Editor’s Note: This text course is an edited transcript of a live webinar. Download supplemental course materials.
Learning Objectives
- After this course learners will be able to list key articles describing cohort studies looking at vestibular function and balance.
- After this course learners will be able to describe the results of key cohort studies that investigated vestibular system function and balance.
- After this course learners will be able to explain some general findings that can be applied across patients based on the results of the key cohort studies reviewed.
Introduction
Dr. Devin McCaslin: Today we are going to talk about the power of cohort studies. We know it is impossible to read everything in the literature, and we will demonstrate that there is value in paying attention to what are known as cohort studies. In particular, we are going to talk about the NHANES (National Health and Nutrition Examination Survey) study. I will cover what the NHANES is, what cohort it is that they are drawing on, and then we will discuss three articles that focus on balance and vestibular issues using a cohort study.
NHANES
NHANES is a large-scale survey research program. At the head, is the National Center for Health Statistics. The goal of this cohort is to assess the health and nutritional issues with adults and children over time, although we are only going to discuss the adult study today. Interviews are included in the studies, many times in the home. Physicians or medical technicians perform physical examinations, and they track closely the demographic, dietary, and socioeconomic status of each of the individuals that are enrolled in this cohort study. Over time, that allows us to discover possible risk factors for the issue in which we are interested. They collect information like blood pressure readings; it is a key indicator for the health of a national issue, such as ours.
The first NHANES study was conducted in 1971. This became an annual event starting in 1999, and the first report of this data was published in 2001. They use the data from these surveys to form national standards for a group of individuals. They look at measurements such as height and weight; it is a key epidemiological issue where they can look at smoking, identify the risk factors and how that affects people’s health over time. This drives public policy. If you remember, once they figured out smoking was cancerous, they did a lot to get people to stop smoking. Funds from the government go into trying to support what they find in these studies.
The sample we are talking about includes people over age 60. They try to get a cross-section of the demographic of the country. All participants visit physicians and have dietary interviews; everyone excluding the children have blood taken. They even have a dental screening. They assess this annually, which gives indicators and predictors for certain disorders.
The health interviews where responses about day-to-day lifestyle are obtained are done in the home. The researchers drive vans, which are essentially like portable hospitals for doing the medical screenings. Many times, they partner with research organizations such as Johns Hopkins, as well as with universities and local health care providers. The National Institute of Health uses a lot of this collected data to drive what we should be funding. It drives public policy for the Food and Drug Administration (FDA) as it is a good means of longitudinal data.
The NHANES team consists of a physician and a medical technician who draws blood. The staff is trained on how to interview, ask about diet, risk factors, the person’s lifestyle, heredity, and in what environment they live, such as living next to a nuclear power plant. They track almost every detail of these peoples’ lives including smoking, alcohol consumption, and regularity of physical fitness. The staff is bilingual. All of this is put in a large scale database which can be drawn upon to adjust different policies.
Article #1: Head Impulse Test Abnormalities and Influence on Gait Speed and Falls
Today we will talk about three articles, the first of which will address dizziness. Yuri Agrawal and colleagues (2013) from the Hopkins group, who work closely with the NHANES group, published a study about the Head Impulse Test, which they use as a screening method for vestibular function and how can that predict either gait speed or falls in older individuals.
Background
The group wanted to know what effect having vestibular dysfunction or a vestibular impairment has on fall risk and gait speed, and they used the NHANES cohort to answer this question. While there are many different reflexes, we are talking about using the canal-ocular reflex, which is what the Head Impulse Test evaluates. There are many others, such as otolith ocular reflex and vestibulo-spinal reflex, in which the vestibular system sends projections to many sites all over the body in lower and upper extremities.
The Head Impulse Test utilizes the vestibulo-ocular reflex (VOR). In a normal patient, when you have them focus on a target and then move the head quickly 180-degrees to the left, the eye will go 180-degrees to the right. The fovea should be able to stay fixed on the target. If there is a problem with the vestibular system, the eye will come off the target, and when the head stops moving, the eye will have to jump back and reacquire the target, which is called a catch-up saccade.
Many of us have seen information about the Video Head Impulse Test (V-HIT). This test uses the same concept. In the research group, the authors use the B-HIT (Bedside Head Impulse Test). This is the test that physicians will do as a gross screening of the lateral semicircular canals. From that, they qualify the patient as either having some kind of vestibular dysfunction or not.
