Introduction
A cochlear implant is a prosthetic device designed to provide hearing to patients with severe to profound sensorineural hearing loss. A portion of the device, referred to as the "implant package," is surgically placed in the mastoid portion of the temporal bone. An electrode array extending from the implant package is inserted into the cochlea. The remaining portions of the device are worn externally and include a microphone, speech processor, cable, and transmitting coil. Figure 1 illustrates the MED-EL Cochlear Implant System and TEMPO+ Speech Processor.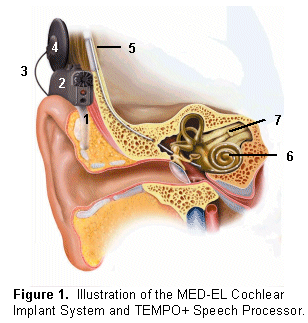
Sound is collected by the microphone (1) and sent to electronic components within the speech processor (2). The speech processor analyzes the input signal (sound) and converts it into an electronic code. This code travels along a cable (3) to the transmitting coil (4) and is sent across the skin via frequency modulated (FM) radio waves to the implant package (5). Based on characteristics of the code transmitted to the internal device, electrode contacts within the cochlea (6) provide electrical stimulation to the spiral ganglion cells and dendrites extending into the modiolus. Electrical impulses then travel along the auditory nerve (7) and ascending auditory pathways to the brain.
The characteristics and code transmitted by a cochlear implant are dependent, not only on the components of the device itself, but also on the way in which the device is fit. Fitting of a cochlear implant, also referred to as "programming" or "mapping," creates a set of instructions (code) that defines the specific characteristics used to stimulate the electrodes of the implanted array. This set of instructions is referred to as the patient's "program" or "map." Implant programming is performed by an audiologist with specialized training in the field of cochlear implants. The audiologist uses interactive software and computer hardware to create individualized patient programs that are stored on the hard drive of the computer and downloaded to the memory of the patient's speech processor.
With advances in cochlear implant technology, the fitting process has become more complex. Today's implants offer a number of sophisticated parameters that can be manipulated to improve sound quality and speech understanding. All cochlear implants require at least two levels to be set for each stimulating electrode. These measurements are referred to as the Threshold level (abbreviated as THR or T) and the Most Comfortable Loudness level (abbreviated as MCL, M, or C). Exact terminology and abbreviations are device specific, but the general purpose of these measures is common across all cochlear implants.
Fitting a Cochlear Implant
The THR and MCL levels are psychophysical judgments of loudness that are measured in clinical units of electrical current, referred to as current units (cu). Threshold levels are comparable to acoustic threshold levels and are related to the softest inputs detected through the implant. Most Comfortable Loudness levels indicate the level at which a sound is loud but comfortable. Together, the two values determine an individual's electrical dynamic range.
In adult cochlear implant patients, THR and MCL levels are typically measured using verbal feedback from the patient. Threshold levels may be obtained using an ascending presentation, followed by a standard bracketing procedure. In some cases, patients may require a reference tone prior to and/or during use of an ascending approach. MCLs are obtained through a method referred to as loudness scaling. The level of current is gradually increased, while the patient reports on the level of loudness and comfort. Ideally, MCL levels should also be balanced across the electrode array. Loudness balancing requires comparison on at least two electrodes at a time until all MCLs are perceived as equally loud.
A cochlear implant performs optimally when THR and MCL levels are set accurately and MCL loudness is balanced across electrodes1. Research indicates that MCL level and loudness balancing have a greater influence on patient performance than THR level. Smoorenburg et al. found that a reduction in THR levels of 25 to 30 current units does not adversely affect speech perception scores. Alternatively, a change in the slope of MCL values may negatively impact speech understanding. Study results "show that speech perception is quite insensitive to the threshold setting," and "imply that one may question the common focus on T levels in fitting cochlear implant speech processors."2
Based on speech acoustics, Dawson et al. hypothesized that inadequate and/or unbalanced MCL levels would adversely affect amplitude cues and spectral information important for phoneme identification. To investigate the potential impact on speech performance, MCL levels were systematically varied. Unbalanced MCLs resulted in qualitative differences and significantly poorer speech perception. Subjects described the sound quality as rough, with echoes and dominant sounds. Mean speech performance was significantly poorer for both sentence and phoneme stimuli in the unbalanced condition.3
Fitting recommendations for the MED-EL COMBI 40+ Cochlear Implant System are consistent with research findings. MCL levels and loudness balancing are of greater importance than THR levels. While THR measurements should not monopolize the fitting session, it is important to confirm that THR values are set below an audible level. For patients that cannot reliably respond to threshold levels of stimulation, THR values may be set between the minimum current level and a value equal to 10 percent of the MCL. This protocol should facilitate THR levels that are inaudible, without a significant impact on speech performance. Therefore, the primary emphasis of each fitting session should be on the MCL.
