Editor's note: Please note that this article was updated and expanded in 2010 and the new version can be viewed here: https://www.audiologyonline.com/articles/fitting-hearing-aids-to-babies-848 The following original article from 2006 article remains on AudiologyOnline for archival purposes, however, readers seeking information on the topic should refer to the 2010 article or other current articles and courses on AudiologyOnline.
Introduction
Today's pediatric audiologist has a goal to identify infants with hearing loss by 3 months, and initiate intervention by 6 months (Joint Committee on Infant Hearing, 2000). In many cases, we are successful in meeting this goal, particularly when babies are otherwise healthy and universal newborn hearing screening programs are part of a comprehensive approach to Early Hearing Detection and Intervention (EHDI). For many families, early intervention will include hearing aid fitting as part of the overall plan, yet few EHDI programs have a written, comprehensive protocol for hearing aid fitting in infancy. This means we are fitting hearing aids in early infancy more often than ever before, but also that each practitioner might use their own approach. This article will discuss a few ways in which this important yet challenging work is a little bit different than fitting hearing aids in young children, and a few reasons why EHDI programs might consider adopting a systematic approach.
Responsible infant hearing aid fitting needs to ensure that babies are neither over- nor under-amplified, and that the hearing aids will provide comfortable yet beneficial levels of sound. What special factors might present barriers to accurate hearing aid fitting for babies in EHDI programs? In our program, we think that special attention needs to be paid to three infant-specific areas:
- Nonbehavioral threshold measurements are used to prescribe hearing aids;
- Babies' small and growing ear canals have special acoustic properties; and
- A growing number of practitioners rely upon software-assisted automatic hearing aid fittings as the primary method for adjusting a baby's hearing aids.
The sections below will talk about each of these, and illustrate why they matter, using case examples.
Assessment of Infant Hearing: The great divide?
Two key developments in the hearing industry have given us the ability to successfully find babies with hearing loss and test their hearing. First, accurate, cost-effective instruments that automate evaluation of the otoacoustic emission and/or auditory brainstem response allow us to screen for hearing loss. Second, the use of evoked potentials for evaluating response thresholds to frequency specific stimuli let us have a substitute for a behavioral audiogram literally months sooner than would otherwise be possible. Together, these two tools let us find and test babies with enough precision that we can fit their hearing aids in the middle part of the first year of life. This is extremely exciting!
The next logical step is to move on to the hearing aid fitting. It may seem obvious how we can do this: simply enter the thresholds from the evoked potential testing into the hearing aid fitting software, right? Unfortunately, it is a little more complicated than that. You see, different procedures exist for evoked potential threshold estimation. Each procedure uses different stimuli and calibrations, which are not entirely equivalent to the calibration used in your conventional audiometer. Audiologists who perform evoked potential assessments are quite familiar with this, but sometimes the audiologist who fits the hearing aids hasn't seen an ABR since graduate school. There have been a lot of developments in the field of evoked potentials - and this article is certainly not meant to cover them - but I would like to give audiologists who fit hearing aids a general sense of some important issues that matter for infant hearing aid fittings in this section.
The ABR has been used the longest for estimating thresholds in babies. It is used with broadband stimuli (clicks), or with frequency-specific stimuli (tone pips). The advantage of click-based ABR is that the clicks evoke a large wave V response that is relatively easy to read. Measures made with clicks are correlated with later behavioral thresholds in the 2-4 kHz region (Gorga, Johnson, Kaminksi, Beauchaine, Garner, & Neely; 2006). However, it is important to note case reports that show individual babies with unusually configured audiograms who do not follow this rule and would be misdiagnosed by click-only testing (Stapells, 2000a). To avoid this problem, ABR testing can also be done using brief tones. These tone-ABR tests give more frequency-specific results than click-ABR, avoiding most configuration problems (Stapells, 2000b). The waves they elicit, however, are less robust in appearance than a click-evoked ABR wave V, making them harder to read, particularly if the clinician has not received training and mentorship. These pros and cons may help to explain why we still see both stimulus types being used, and why some clinics choose to use them in combination. Figure 1 shows an example of tone-ABR threshold estimation for a baby seen in the H.A. Leeper Speech and Hearing Clinic at the University of Western Ontario. The results are shown for 2000 Hz and responses were observed at test levels of 60 dB nHL and above. This process gets repeated at other test frequencies (and in the other ear!) until at least one low and one high frequency threshold has been obtained and replicated.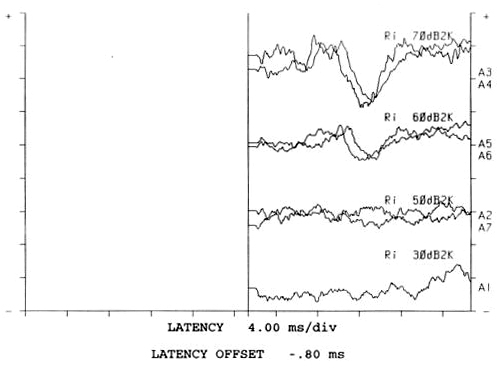
Figure 1. Example of tone-ABR threshold.