In a normal Head Impulse Test, the directions to the patient are to keep their eyes directly on your nose. Then you move their head quickly. If the eyes come off the nose, the examiner will see that the eye has to jump to reacquire the nose if that side is weaker. The following video is a normal B-HIT exam.
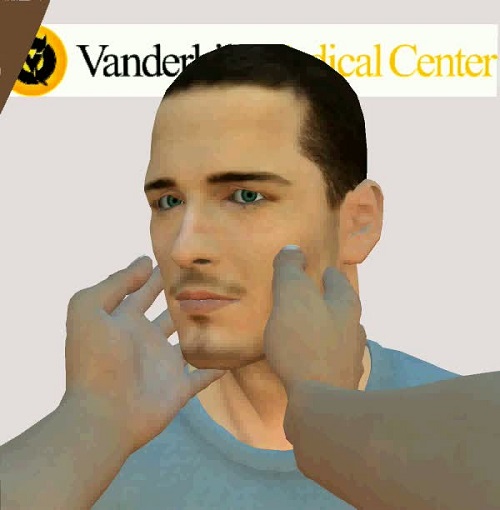
This response is indicative of normal peripheral function, in particular, lateral semicircular canal function.
The next video shows what happens when the left side is impaired. If you thrusted toward the impaired side, you will end up seeing the eye come off the nose, and that would be qualified as an abnormal Head Impulse Test.
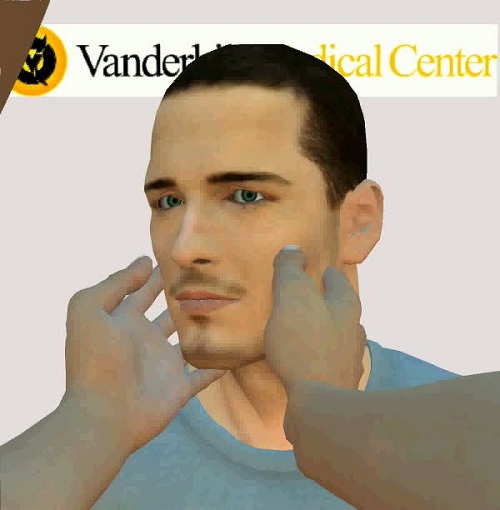
This is a positive Head Impulse Test with catch-up saccade after the head has stopped moving, which is indicative that the lateral canal on the left side is weaker. You can say that this person has a vestibular impairment.
Methods
So how did the researchers get to looking at the vestibular function, gait speed, and risk of falls? They did a cross-sectional study, conducted primarily in an academic tertiary-care medical center. They looked at 50 community-dwelling individuals who were 70 years or older. These people had no identification of vestibular function impairments. Patients were excluded if they had poor neck range or cervical neck issues. They could not have visual impairments, either. If they had a positive history of vestibular disease, they were not included. Diabetes mellitus we now know has several connections with vestibular impairments, so that diagnosis was also exclusionary.
They did a number of tests to determine if the vestibular system function was causing the falls or slower gait speed. There are many systems that are involved in balance, so they had to make sure that they tested those also to isolate the effect of the vestibular system impairment on those two outcomes.
They did visual acuity testing using the Early Treatment of Diabetic Retinopathy Study Eye Chart. This was used in the NHANES study for everyone. They performed cutaneous sensory testing, where you poke a person in the foot with a nylon monofilament at three sites so that you can look at the proprioceptive or somatosensory system.
Grip strength has also been shown recently in the literature to be predictive of issues with gait speed, and, in some cases, falls. They measured grip strength in kilograms with a handheld dynamometer. In order to measure gait speed, they had the person walk four meters at a normal pace. They timed this three times and would take the fastest time. The history of falls was based on a history from the patient. They would ask the patient how many times they have fallen in the last year and last five years and then calculate their responses.
Results
They found the mean age to be 77 years old. They key groups were those with an abnormal B-HIT and those with a normal B-HIT, with the idea that there was a difference in the people who had abnormal vestibular screening tests and those with normal B-HITs. They found there was no significant difference in visual acuity in the group of people who had an abnormal HIT and those with a normal HIT. Similarly, there was no significant difference in insensate sites, which is where we poke the person with the monofilament in the bottom of the foot for either group. There was also no significant difference in grip strength between people with an abnormal HIT and people with a normal HIT.