Loudness scaling and loudness balancing of the MCL require subjective responses to sophisticated listening tasks. Even adult patients who have experienced a relatively short duration of deafness can find the fitting process to be stressful and fatiguing. For children, who often lack the listening experience, language, or conceptual development to perform specific fitting tasks, the challenge is much greater.4 While many young children can be conditioned to provide information regarding threshold, "clinicians must often rely on clinical intuition and trial and error in order to estimate appropriate comfort levels for very young patients."5
Fitting Techniques for the Pediatric Patient
It is widely accepted that children diagnosed with hearing loss must receive adequate auditory stimulation and appropriate intervention as soon as possible to maximize the potential for learning language. With the introduction of neonatal screening programs in many states, children with hearing loss are often identified and fit with amplification at very young ages. For children with congenital and/or prelingual hearing loss who do not benefit from conventional amplification, cochlear implantation at younger ages can lead to improved outcomes.4
Research has shown that children implanted prior to 2 years of age achieve open-set speech perception equal to or faster than children implanted after age 2.6 Moreover, prelingually deafened children have demonstrated the capacity to learn language at rates equal to that of their hearing peers.7
Cochlear implant technology and related surgical techniques have undergone a number of advances over the past two decades. As a result, the candidacy criteria for implantation continue to expand. In addition to very young children, an increasing number of children with multiple developmental delays are now being evaluated for cochlear implant candidacy.8 With expanding criteria, the fitting of pediatric cochlear implant patients has become more difficult.
Over the past several years, audiologists and researchers have explored more effective means of fitting pediatric cochlear implant patients. Fitting techniques may incorporate a number of traditional testing procedures, including both behavioral and objective methods for working with young children. The preferred method(s) often differ based on the age and abilities of the individual patient.
I. Behavioral Measures
Behavioral fitting methods require the patient to indicate when he or she has heard a specific stimulus. An electrical stimulus is presented through the implant via interactive software and a fitting system designed by the device manufacturer. Children are able to indicate that they've heard the stimulus in a wide variety of ways, depending on their age, listening experience, and cognitive abilities. Regardless of the specific method employed, there are a number of critical factors that must be considered when performing behavioral measures with children.
When fitting a cochlear implant, the patient's speech processor is hard wired to the fitting system via a programming cable. Thus, the audiologist, patient, and fitting equipment are in close proximity to one another. There may be a number of visual indicators when the stimulus is presented, and it is crucial that the patient not have access to these cues. For patients that rely heavily on visual input, the audiologist may choose to have the patient face away from the computer during programming. When working with young children, it is advantageous for a second professional to keep the child engaged while looking for responses from the child.
For behavioral measures that require a conditioned response, it is advisable to first condition the child to a non-auditory stimulus. Use of a light or vibrotactile stimulus can yield valuable information regarding the child's ability to provide a conditioned response and the type of responses that might be observed. If the child does not display a conditioned response to non-auditory stimuli, he or she is not likely to provide conditioned responses to auditory stimulation. By obtaining this information prior to use of an auditory stimulus, you can potentially reduce testing time and frustration.
During the use of behavioral measures, it is important to engage the child in a suitable activity. The activity selected should be age appropriate and should provide an adequate level of distraction for the child. Effective use of distraction will help reduce the risk of habituation to the stimulus and will divert the child's attention away from programming equipment. If the child becomes too engaged in an activity, however, it will likely hamper his or her ability to respond to a given stimulus. Clinical reports suggest that pre-implant training of tasks similar to those required during post-implant fitting and therapy can facilitate initial fitting sessions and post-implant progress.4,9 If the child does not have sufficient residual hearing, a tactile aid can be effective for training purposes.9
Standard Adult Procedures:
For older children who have prior experience with sound, the same fitting techniques utilized with adult patients can often be used. When setting THR levels, the child is asked to indicate when he or she detects the stimulus. An ascending approach and standard bracketing procedure are employed. The child may respond by saying "yes" or by raising his or her hand. This process is repeated for each stimulating electrode.