Let's take this back to the hearing aid fitting for a moment. Assume that we had a complete audiogram done using the technique shown in Figure 1, including ear-specific thresholds for both air- and bone-conducted stimuli, for low (500 Hz) and high (2000 Hz) frequencies, and that the results (along with the rest of the test battery) told us that a sensorineural hearing loss had been found. However, that doesn't necessarily mean that we can type a "60 dB" threshold into hearing aid fitting software.
Why not? Well, that 60 dB threshold was measured in dB nHL, not dB HL. If that seems unimportant, consider that the differences between nHL and HL thresholds have been shown to be as high as 20 to 25 dB (Stapells, 2000b). These differences need to be subtracted from the nHL thresholds before the measurements can be used in hearing aid prescription and fitting. Otherwise, the hearing aids may be set to have too much gain, particularly in the low frequencies.
So what is the solution? There are several, with more on the horizon all the time. First, if you use tone-ABR measures, published corrections can be used to convert the thresholds from nHL to HL (Stapells, Gravel, and Martin, 1995). Or, version 5.0 of the DSL method offers built-in or user-defined corrections from nHL to estimated HL (Bagatto, et al., 2005; Scollie, et al., 2005). For either of these approaches, the correction you use should come from data that match the test procedure (stimulus type, calibration levels, etc.) that was used to test the baby. Other types of evoked potential procedures offer built-in calibration levels, and therefore do not require further correction (e.g., Gorga, Kaminski, Beauchaine, & Bergman, 1993; Gorga et al., 2006). Others have published corrections much like those for tone-ABR measures (Rance, Roper, Symons, Moody, Poulis, Dourlay, & Kelly, 2005, Picton, Dimitrijevic, Perez-Abalo, & Van Roon, 2005). HL-based evoked potentials may be used without further correction, although note that some have been evaluated only on adults - look for infant studies to check that the procedure gives accurate threshold predictions in babies.
To summarize, the starting point of an early hearing aid fitting for a baby is a prediction of the audiogram from an evoked potential assessment. Within the three main classes of evoked potentials (click ABR, tone ABR, ASSR), there are several subtypes, any of which may use different calibration levels. This is not currently standardized. Therefore, some procedures may need some correction before they can be used to predict the audiogram in HL. A word to the wise: find out what type of procedure you (or your colleagues) are using, and whether it needs correction, before using the data to fit a baby's hearing aids. The reason for this caution is simple: missing the correction can lead to overamplification. Applying the correction is quite straightforward - an example is shown in Table 1. The baby was tested with tone-ABR, calibrated according to Stapells' recommendations (see www.audiospeech.ubc.ca/haplab/clinic.htm). Corrections are subtracted at each frequency to convert the nHL measurements to a predicted HL audiogram. This predicted audiogram was used to fit the baby's hearing aid on this ear.
Table 1. Tone-ABR assessment results in dB nHL and their correction to estimated HL values.
Note. Correction values are those used by the Ontario Infant Hearing Program, and assume ABR calibration as described in the text.