When looking at gait speed and falls, they found significant differences between HIT groups. In people who had an abnormal HIT, their gait speed was significantly slower than people who had a normal HIT. They also found that people who had an abnormal HIT had more falls than people who had a normal HIT. Remember that these were people who did not report any vestibular problems and had no history of vestibular dysfunction.
When patients had an abnormal HIT, the odd were five times greater that they would also have a slower gait speed, which was a significant finding. Additionally, the HIT was significantly different when looking at the odds of falling in five years. Visual acuity affected gait speed and the one-year history of falling.
Discussion
It is surprising that these people do not report any issues. Those with vestibular dizziness issues do end up having a high proportion of vestibular impairments. The most surprising thing is that almost 50% of these individuals had an abnormal vestibular screening test on the HIT. If you were to test people at a nursing home, you would find almost half of them, based on this data, have a vestibular impairment that is undiagnosed.
There were some limitations in this study. One was that some participants may not have remembered all falls on their self-report of falls. They did not control for cognitive function or psychological issues. One thing to know is that the HIT is not always significant. We find in the caloric exam that there is almost 40% unilateral weakness before a HIT becomes significant. We also do not know the test/retest of the HIT is at this time.
In conclusion, screening for vestibular impairments using the simple HIT is important in these older individuals. Regardless if they report dizziness or not, we need to consider that it is worthwhile to screen the vestibular system function in this elderly cohort of patients.
Article #2: Diabetes, Vestibular Dysfunction, and Falls: Analyses From the National Health and Nutrition Examination Survey
Dr. Kaylee Todd: I will be discussing diabetes, vestibular dysfunction, and falls analysis from the NHANES survey (Agrawal, Carey, Della Santina, Schubert, & Minor, 2010). According to the Centers for Disease Control (CDC), diabetes affects 380 million people worldwide and 29 million in the United States.
Diabetes is a chronic disease where cells either do not generate insulin or they resist against insulin. As a result, there are high levels of glucose in the blood. Insulin is a hormone produced by the pancreas to control for blood sugar. After a person eats, insulin moves glucose into the bloodstream, and glucose is then used as fuel for the body. People develop diabetes when they have high blood sugar because their body does not take that glucose out of the blood stream. The risk of falling is increased in patients with diabetes. There have associated complications and risk factors for falls, such as peripheral neuropathy and retinopathy.
According to a study by Agrawal in 2009, patients with diabetes are 70% more likely to have vestibular impairment. We know that diabetes causes microvascular injury involving small blood vessels that supply nerves and this affects small distal nerves first and then progresses to larger nerves. That is why people with diabetes will develop peripheral neuropathy in their hands and feet.
The relationship between diabetes and vestibular dysfunction is not well characterized. Past studies have looked at possible causes of diabetes and impairment in the vestibular system. There have been studies that support pathologic changes from diabetic microangiopathy in rat vestibular end organs as well as type I hair cell degeneration. Other animal studies have also indicated pathological myelin sheath thinning around the vestibular nerves in those with diabetes.
There have also been some studies completed on small samples sizes of people with diabetes. Some ENG testing revealed evidence of abnormal vestibular dysfunction, and another study that was completed in 2002 found a significant difference in the time constant of the VOR during rotational chair testing. Other studies have found peripheral and central findings during ENG testing. The results from previous studies are variable, and more research needs to be completed evaluating the relationship between diabetes and vestibular impairments.
This Agrawal study (2010) was designed to look at the effect of diabetes on the vestibular system and whether vestibular impairments have an independent effect on fall risk in those with diabetes.
Methods
The participants were first given a balance questionnaire, with a focus of any history of previous falls. The balance testing consisted of a modified Romberg test, which had four different test conditions, designed specifically to look at the sensory components of the balance system such as the visual, vestibular, and proprioceptive systems. The participants were excluded if they could not stand on their own, were having dizziness that caused unsteadiness, if they weighed more than 275 pounds due to limitations of the equipment, or if they had any foot or leg amputations.