Because most pediatric cochlear implant patients do not have enough prior experience with sound to understand the concept of soft versus loud, obtaining accurate MCL levels using standard adult procedures can be much more difficult. Graphic representations can be used with children who are able to make the symbolic transfer, a skill that is likely to exceed the conceptual abilities of children below 6 years of age.10 Pictures should be age appropriate and should indicate various levels of loudness, ranging from very soft to uncomfortably loud. The stimulus is increased gradually until the child points to an image corresponding to a sound that is loud but comfortable.
For older children who may be more sophisticated listeners, feedback may be verbal in nature. As the stimulus is increased gradually, the child can verbally advise the audiologist to increase or decrease the level of the stimulus. For example, teenage patients who have been implanted for quite some time may simply respond by saying "up" or "down."
Conditioned Play Audiometry (CPA):
Play audiometry is typically used for children between the developmental ages of approximately 2 and 5 years. For the most part, play audiometry is used to determine when a child detects the presence of a stimulus. Because the task is not highly sensitive to determinations of loudness perception, it is obviously more effective for obtaining THR levels than MCL values. It is possible, however, to find creative ways in which to adapt the procedure for the purposes of loudness scaling. Examples would involve toys that include pieces of varying sizes.
If the child understands the concept of big versus small, then it may be possible to condition the child to associate big with loud and small with soft. Once THR levels have been obtained, this value can be used as a starting point for conditioning. The child would be trained to associate stimuli just above THR level with "small" and sounds significantly above THR with "big." It is important to increase the stimulus above THR gradually, while monitoring the child's behavior, to avoid overstimulation.
Visual Reinforcement Audiometry (VRA):
VRA is a testing procedure often useful for children with developmental ages below 24 months. Visually appealing toys (reinforcers) that light up and move are hidden in compartments around the testing room. During the conditioning phase, these toys are activated with, or immediately following, each presentation of an audible stimulus. Caregivers and/or audiology personnel work to keep the child's attention focused away from the reinforcers between presentations. Once the child has discovered that the toy lights and/or moves when sounds are presented, he or she will likely turn toward the toy when a stimulus is heard. With the child conditioned, electrical stimuli are presented on each electrode to obtain THR levels. The reinforcers are then only activated when the child turns toward the toys in response to stimulation. Care should be taken to avoid reinforcement of false positive responses.
To use VRA as a fitting method, the fitting system and VRA reinforcers must be located in the same room. Time spent in the sound booth is often kept to a minimum in clinical settings. Sound booths are often small in size and not well ventilated. Furthermore, there is typically a high demand on sound booths in many centers, and the sound attenuation qualities are not necessary for programming a cochlear implant. For these reasons, most clinicians are not likely to perform fitting sessions within the sound booth. It is possible, however, to install VRA equipment in a separate programming room.
Children in this age range (below 24 months) are rarely capable of providing deliberate feedback regarding loudness perception. MCL values must, therefore, be obtained through Behavioral Observation Audiometry, objective measures, or a combination of these methods.
Behavioral Observation Audiometry (BOA):
BOA is used for children who do not exhibit a conditioned response. BOA is largely subjective and requires the audiologist to make inferences from the child's behavior. Using BOA, the audiologist observes the child for any change in behavior that is time-locked to the stimulus. BOA responses may include: sudden quieting, decrease in activity level, increased vocalization, turning in search of the sound source, touching the ear or head, smiling, laughing, crying, looking for reassurance, or blinking.
In some cases, observational audiometry can provide information regarding both THR and MCL levels. Children may exhibit different behaviors, based on whether the stimulus is soft or loud. Because every child is unique and responds to sound differently, however, interpretation of such behaviors can be problematic. It is important to develop a rapport with each child and to learn the behaviors that are specific to that child. Young children will often only respond to suprathreshold levels of stimulation. It is also not uncommon for children with congenital and profound hearing loss to exhibit negative responses to soft sounds, particularly to high-frequency sounds. Other children may demonstrate no behavioral change until stimulation has exceeded MCL. Thus, THR levels may represent stimulation above the child's true electrical threshold, and MCL values can potentially be set above a level that is comfortably loud.
II. Objective Measures
Electrophysiologic (objective) measures do not require active participation from the patient and can provide useful information regarding MCL levels. Thus, objective fitting procedures can be useful for fitting very young children. Objective methods may also be useful for verifying results obtained during behavioral procedures or for fitting patients who are otherwise unable to fully participate in behavioral testing. There are primarily three objective measures that have been investigated for the purposes of fitting a cochlear implant. Studies have included the Electrically Evoked Stapedius Reflex Threshold (ESRT), the Electrically Evoked Whole Nerve Action Potential (EAP), and the Electrically Evoked Auditory Brainstem Response (EABR).