The next step: ear canal acoustics
We all know what this section is about: the real ear to coupler difference (RECD). That is true, it is, but a few new spins have come along concerning RECDs for babies: how to get them, how to predict them. It isn't news that the smaller ears of babies, toddlers, and children have large ear canal resonances. For hearing aid fitting, this means that a hearing aid with 57dB of gain in a coupler will produce higher sound pressure levels in the average baby's ear than it would in the ear of an average toddler, followed by the ear of an average child, followed by the ear of an average adult. Fewer of us are aware that the same pattern exists when we assess hearing sensitivity: baby ears receive a higher level of sound at 50 dB HL than would be received in an adult's ear, particularly at the higher frequencies (Seewald & Scollie, 1999; Voss & Hermman, 2005). This means that when we do infant hearing aid fitting, the infant's ear canal plays a role both in how we define threshold and in how we define what is needed from the hearing aid. In addition to this general age-related trend, individuals of all ages vary substantially from the average, meaning that each person's ear is unique and may be poorly described unless measured directly.
The RECD can account for the effects of the infant's ear canal on the hearing aid requirements. If insert phones were used for audiometric or evoked potential testing, the RECD can also be used to account for ear canal effects during hearing assessment. For these reasons, many infant hearing aid fitting protocols mention measurement of the RECD. Typically, RECD-based corrections are automated by hearing aid fitting software - detailed information about how this is done is available elsewhere (Bagatto et al., 2005).
One of the main obstacles in clinical practice is difficulty in measuring the RECD in a particular infant. We know that RECDs vary significantly from infant to infant, child to child, or adult to adult (Bagatto, Scollie, Seewald, Moodie & Hoover, 2002; Saunders & Morgan, 2003). So, it is important to measure them whenever possible, but at the same time this can feel like a challenge - especially in babies. Clinicians may be new to RECD measurement, and/or they may be reluctant to place a probe tube deeply into the ear canal of an infant. Moving toddlers present even greater challenges. The following sections will present a review of two solutions to these problems, and assume this basic familiarity with the RECD: it consists of measuring the sound pressure level of a test signal in a 2cc coupler, followed by measurement of the same signal in the ear. The RECD is the difference between the two. For novices to this topic, detailed measurement definitions and procedures are available (e.g., Moodie, Seewald & Sinclair, 1994, user's manuals of most probe mic systems) as well as tips and tricks suitable for helping us to make this measurement with older toddlers and children (Bagatto, 2001).
A special technique for measuring the RECD in babies
There is a modified strategy for making RECD measurements in infants and toddlers. This infant-friendly approach was designed to be more feasible for use with babies, and has been shown to produce reliable and valid results (Bagatto et al., 2006 in press). First, look in the baby's ear canal to determine that it is clear of debris. Second, tape the probe tube to the eartip or earmold (Figure 2) at the correct insertion depth (3 to 5 mm past the end of the eartip or mold - this took us to 11mm from the outer edge of the eartip in four month olds). Third, insert the taped combination into the baby's ear canal and measure the real ear portion of the RECD, taking care not to fold or pinch the probe tube during insertion. This procedure makes it easier to measure an RECD on a baby because the taped combination of probe tube and earpiece are inserted simultaneously. This reduces the number of times that the audiologist must insert objects into the infant's ear, reducing upset to the infant and increasing the likelihood of performing the measurement while the infant is sleeping or in a quiet state. Also, the taping keeps the probe tube at a reasonable insertion depth - a feat that is hard to accomplish with confidence using the more conventional placement methods. It is unlikely, however, that this method places the probe tube at the eardrum. Despite this, it generated RECD values similar to normative data in a sample of 30 infants, indicating that it may be at least as acceptable as conventional methods for RECD measurement. An example of a measurement made with this technique is shown in Figure 3.
Figure 2. An example of a probe tube microphone attached to an eartip to ease the insertion of tube/eartip combination in a baby's ear. The tube has been attached with Moisture Guard tape so that it extends past the eartip by 3 mm. The external marker on the tube is inserted flush with the opening of the ear canal.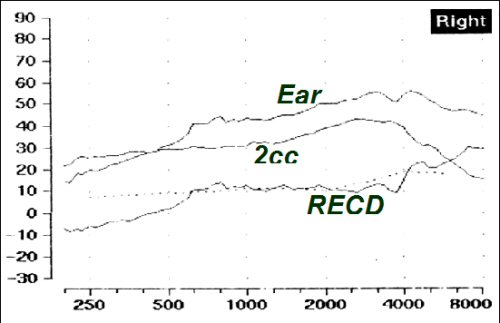
Figure 3. An example of an RECD measured on a young infant.