Condition one of the Romberg test was where participants would stand with their eyes open on a firm surface. Condition one of the Romberg test is roughly equivalent to condition one of the Sensory Organization Test (SOT), for those who are familiar. Condition two of the Romberg test had the participants stand with their eyes closed on a firm surface. This would eliminate any vision, but their proprioception was still available. This was equivalent to the SOT condition two. In condition three, the participant would stand with their eyes open on a compliant surface, such as a six-inch thick foam rubber square, and this would distort the proprioceptive sense. This was equivalent to condition four of the SOT. Finally, condition four for the Romberg test had the participant stand with their eyes closed on a compliant surface. This would eliminate any vision and distort the proprioceptive sense. The participant would have to rely more on the vestibular input. This was roughly equivalent to condition five of the SOT.
Only subjects who passed conditions one through three were included in the study. It is important to remember that the authors contended condition four in the study assesses a patient’s ability to maintain balance when vestibular information is the only reliable sensory input in the absence of parallel visual and proprioceptive cues.
It was considered a pass if the participant was able to stand for 15 seconds in conditions one and two, and 30 seconds in conditions three and four. It was considered a fail if the participants had to open their eyes during any of the testing or if they had to move their arms or feet to maintain that stability, or if they needed assistance from others to maintain stability. If the participant did fail, they could repeat the test and if they failed the test a second time, the test was ended.
There were also examinations for peripheral neuropathy to look at the sensitivity in the feet. These participants were lying in a supine position and a standard monofilament was used to apply pressure to three different locations on the foot. It was considered mild neuropathy if there was no sensitivity in one to three areas, and it was considered a severe neuropathy if they had no sensitivity in four to six areas. The participants were excluded from this part of the study if they had a bilateral amputation.
The next part of the examination was a visual examination to measure visual acuity; this was used as a proxy for retinopathy. 20/20 was considered as no visual acuity impairment, 20/20 to 20/40 was considered to be a mild visual acuity impairment, and worse than 20/40 was considered a severe visual acuity impairment. Participants were also excluded from this part of the study if they were determined to be completely blind or have no light perception in both eyes or if they had a severe eye infection during the time of testing.
Results
From the total sample of U.S. adults aged 40 years and older, 17% had diabetes. Of that 17%, 54% were found to have vestibular dysfunction. That was found from the condition four, where subjects had to rely on more vestibular input when the visual and proprioceptive input was distorted or eliminated. They also found that 22% of participants with diabetes had a six- to ten-year duration of disease, and 38% had greater than ten-year duration of the disease. These were statistically adjusted for sociodemographic and cardiovascular variables.
Next, they found that hemoglobin A1C levels were 7.0% and greater in 41% of individuals with diabetes. The hemoglobin A1C level test is a glycated hemoglobin test that assesses the amount of sugar in the blood. For those of you who are not familiar with this test, normal is considered less than 5.7%, prediabetes is between 5.7% and 6.4%, and anything greater than 6.4% would be considered to have diabetes.
Next, they found that the prevalence of mild and severe peripheral neuropathy among patients with diabetes were 19% and 7.1%, mild and severe retinopathy were 54% and 22%, and the prevalence of falls in individuals with diabetes was 12%.
The prevalence of vestibular dysfunction in this group is 54%, and the prevalence of falls was 12%.
The findings suggest that diabetes increases the risk of vestibular dysfunction. It was observed that those with diabetes and vestibular dysfunction had a significantly longer duration of the disease and higher hemoglobin A1C levels. There was also a higher prevalence of vestibular dysfunction among participants with diabetes that also had other complications.
In the comparison among the different complications, vestibular dysfunction had the most effect on the association between diabetes and falls risk. It is also noteworthy that when people only had retinopathy or neuropathy, there was no significance with the complication and risk for falls. These findings suggest that vestibular dysfunction may represent a newly recognized diabetes related complication.
Conclusion
In conclusion, as diabetes progresses or hemoglobin A1C levels are increased, the person is at greater risk for vestibular impairments and other complications. It is important that people with diabetes learn to manage their diabetes and help control for additional complications. It is important for us to also communicate strategies with this specific population to help reduce any falls that could occur in the future.
Further research is still needed to better understand the relationship between diabetes and vestibular impairments, and it is needed to more specifically look at specific vestibular tests that evaluate the different vestibular end organs to help understand this relationship.