Electrically Evoked Stapedius Reflex Threshold (ESRT):
A stapedius reflex involves contraction of a tiny muscle in the middle ear in response to loud sounds. In the normal hearing ear, the reflex is elicited bilaterally in response to acoustic stimulation in either ear. The reflex can be measured in either the ipsilateral or contralateral ear using a standard tympanometer. In cochlear implant patients, a stapedius reflex can be measured in the contralateral (non-implanted) ear in response to electrical stimulation through the implant. An Electrically Evoked Stapedius Reflex Threshold (ESRT) is defined as the lowest level of electrical stimulation that elicits a measurable response. Measurement of the ESRT requires passive cooperation, meaning that the patient should remain relatively still and quiet during each recording. Excessive swallowing, talking, or head movements could disrupt the measurement. To record an ESRT, the patient must exhibit a healthy middle ear status. Fluid in the middle ear or dysfunction of the eardrum or middle ear ossicles can prevent measurement of the ESRT. In some cases, a patient with normal middle ear function may not exhibit a measurable reflex response.
The ESRT is measured using a standard tympanometer, cochlear implant software, and the patient's cochlear implant equipment. A soft recording probe is placed in the ear contralateral to the cochlear implant. Tympanometry is performed to confirm normal middle ear status. A good seal of the recording probe and peak compliance of the middle ear should also be confirmed prior to recording an ESRT. The tympanometer is set for measurement of reflex decay, providing a longer recording window. Electrical stimulation is presented through the cochlear implant via interactive software. Stimulation is gradually increased until a sufficient deflection is observed in the reflex decay window, and a standard bracketing procedure is used to determine the stapedius reflex threshold.
ESRT measurements are performed using the same software platform and electrical stimulus utilized for fitting the cochlear implant.10 Thus, stimulus parameters, including pulse duration and repetition rate, can be identical to those used to obtain behavioral judgments for psychophysical levels.1 This allows a direct comparison between ESRT and behavioral measurements, making the ESRT data potentially more predictive for purposes of fitting the cochlear implant. Studies have been conducted to determine the correlation between ESRTs and behavioral measures of THR and MCL values. A positive correlation between two values is represented by a correlation coefficient value between 0.0 and 1.0. A coefficient value of 0.0 would represent a random relationship, and a perfect correlation would be represented by a coefficient of 1.0.
Measurement of the ESRT can be performed both intraoperatively and postoperatively. Studies have shown that postoperative ESRT results are more closely correlated to behaviorally measured MCL values. The overall correlation between postoperative ESRT and MCL is extremely high, indicating that ESRTs are highly predictive of MCL values. Stephan and Welzl-Müller reported a correlation coefficient of 0.92.1 Similarly, Hodges et al. found a correlation of 0.91.10 In most cases, the behavioral MCL is recorded at levels exceeding the ESRT.1 A number of investigators have confirmed that the ESRT can be measured with no discomfort to the patient.1, 5, 10, Stephan and Welzl-Müller concluded that measurement of ESRTs during the fitting process can help avoid overstimulation via the cochlear implant, which is of particular importance when fitting children.1
The ESRT pattern across electrodes is similar to the pattern observed in behaviorally measured MCL values. Thus, individuals with flat MCL responses also demonstrate flat ESRT responses. Likewise, an irregularly shaped MCL response will be replicated by the ESRT response. ESRT data for the MED-EL COMBI 40 and COMBI 40+ cochlear implant systems reveal no significant variance in the ESRT/MCL based on location of the electrode stimulated. This finding is significant in relation to the loudness balancing between MCL values across electrodes.1
Collection of ESRT data on all stimulating electrodes can be completed in less than one hour in most cases.10 Based on findings that patterns for ESRT and behavioral MCL responses are similar across the array,1 it may not be necessary to record ESRT data for every stimulating electrode. For young children, recordings can be performed on a sampling of basal, medial, and apical electrodes. In this way, ESRT measurements could likely be completed on a total of 3-5 electrodes in approximately 15 minutes. For all other electrodes that utilize the same pulse duration, the ESRT-derived MCL value can be interpolated based on the electrodes measured. The use of cartoon videos or the child's natural sleep schedule has been found successful for measuring ESRTs with less cooperative and very young children.10 Hodges et al. concluded that the ESRT is "an accurate and rapid method of estimating maximum comfortable loudness levels, which may be useful in the initial programming of young implant recipients."10