If you are new to RECD measures, I suggest that you learn to measure it in a few stages before attempting it with infants and toddlers. First, get some information about how to measure it, including specific instructions for your probe microphone system. Second, it helps to watch one being done by a seasoned clinician, either in person or on a video (Roush, Seewald & Gravel, 2004; Seewald, Moodie, Gravel & Buerkli-Halevy, 1997). Third, get to know the procedure and your probe mic system by practicing with a coworker or cooperative adult. Once you are comfortable, begin trying it with your more cooperative patients - it is surprisingly easy to measure an RECD on most sleeping infants. Active toddlers are the biggest challenge - making good use of distractions is really helpful. It is also important to remeasure the RECD at regular intervals - it will change over time. Measuring with each new earmold is a good minimum guideline.
What if I couldn't measure the RECD this time?
Sometimes, you just can't. Sometimes you can measure it on one ear but not the other - this happens when the baby is sleeping for the first ear but rouses and is not cooperative for the second. In this case, we check our impedance results - if the middle ear is normal on both sides, it is likely OK to use the measured RECD on both sides. However, sometimes you can't measure it at all - about 2 to 25% of the time in clinical reports I have seen. You can always try to measure it the next time you see them, but this is also where normative data come in and are really very helpful. Age-appropriate predictions for the RECD have been around for years, and certainly predate EHDI program (Feigin, Kopun, Stelmachowicz & Gorga, 1989). Newer sets of normative data are now available that tell us more about infant ear canal acoustics - for example, the biggest changes in the RECD happen in the first two years of life (Bagatto et al., 2002). These new norms have been re-analyzed and implemented into version 5.0 of DSL for use when the RECD isn't available (Bagatto et al., 2005). So, you'll be seeing these new norms appear in software systems near you over the next year or so. They are different from the older norms in a few important ways. First, they predict the RECD in age by months - so now a four month old and an eleven month old will have different predicted RECDs. Second, there are now different RECD predictions for eartips versus earmolds, and these will often be generated separately for audiometry (which often uses eartips) and hearing aid (earmolds, obviously) applications.
Moving on to the Hearing Aids!!
Now that we know the baby's ear-specific hearing status and have some way to describe his or her ear canal acoustics, it is time to go ahead and fit the hearing aids. (Assuming, of course, that this is a part of the overall intervention approach chosen by the family.) Commonly selected hearing aids for babies will include programmability, some type of nonlinear (WDRC or similar) signal processing, and lockable features such as childproof battery doors, and lockable volume controls. Other important features are pediatric sized tone hooks to improve the physical fit to the baby's tiny pinnas, well-made soft earmolds, and a care kit that lets families check and care for the devices. Some clinics make use of FM systems in the toddler years (Moeller, Donaghy, Beauchaine, Lewis & Stelmachowicz, 1996; Gabbard, 2005), or second memories for use in noisy places like the car (rather than turning the aids off in this environment, as many parents do). Anticipating these needs in the first hearing aid selection (along with the extra 5 to 10 dB of gain that will likely be needed as ears grow) may extend the usable life of the first purchased instruments.
Once loaner or purchased hearing aids are in place, they need to be set for the individual needs of the baby. One common approach is to enter all of the baby's information into the software that programs the hearing aids, and let the automatic fitting take place. Recent studies tell us that this approach is not consistently accurate even for adult patients... should we be relying only on this for babies (Hawkins & Cook, 2003)? Likely not, since the errors shown for adult-derived automated fittings were related to widely varying definitions of ear canal acoustics across manufacturers (Hawkins & Cook, 2003). It is relatively safe to assume that similar, or even greater, errors could occur for infants.