Article #3: Association between vestibular and cognitive function in U.S. Adults: Data from the National Health and Nutrition Examination Survey
Dr. Gary Jacobson: The article I am going to discuss was authored by Semenov and colleagues and published in 2015. The data they used emanated from the NHANES study.
We know that aging results in a reduction in function of the three interdependent senses supporting balance, and those include vision, the somatosenses and vestibular system function. These reductions in function and the effect of age on the brain that integrates these senses place patients at risk for falls. Information published from about 2005 onward has suggested that the vestibular system may contribute to higher cortical functions including memory, attention, and even some executive functions.
Taking this one step further, the behavior of animals and humans who have been deafferented, (i.e. meaning that both peripheral vestibular systems have been destroyed through surgery or as a function of exposure to ototoxic medications), has suggested that the loss of a peripheral vestibular system has a negative effect on spatial orientation, including spatial memory and spatial navigation. The definition of visuospatial memory is the part of memory responsible for recording information about one’s environment and its spatial orientation. This is a function you need in order to navigate around a familiar city. You need to know where you are and what landmarks there are on the way to your destination. You also need to know when you have reached your destination as well. Unfortunately, visuospatial abilities also deteriorate with age.
Older individuals have greater difficulty than younger subjects navigating in real world or virtual environments. In particular, errors occur in returning to starting places. My example is trying to find your car in a large parking lot after you have been busy shopping at a large mall.
Methods
Prior to this study, it was unknown whether age-related changes in the vestibular system had an effect on spatial cognitive function in older adults, which brings us to the data collected between 1999 and 2002 in the NHANES. During this period of time, investigators evaluated vestibular function, although some might say they were measuring balance function. They also assessed cognitive function. They conducted balance testing on individuals 40 years of age and older using the Romberg test that Dr. Todd discussed in this paper.
Patients who underwent cognitive testing were 60 years of age or older. Once the subject selection criteria were applied, about 3,200 subjects underwent balance testing, about 1,300 subjects who underwent cognitive testing, and this is the value of cohort studied. These studies with sample sizes in the thousands can give you statistical power that you would not have otherwise. If patients fail condition four of the Romberg test, which is equivalent to condition five of the SOT of the EquiTest protocol, they are classified as having “vestibular dysfunction.”
The cognitive test was the Digital Symbol Substitution (DSS) test of the Wechsler Adult Intelligence Scale, third edition. The patients were given a key that paired the numbers 1through 5 with various symbols. The subjects had to supply the correct symbol for each number. There were a maximum of 133 items, and subjects were given two minutes to supply as many correct answers as possible. It is believed that the DSS assesses psychomotor speed, attention, visuospatial skills for our purposes, associative learning, and memory.
Results
The mean total score was 45.5 points, with a range from 0-100. There obviously were some people who could not do it at all. Only 58% of the patients were classified as having vestibular dysfunction. Vestibular dysfunction was associated with a significant mean reduction in the DSS score of 3.4 points, which the investigators said is equivalent to an increase in five years of age in cognitive functioning. Further, the investigators stated that vestibular dysfunction explained 8.8% of the association between age and the DSS score.
The investigators felt that they had demonstrated a connection between vestibular dysfunction and cognitive function; in this case, that was visuospatial memory. Further, (their words) “…vestibular loss associated with aging may contribute to the far more common cognitive deficits observed in older adults. Indeed, it has been hypothesized that vestibular loss contributes to the onset of Alzheimer’s disease.” (Italics are our words.)
Lower cognitive performance related to vestibular dysfunction was associated with an almost three-fold increase in falls risk. Further, individuals with vestibular loss have been shown to have impairments in the ability to generate internal representations of their external world.
One interesting paper the authors referenced in their discussion was written by Thomas Brandt and colleagues out of Germany in 2005. The investigators examined 10 individuals with neurofibromatosis Type 2 (NF2) who had undergone bilateral surgery and were effectively deafferented bilaterally. He compared the performance of this group of patients to a group of age and sex-matched normals. They used a human virtual reality adaptation of the Morris Water Test, which is shown in the course handout.
In this test, a rat is placed in a pool of water at one of four compass points (north, south, east, and west). There is a visual landmark, a star for example, on the wall above the pool; this serves to provide visual orientation to the rat. A platform is above the water level in one of the four quadrants of the pool. The rat learns to swim to the exposed platform using the actual location of the platform and its orientation to the visual landmark. Eventually, the platform is submerged a bit and the rat must use its visuospatial memory to successfully reach the submerged platform. At any rate, the deafferented patients showed impaired performance when required to retrace their path to the hidden platform. The human research done was done in virtual reality.