Researchers have compared speech perception results between maps with ESRT-based MCL values and maps based on behavioral measurements of MCL. Spivak et al. showed that 5 of 7 subjects performed either better with the ESRT map or equally well with both maps. This study also found that 4 of 7 subjects preferred the sound quality of the ESRT map when compared with conventionally determined maps.5 Other researchers have reported similar findings.1, 10
Hodges et al. found that the majority of adult subjects with the Nucleus multichannel cochlear implant system prefer a map set using ESRTs over a map set behaviorally, "generally describing the sound as sharper and clearer."10 Spivak et al. concluded that ESRTs "may be an adequate substitute for comfort levels when programming the implant for patients who are unable to make reliable psychophysical judgments."5 Finally, because the ESRT has been shown to be a more stable measurement over time than behavioral MCL values, it has been speculated that the ESRT may be "a more reliable and consistent measure on which to base the map." 5
Electrically Evoked Whole Nerve Action Potential (EAP):
An action potential is a response from neural elements. With regard to cochlear implantation, action potentials from the peripheral auditory nerve can be measured in response to stimulation through the implant. According to Brown et al., "biphasic current pulses are applied to one of the intracochlear electrodes in a monopolar stimulation mode. These current pulses activate the auditory nerve and an EAP is generated. Voltage at a second intracochlear electrode, the recording electrode, is then sampled, amplified and transmitted back across the skin to the externally worn coil. Because intracochlear electrodes are used to both stimulate and record, and because the EAP has a relatively short latency, it is generally not possible to record this potential without significant artifact contamination." In the EAP recording systems currently available for commercial use, a subtraction technique must be implemented to extract the EAP response from the stimulus artifact.11,12
While additional recording equipment is not needed, measurement of the EAP requires a separate software application and uses stimulus parameters that differ from those typically used for fitting purposes. As an example, Neural Response Telemetry (NRTTM) is an EAP software application. Although the acronym "NRT" is often used interchangeably with EAP, the two are not synonymous. The EAP is a neural response. NRTTM is the trademark name for a software application. The NRTTM system includes a number of parameters that must be implemented correctly to optimize EAP results. Among these parameters are the stimulation mode, sampling window and rate, probe level, masker level, masker advance, sampling delay, amplifier gain, pulse duration, and number of sweeps.2, 8, 11, 13
With the NRTTM system, the masker pulse is utilized within the subtraction technique to separate the EAP response from stimulus artifact.12 Thus, the masker level must be set appropriately to record an adequate neural response. Varying implementations of the masker pulse have been utilized within and across various research studies.2, 11, 12 Hughes et al. reported a potential influence on the slope of EAP growth functions with use of different masker levels over time.12 Similarly, the number of sweeps has been shown to impact EAP results. Many investigators vary the number of sweeps based on amplifier gain settings or amplitude of the EAP waveform.2, 8, 11, 13 Brown et al. found that use of 1000 sweeps resulted in a small but statistically significant improvement in the average error of EAP results when compared to use of 100 sweeps.11
Certain stimulus parameters available within the NRTTM system differ from those typically implemented via the fitting software. The majority of research studies conducted with the Nucleus 24 cochlear implant and NRTTM system cite a stimulation rate of either 35 Hz (for software version 1.4) or 80 Hz (for software versions 2.01 and 2.04). In most cases, however, study subjects were utilizing a rate of 250 Hz within their cochlear implant maps.2, 8, 11, 12, 13 This difference in repetition rate has been cited as a probable cause for EAP thresholds that exceed the level of comfort.2, 13
As with the ESRT, there are patients for whom a clear EAP response cannot be recorded.2, 11, 13 Likely conditions under which a reliable EAP response cannot be recorded include cochlear malformation, cochlear ossification,13 or a narrow electrical dynamic range.11 In a study by Smoorenburg et al., it was possible to record an EAP response on each stimulating electrode in only 13 of 27 subjects (48%). For studies reporting higher success rates, measurement of the EAP response is typically restricted to recordings on fewer electrodes.2
Studies similar to those performed with ESRT responses have been conducted with the EAP response. Researchers have compared EAP data to behaviorally obtained THR and MCL levels. Comparative analysis of speech perception results between EAP-based maps and those created using conventional fitting techniques has also been performed. All studies reported here were conducted with the Nucleus 24 cochlear implant system (CI24M) and NRTTM software.