Let's look at a few examples of this. I entered a flat 50 dB HL audiogram and a flat 90 dB HL audiogram into two popular Noah modules, along with the baby's age. I then connected several modern digital hearing aids, and chose an automatic fitting for each. The hearing aids were then measured to see how the automatic fitting turned out. Any automatic fitting can (and should) be cross-checked with an electroacoustic verification of the aided responses. This is preferably done using speech, or highly speech-like signals, in combination with high-level narrowband tests of maximum output (Bagatto et al., 2005, Scollie & Seewald, 2002). In the DSL Method, the measured hearing aid responses are compared to the baby's thresholds, targets, and predicted upper limits of comfort on one graph called a SPLogram. The hearing aids are measured on the coupler, and real ear responses are predicted based on the coupler data, the RECD, and other factors. This lets the audiologist see how the hearing aid responses relate to both audibility and comfort for the baby, acting as a surrogate for verbal feedback from the baby. This form of verification can be paired with validation measures that cross-check whether the expected audibility and comfort of the fitting translates into awareness of sound and development of responses to sound as would be expected. Validation measures can be questionnaires, measurement of aided thresholds, or measures of infant speech discrimination.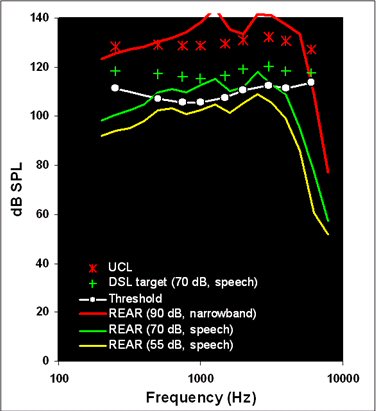
Figure 4. A verification of an automatic fitting for a 6 month old infant with a flat 90 dB HL audiogram.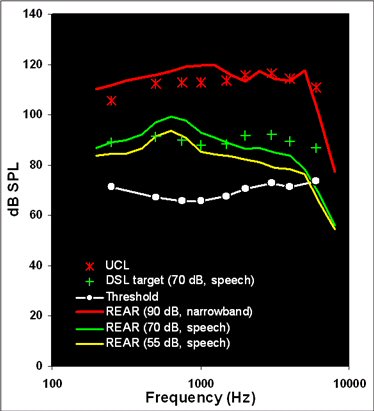
Figure 5. A verification of an automatic fitting for a 6 month old infant with a flat 50 dB HL audiogram.
The verification cross-check of these two automatic fittings are shown in Figures 4 and 5. These illustrate two of the things we sometimes see when we verify routinely. In Figure 4, the flat 90 dB HL audiogram has been fitted with too little gain/output for speech, but also with an output limiting setting that is too high. In fact, it exceeds the infant's predicted Upper Limits of Comfort by more than 10 dB at some frequencies. In a real clinical situation, we would add a filter to the earhook to smooth the response (especially in the 1000 Hz region), use a lower output limiting setting, and re-adjust the response shape for speech. In Figure 5, the flat 50 dB HL audiogram has received a more acceptable output limiting setting, but has a frequency response for speech that over amplifies the low frequencies while under amplifying the high frequencies. This type of fitting would likely lead to long term problems in detecting high frequency speech sounds, perhaps leading to speech production deficits and problems in the correct use of "s"-related cues such as pluralization and possessive marking. It would be difficult for parents or teachers to attribute any of these problems to the hearing aid itself, because children with hearing loss often have difficulty detecting and using high-frequency cues in speech, even with well-fitted modern hearing aids (Stelmachowicz, Pittman, Hoover & Lewis, 2004). Therefore, it is entirely possible that this fitting could persevere for years without adjustment, despite the fact that it is a sub-optimal when compared to any evidence-based pediatric hearing aid prescription. The audiologist who routinely verifies would know, however, that the automated fitting did not set this hearing aid correctly. Simple programming readjustments to the frequency-gain response of this device would quickly resolve the problem.
The conclusions? Put simply, routine hearing aid verification is still a must, and is more meaningful when the audiologist verifies the hearing aid against a validated prescription. Recently, there has been renewed interest in this type of procedure, in part because of excellent new practice guidelines (American Academy of Audiology, 2004) and also because of the newer speech-based verification systems that have recently come into the market. I hope that this trend continues - audiologists are responsible for creating infant hearing aid fittings that are beneficial and comfortable - and a high-quality electroacoustic verification can help us detect troublesome fittings before they are placed on a baby's ears.