Discussion
The inability to generate mental maps of the environment may make it difficult for patients with vestibular loss to navigate around obstacles in their way, leading to falls and fall-related hip and wrist fractures. Also these impairments may result in a reduction in the ability to spatially interact with the environment making it difficult to conduct common activities of daily living, such as traveling and shopping.
What are some explanations for the connection between vestibular impairment and cognitive function? One answer may be that bilateral vestibular loss may produce atrophy in the hippocampus. The paper by Brandt and colleagues in 2005 showed that patients with bilateral vestibular loss demonstrated a 17 to 18% loss of hippocampal mass. This atrophy could result in impairments in visuospatial memory and perception. Deafferentation was associated with atrophy of almost a cubic centimeter of hippocampal mass regardless of the subject’s gender.
We know that cognitive and spatial memory deficits remain for at least five to 10 years post-deafferentation, attesting to the link between vestibular nuclei and the hippocampus. The role of the hippocampus in spatial memory is known; however, the pathway connecting the vestibular system to the hippocampus still is unclear. One theory is that the pathway may pass from the vestibular nuclei through the posterior lateral thalamus on its way to the hippocampus.
Other Explanations?
What are other possible explanations for the association between vestibular impairment and cognitive dysfunction? Postural and gaze instability from the bilateral loss may require the brain to allocate more attentional resources for balance and orientation so that the patient has less cognitive reserve. Cognitive dysfunction may also result from an anxiety and depression that are common for dizzy, vertiginous, and unsteady patients.
So what do we know now that we did not know before? We know that connections probably exist between the vestibular nuclei and the hippocampus via the thalamus. We know that patients with impaired balance, not explicitly vestibular impairment, demonstrate evidence of impaired visuospatial memory. We also know past research shows that bilateral vestibular loss results in 17 to 18% reduction in hippocampal mass and degradation in visuospatial memory. We know that real-world consequences of bilateral loss may include a corruption of the visuospatial map the person has of their environment and may predispose patients to falls. Lastly, visuospatial memory may represent a dimension we need to assess in patients who have bilateral vestibular impairments.
Questions and Answers
Did the study differentiate between type I and type II diabetes, and would you see the same results in type I diabetes?
This particular study did not differentiate between type I and type II diabetes, so I do not have the answer as to what the results would be.
References
Agrawal, Y., Carey, J. P., Della Santina, C. C., Schubert, M. C., & Minor, L. B. (2010). Diabetes, vestibular dysfunction, and falls: analyses from the National Health and Nutrition Examination Survey. Otology & Neurotology, 31(9), 1445-1450. doi: 10.1097/MAO.0b013e3181f2f035
Agrawal, Y., Davalos-Bichara, M., Zuniga, M. G., & Carey, J. P. (2013). Head impulse test abnormalities and influence on gait speed and falls in older individuals. Otology & Neurotology, 34(9), 1729-1735. doi: 10.1097/MAO.0b013e318295313c
Agrawal, Y., Platz, E. A., & Niparko, J. K. (2009). Risk factors for hearing loss in US adults: data from the National Health and Nutrition Examination Survey, 1999 to 2002. Otology & Neurotology, 30(2), 139-145.
Brandt, T., Schautzer, F., Hamilton, D. A., Bruning, R., Markowitsch, H. J., Kalla, R., et al. (2005). Vestibular loss causes hippocampal atrophy and impaired spatial memory in humans. Brain, 128(Pt 11), 2732-2741.
Semenov, Y. R., Bigelow, R. T, Xue, Q-L, du Lac, S., & Agrawal, Y. Association between vestibular and cognitive function in U.S. Adults: Data from the National Health and Nutrition Examination Survey. 2015. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 71(2), 243-250. doi: 10.1093/gerona/glv069
Citation
Jacobson, G., McCaslin, D., & Todd, K. (2016, March). Vanderbilt Audiology's Journal Club: The value of cohort studies in the study of vestibular system function and balance. AudiologyOnline, Article 16612. Retrieved from https://www.audiologyonline.com.