Raw EAP data from adult cochlear implant patients has shown weak to poor correlations with both THR and MCL levels. Brown et al. demonstrated a correlation of 0.547 between EAP threshold and THR level and 0.565 between EAP threshold and MCL level.11 Smoorenburg et al. reported correlation coefficients of 0.64 between overall EAP and THR levels, 0.39 between overall EAP and MCL levels, 0.82 between EAP slope and THR, and 0.36 between EAP slope and MCL.2 Table 1 provides a summary of these values.
Research findings have also demonstrated a great deal of intersubject variability. 2, 8, 11, 13 For some subjects, the EAP response falls closer to THR level; whereas, responses fall closer to MCL levels in other subjects.8, 13 Brown et al. found that EAP thresholds fall between THR and MCL levels for some patients and exceed MCL values for others.11 It should also be noted that each of the correlation coefficients cited above were obtained using the SPEAK coding strategy at a stimulation rate of 250 Hz. Higher stimulation rates, such as those available with the ACE coding strategy, would result in a greater discrepancy in these rates and, possibly, a greater error in the correlation.
It is widely accepted that raw EAP data is not adequate for estimation of psychophysical levels. At this time, a correction factor must be applied for the EAP to be of any predictive value. A number of investigators have described various correction factors, and there does not yet seem to be a universal approach for calculation of predicted map values from the EAP. Brown et al. described a calculation based on comparison of the EAP and behavioral responses from a single, medial electrode. This correction factor was applied to each electrode for which an EAP was recorded. THR and MCL levels on electrodes for which an EAP response was not recorded were interpolated, based on mapping levels from adjacent electrodes. Using this calculation, Brown et al. noted a stronger correlation between EAP data and behavioral measures in adults, reporting a coefficient value of 0.83 between EAP threshold and THR level and 0.77 between EAP threshold and MCL level.11 Alternatively, Smoorenburg et al. measured EAP thresholds across the full electrode array and described a correction factor based on stimulation to live voice and the "EAP threshold profile."2
The correction factor utilized by Brown et al. was developed with the assumption that the relationship between EAP measures and THR and MCL levels are fairly constant across the array. Several studies, however, have demonstrated intrasubject variability in the EAP.2, 8, 11, 13 Seyle and Brown found that differences between EAP-derived maps and conventionally-derived maps were greatest at the apical end of the electrode array.8 Similarly, Brown et al. reported that EAP thresholds occur at lower levels in the dynamic range for the apical electrodes.11 Additionally, Smoorenburg et al. demonstrated that individual EAP thresholds tend to exceed MCL values for the basal electrodes.2
A number of studies have demonstrated a tendency for EAP thresholds to exceed levels of maximum comfort. Brown et al. reported a 38 percent chance that individual EAP thresholds will exceed the behavioral MCL and a 28 percent chance that individual EAP thresholds will exceed the MCL by 5 or more programming units.11 Smoorenburg et al. found EAP thresholds that exceeded MCL values by as much as 29 programming units.2 Hughes et al. found that, for older children, MCL values based on the NRT stimulus were higher than behavioral MCL values in most cases.13 In part, EAP thresholds corresponding to levels above behavioral MCLs can be attributed to the lower repetition rate used for EAP measures compared to those utilized during fitting of the speech processor.2, 13
In 2001, Hughes et al. revealed changes in the EAP over time. Statistically significant changes in the EAP thresholds of children were observed until 3 to 8 months following initial stimulation. Measures of EAP slope in children did not stabilize until the 12-month post-implant evaluation. Moreover, longitudinal trends in EAP measures mirrored THR level more closely than MCL level;12 yet, previous studies have demonstrated that accurate estimation of MCL levels and loudness balancing are of greater value than THR levels.2, 3 Smoorenburg et al. concluded that EAP thresholds "cannot be used to predict the overall C level or the tilt of the C profile" and that "prediction of the most critical factor in speech perception, the slope of the maximum stimulation curve, from the [EAP] thresholds is poor." 2
The study by Hughes et al. demonstrated serious implications for the collection of EAP recordings over time. Comparison of EAP data with behavioral responses in children should be restricted to measurements obtained during a single fitting session. Because the EAP thresholds and slope, as well as psychophysical levels, show significant changes throughout the first year of implant use, EAP recordings obtained at multiple fitting sessions cannot be combined to form a complete EAP profile. Consequently, if the EAP is to be used for fitting of the speech processor, it is important that EAP measures are repeated several times during the first 12 months following initial stimulation. Only after the EAP has stabilized can recordings from a single fitting session be of use in future fitting sessions. 12
Finally, mean speech perception data has shown no benefit when the speech processor is fit based on EAP measures. While Seyle and Brown found significant variability in speech perception results, subjects tended to perform more poorly with maps based on the EAP than with conventionally derived maps. These differences were statistically significant at presentation levels of 70 dB SPL, the industry standard for speech testing with cochlear implant patients.8
Electrically Evoked Auditory Brainstem Response (EABR):
The ABR is recording of bioelectric activity originating from the auditory nerve and portions of the brainstem in response to sound. In cochlear implant patients, the ABR can be elicited and utilized in much the same manner that the EAP is used.14 Like the EAP, an EABR measures the response from neural elements in response to electrical stimulation through the cochlear implant. Recording the EABR requires external recording equipment and surface recording electrodes. To obtain adequate waveforms, the patient must remain still and quite. Results can be difficult to obtain on infants and children while they are awake. Prescription of a sedative may increase quality of the waveform and reduce testing time. Sedation allows the child to remain calm and still, reducing the noise floor and muscle artifact.