Summary
Infant hearing aid fitting carries, in some ways, a higher level of responsibility than hearing aid fittings for older patients. The infant cannot tell us anything about the appropriateness of our fittings, and will not be able to for months or even years. We aggressively pursue early intervention for babies with permanent hearing losses, in the hopes that our services will allow them to develop better speech and language. In this article, I have tried to point out three main areas in which most EHDI programs do not have specific protocols, but that likely affect the benefit that many infants will or will not receive from their hearing aids. First, the link between hearing assessment (with evoked potentials) and hearing aid fitting in infancy is not always well understood, nor is it supported by highly standardized calibration at this time. This means that audiologists who fit hearing aids to babies must ensure that corrections, if necessary, are applied to the threshold estimates that they use for early hearing aid selection and fitting. Second, the highly unique properties of most infant ear canals are likely not incorporated into most infant fittings, either because the audiologist doesn't know how to measure an RECD, or because it cannot be measured during a particular appointment. A modified, baby-friendly approach is described in this article, along with some facts about new RECD normative data. Together, I hope that these two new strategies will improve the accuracy with which we estimate infant ear canal acoustics during hearing aid fitting in the future. Third, the advent of automated hearing aid fits seems to have persuaded many clinicians that routine electroacoustic verification is no longer necessary. I've shown two examples in which inappropriate hearing aid fittings would likely result unless the automated fitting is cross-checked by a high-quality verification protocol. Without these three protocol elements (threshold corrections, high quality RECDs, routine verification), some babies will receive appropriate hearing aid fittings while others will not. In the interest of consistency, I hope that audiologists who participate in EHDI programs will consider including these elements in their routine protocols.
About Susan Scollie:
Susan Scollie, Ph.D., is an Assistant Professor in the School of Communication Sciences and Disorders at the University of Western Ontario, and a Principal Investigator at Canada's National Centre for Audiology. Dr. Scollie has served on committees to develop Preferred Practice Guidelines for the prescription of hearing aids in both adults and children for the College of Audiologists and Speech-Language Pathologists of Ontario, and is a consultant for the Ontario Infant Hearing Program. She has completed efficacy studies of digital and directional hearing aids, evaluated the accuracy of probe microphone measures for completely-in-the-canal hearing aids and pediatric transfer functions, and provided technical support for the DSL[i/o] prescriptive algorithm to clinicians and industry. Her recent research has focused on the development of Version 5.0 of the DSL Method, the validity of electroacoustic verification of hearing aids, and the application of the Speech Intelligibility Index to predict speech recognition scores in children. Dr. Scollie is an Audiology Online Contributing Editor in the area of Pediatric Amplification.
Works Cited:
American Academy of Audiology. (2004). Pediatric Amplification Protocol. Audiology Today, 16(2), 46-53.
Bagatto, MP. (2001). Optimizing your RECD measurements. The Hearing Journal, 54(9), 32,34-36.
Bagatto, MP., Moodie, S., Scollie, SD., Seewald, RC., Moodie, K., Pumford, J., & Liu, K.P.R. (2005). Clinical protocols for hearing instrument fitting in the Desired Sensation Level method. Trends in Amplification, 9(4), 199-226.
Bagatto, MP., Seewald, RC., Scollie, SD. & Tharpe, AM. (accepted for publication, January 2006). Evaluation of a technique for measuring the real-ear-to-coupler difference (RECD) in young infants. Journal of the American Academy of Audiology.
Bagatto, MP., Scollie, SD., Seewald, RC., Moodie, KS. & Hoover, B. (2002). Real-Ear-to-Coupler Difference (RECD) Predictions as a function of age for two coupling procedures. Journal of the American Academy of Audiology, 13(8), 416-27.
Feigin JA., Kopun JG., Stelmachowicz PG. & Gorga MP. (1989). Probe-tube microphone measures of ear-canal sound pressure levels in infants and children. Ear and Hearing, 10, 254-258.
Gabbard, S. (2005). FM Systems for Infants and Young Children: A Tool to Optimize Early Listening. In R Seewald & J Bamford, (Eds.). A Sound Foundation Through Early Amplification: Proceedings of the Third International Conference (155-161). Stäfa Switzerland: Phonak.
