see you with scissors snipping foam tips, it doesn’t convey a modern, hi tech, competent strategy for hearing assessment. Once the adaptors became available, I stopped snipping the foam tips. Buy the little plastic adapters, slip them on to the end of the acoustic tubing, and then use any size you need. What happens if you cut the foam and you cut too much? Then you’ll need to do it again and you’ll be wasting precious test time. Once you start using the adaptors you will know what colors will fit on which size child, and you can quickly switch to another size if you choose the wrong one. It is more efficient and professional than cutting foam tips.
Is replication necessary when using equipment that splits the response into A and B buffers?
Great question. I've debated with the manufacturers who have this option about this very question. The A and B buffers are great option and I use it at the beginning of an ABR with an infant so I can use an alternating polarity. Then I can separate that big waveform into the rarefaction waveforms and the condensation waveform and look to see if there's a cochlear microphonic. I don't believe that looking at the A and B buffers is a true replication, however. If you have muscle artifact, and I've seen this with the post auricular muscle, or you have any kind of electrical artifact that goes on for a few seconds, it will appear in both those buffers. Everything including artifact can replicate, so I always prefer near threshold to do a true replication.
You run the response for say 2,000 stimuli or 1,500 stimuli, and then do it again. If that really replicates, it's a response.
Is there a minimum absolute amplitude of wave I as a response criteria?
The British ABR guidelines talk about this. As a rule, when you get to a response that's less than about 0.2 microvolts, which is about a third of a normal response, you'd have to have a very quiet patient with almost no noise to feel confident about that. However, rather than consider minimum amplitude, I focus on getting a SNR of 3:1 and replicability.
If I get a very small wave V that replicates, and if the noise as much lower, I will still consider it a response. Eventually, when you decrease the intensity of the stimulus, the response will keep getting smaller and smaller. If the response is very small but it is 3 times bigger than background noise and it replicates, I would not want to say it is not a response.
Can you talk about ASSR air conduction and its role?
The next course in this series will go into more detail about ASSR. For diagnostic purposes, the tone burst ABR, particularly with chirp stimuli, will provide you with all the information you need for estimating hearing sensitivity. You may not need ASSR unless there's no ABR or you're at maximum intensity levels and you're barely recording an ABR. For bone conduction, I prefer ABR to ASSR mainly because with the bone conduction ABR, I can look for wave I and I can know whether or not I'm testing the test ear. My opinion is to use ABR with tone burst or chirp stimuli, and ABR for bone conduction, and only when that doesn't give you the information you need, consider ASSR. That's not for screening purposes, but for diagnostic purposes.
You mentioned calibrating to a physical reference.
There are 2 or 3 physical references designed for transient stimuli. It seems like more people are using ISO, which is used in other parts of the world, as opposed to ANSI, which is a US standard, mainly because testing equipment has been marketed throughout the world. My book discusses the specific references for physical calibration that are designed not for pure-tones or speech stimuli but specifically for transient stimuli, and most manufacturers are using them.
Do you correct your tone burst thresholds to get estimated hearing level for fitting hearing aids?
The ABR will not give you a behavioral threshold, but the ABR is a valid predictor of behavioral thresholds. Correction factors are needed to apply to your ABR thresholds to obtain behavioral thresholds, and it is beyond the scope of today’s course to get into the details.
In general, for a normal hearing patient, if you average enough and the patient's very, very quiet, you can record an ABR wave V down to 10 dB above the behavioral threshold. For example, if you get ABR down to 20 dB (but not at 15 dB) at 2000 Hz, then you can estimate that there's probably a 10 dB threshold. Work from Boys Town suggest that the agreement between ABR thresholds and behavioral thresholds varies as a function of degree of hearing loss.
References
Hall, J.W.III. (2015). eHandbook of auditory evoked responses. Available from Amazon.com
Hecox, K.E., & Galambos, R. (1974). Brain stem auditory evoked responses in human infants and adults. Archives of Otolaryngology, 99(1), 30-33. doi:10.1001/archotol.1974.00780030034006
Jerger, J. & Hayes, D. The cross-check principle in the diagnosis of hearing loss in children. Arch Otolaryngol, 102(10), 614-20.
Jewett, D.L., & Williston, J.S. (1971). Auditory evoked far fields averaged from the scalp of humans. Brain, 94(4), 681-96.
McCreery, R.W., Kaminski, J., Beauchaine, K., Lenzen, N., Simms, K., & Gorga, M.P. (2014). The impact of degree of hearing loss on auditory brainstem response predictions of behavioral thresholds. Ear & Hearing, 36(3), 309-319. doi: 10.1097/AUD.0000000000000120
Sackett, D.L., Straus, S.E., Richardson, W.S., Rosenberg, W., & Haynes, R.B. (2000). Evidence-based medicine: How to practice and teach EBM. London: Churchill Livingstone.
More references are listed in the course handout.
Citation
Hall, J.W.III (2017, January). Update on auditory evoked responses: Evidence-based ABR protocol for infant hearing assessment. AudiologyOnline, Article 19040. Retrieved from https://www.audiologyonline.com