According to Brown et al., "recording the EABR is fairly straightforward. With the EABR, no subtraction procedure is used and because wave V of the EABR has a latency of approximately 4 msec, the response is fairly easy to record, even in situations like deformed cochleas or instances where extensive drilling of the cochlea was required."11 For this reason, the EABR has been more successful in recording neural responses than the EAP for patients with cochlear malformations and ossification.11, 13 Another significant difference between EABR and EAP recordings involves the population of nerve cells that contribute to the response. "The EABR is recorded using scalp electrodes and is a gross potential that reflects contributions of all the neural units that respond to the electrical stimulus." 11 The EAP, however, is recorded from an electrode within the cochlea and may, therefore, reflect activity from a smaller population of nerve cells. The EAP does not reflect neural activity from higher levels in the brainstem. 11 For this reason, information about processing at higher levels, such as temporal summation effects, cannot be determined by EAP recordings.
Correlation coefficients between the EABR and behavioral responses are similar to those obtained with the EAP. Like EAP data, EABR measures require a correction factor to be of any predictive value. Interestingly, EABR thresholds have been found to occur at levels slightly below the EAP threshold in adult patients. This difference is potentially due to differences in the noise floor between the two measures and in the number of sweeps used to record each measure. 11
The EABR offers advantages and disadvantages when compared to the EAP. The EABR is used less frequently, because it requires external recording equipment, scalp electrodes and, in some cases, sedation. Additionally, EABR measurements can be more time-consuming and more susceptible to problems with artifact. With slightly lower overall response levels, however, the EABR may be less likely to result in overstimulation. Finally, an EABR may be present in patients for whom the EAP cannot be recorded.
III. Speech Perception Measures
One additional method that can assist in successful fitting of the speech processor includes speech perception measures. Speech perception measures can be used to determine fitting needs and to assess performance and progress over time. Speech perception testing should incorporate a battery of measures designed to evaluate a broad range of perceptual abilities. To assess speech understanding in children, separate test batteries may be required for preschool- and school-age children. For example, a preschool battery, should require no more than 1 hour to complete, should incorporate materials along a continuum of difficulty, should include tasks familiar to the child and examiner, should require little training, and should use objects (rather than pictures) whenever possible. A battery for school-age children should include measures that assess skills along a continuum, ranging from closed-set tests of pattern perception to open-set tests with sentences and single words.9
Perceptual abilities develop in a non-sequential manner, with open-set skills emerging before closed-set skills have been mastered. Thus, all materials in a test battery should be administered at each testing interval. Rather than waiting for a child's performance to reach the skill level required for each test, a clearly defined set of criteria should be determined for discontinuing testing, and each examiner should adhere to the same standards. When testing is discontinued due to a child's inability to perform the task, a score of zero percent should be assigned for open-set measures and chance performance for closed-set measures. 9
Closed- and open-set speech perception measures cannot fully identify and evaluate a child's performance. A great deal of beneficial information can be observed prior to and during development of the requisite skill need for closed- and open-set tests. For this reason, feedback from parents and professionals should also be considered during follow-up care and fitting. It is important for the cochlear implant team to maintain a good working relationship and open communication with a child's family members, caregivers, educators, and therapists. A formal questionnaire, like the Meaningful Auditory Integration Scale (MAIS), can be useful in obtaining and categorizing feedback from parents and professionals. Depending on the age and abilities of the child, an Infant-Toddler version of the scale, the IT-MAIS, is also available.