Gorga MP., Kaminski JR., Beauchaine KL., & Bergman BM. (1993). A comparison of auditory brain stem response thresholds and latencies elicited by air- and bone-conducted stimuli. Ear and Hearing, 14, 85-94.
Gorga MP., Johnson TA., Kaminski JR, Beauchaine KL, Garner CA, Neely ST. (2006). Using a combination of click- and tone burst-evoked auditory brain stem response measurements to estimate pure-tone thresholds. Ear and Hearing, 27: 60-74.
Hawkins, D., & Cook, J. (2003). Hearing aid software predictive gain values: How accurate are they? The Hearing Journal, 56(7), 26,28,32,34.
Joint Committee on Infant Hearing. (2000). Year 2000 Position Statement: Principles and Guidelines for Early Hearing Detection and Intervention Programs. American Journal of Audiology, 9, 9-29.
Moeller MP., Donaghy KF., Beauchaine KL., Lewis DE., & Stelmachowicz PG. (1996). Longitudinal study of FM system use in nonacademic settings: effects on language development. Ear & Hearing, 17, 28-41.
Moodie KS., Seewald RC. & Sinclair ST. (1994). Procedure for predicting real-ear hearing aid performance in young children. American Journal of Audiology, 3, 23-31.
Picton TW. Dimitrijevic A. Perez-Abalo MC. Van Roon P. (2005). Estimating audiometric thresholds using auditory steady-state responses. Journal of the American Academy of Audiology, 16, 140-56.
Rance G., Roper R., Symons L., Moody LJ., Poulis C., Dourlay M., Kelly T. (2005). Hearing threshold estimation in infants using auditory steady-state responses. Journal of the American Academy of Audiology, 16, 291-300.
Roush, PA., Seewald, RC. & Gravel, J. (2004). Hearing care for infants: Strategies for a sound beginning. Phonak Video Focus 4. Phonak AG: Stafa, Switzerland.
Saunders GH. & Morgan DE. (2003). Impact of measuring threshold in dB HL versus dB SPL on hearing aid targets. International Journal of Audiology, 6, 319-326.
Scollie, SD. & Seewald, RC. (2002). Electroacoustic verification measures with modern hearing instrument technology. In RC Seewald & JS Gravel, (Eds.), A Sound Foundation Through Early Amplification: Proceedings of the Second International Conference (pp.121-137). Stäfa Switzerland: Phonak.
Scollie, S., Seewald, R., Cornelisse, L., Moodie S., Bagatto, M., Laurnagaray, D., Beaulac, S. & Pumford, J. (2005). The Desired Sensation Level Multistage Input/Output Algorithm. Trends in Amplification, 9(4), 159-197.
Seewald, RC., Moodie, KS., Gravel, JS. & Buerkli-Halevy, O. (1997). Pediatric hearing instrument fitting. Phonak Video Focus 2. Phonak AG: Stäfa, Switzerland.
Seewald, RC. & Scollie, SD. (1999). Infants are not average adults: Implications for audiometric testing. The Hearing Journal, 52(10), 64-72.
Stapells DR. (2000a). Frequency-specific evoked potential audiometry in infants. In: Seewald RC, (Eds), A Sound Foundation through Early Amplification: Proceedings of an International Conference (pp. 13-32). Stäfa, Switzerland: Phonak AG.
Stapells DR. (2000b). Threshold estimation by the tone-evoked auditory brainstem response: A literature meta-analysis. Journal of Speech-Language Pathology & Audiology, 24, 74-83.
Stapells DR., Gravel JS. & Martin BE. (1995). Thresholds for auditory brain stem responses to tones in notched noise from infants and young children with normal hearing or sensorineural hearing loss. Ear & Hearing, 16, 361-371.
Stelmachowicz PG., Pittman AL., Hoover BM & Lewis DE. (2004). The importance of high-frequency audibility in the speech and language development of children with hearing loss. Archives of Otolaryngology Head Neck Surgery, 130, 556-562.
Voss, S.E. & Hermman, B.S. (2005). How does the sound pressure generated by circumaural, supra-aural, and insert earphones differe for adult and infant ears? Ear & Hearing, 26, 636-650.