A formal speech perception test battery is only administered at specific intervals. Alternate techniques may be incorporated into each fitting session. The Ling 6 Sound Test, for example, is quick and easy to perform and can be administered at any time. However, because children often require a period of adjustment with a new map, performance on any measure of speech perception should be examined over time. Care must also be taken not to place too much emphasis on single phonemes.
Discrimination between specific phonemes depends on more than one formant. For many phonemes, formant frequencies include both low- and high-frequency information, and phoneme errors can be related to any of these formants. Changes to the map based on specific phoneme errors should be performed with extreme caution and only when the specific formant frequency responsible for the error can be identified. Finally, according to Robbins and Iler-Kirk, "it should be kept in mind that speech perception is a phenomenon that cannot be observed directly. It can only be inferred based upon the child's responses. This inference may be an imperfect estimate, particularly in the case of the young or uncooperative child who often can do more than he or she will do during testing." 9
Conclusion
At a minimum, fitting a cochlear implant requires accurate measurement of the Threshold (THR) and Most Comfortable Loudness Level (MCL) for each stimulating electrode. THR and MCL are psychophysical judgments of loudness that are measured in clinical units of electrical current. Current units are not directly comparable to the decibel scale and cannot be directly correlated to the audiogram. Furthermore, the frequency bands afforded by a cochlear implant system are not always represented on the audiogram. Certain frequency bands may include frequencies not represented by the octaves and interoctaves available on standard audiometers. Finally, because the speech processor is specifically designed to process speech (a wide spectrum input signal), soundfield thresholds obtained using frequency-specific warble tones do not necessarily reflect the true sensitivity or performance of the cochlear implant. For these reasons, it is not recommended that cochlear implant patients be fit based primarily on information obtained from the audiogram.
In some cases, the audiogram may offer a potential benefit in identifying problematic peaks in the frequency response of a patient's map. If the audiogram shows a soundfield threshold that is significantly better at 500 Hz, for example, it could indicate the need to re-establish accurate THR and MCL values for the electrode corresponding to that particular frequency band. However, it is important to note that each electrode is responsible for a range of frequencies. For example, if 500 Hz is assigned to the same electrode responsible for 750 Hz, any change in the psychophysical levels for that electrode would likely affect both 500 Hz and 750 Hz. However, as an interoctave, 750 Hz may not be routinely tested during soundfield testing with the implant.
In reality, fitting techniques often incorporate a variety of approaches, particularly for pediatric cochlear implant patients. Older children can often be fit based on verbal feedback, with techniques similar to those used for adult cochlear implant patients. For younger children or children with special needs, the fitting process is more complex. Many children do not possess the hearing experience, language skills, or conceptual development necessary to provide sophisticated feedback regarding subjective percepts of loudness. Behavioral testing procedures typically employed in pediatric audiology can be adapted for fitting purposes. Most behavioral measures are primarily useful for obtaining THR information. Measurement of MCL levels and loudness balancing are much more difficult to obtain using behavioral fitting methods.
Research has shown that MCL levels and loudness balancing have a greater influence on speech understanding than THR levels.2, 3 Studies demonstrated that a significant decrease in THR levels did not negatively affect speech perception scores, while a decrease in MCL values resulted in poorer speech perception.2 Additionally, changes to the slope or balancing of MCL levels resulted in poorer speech understanding.2, 3 Therefore, cochlear implant fittings must, devote a greater emphasis on obtaining and balancing MCL levels.
Finding an effective means for obtaining accurate MCL levels with very young or special needs children is a formidable task. A number of studies have researched potential benefits of various objective testing procedures for the purposes of fitting a cochlear implant. There are primarily three objective measures that have been investigated: Electrically Evoked Stapedius Reflex Threshold (ESRT), Electrically Evoked Whole Nerve Action Potential (EAP), and Electrically Evoked Auditory Brainstem Response (EABR).
Research findings indicate that the ESRT is of greater predictive value than the EAP or EABR for estimation of psychophysical levels. Postoperative ESRT thresholds show an extremely high correlation with behaviorally obtained MCL levels and the MCL pattern across electrodes. Additionally, fitting the speech processor based on ESRT data has been shown to result in speech perception scores equal to or better than those achieved with conventional fitting techniques.5
For patients implanted with the MED-EL COMBI 40+ Cochlear Implant System, the fitting process can be extremely straightforward. Psychophysical levels are typically similar between adjacent electrodes and follow a predictable pattern across the electrode array. Measurement of an ESRT on 3-5 electrodes can be accomplished in approximately 15 minutes and is sufficient to accurately predict MCL levels and the MCL response pattern across the full array. Because the ESRT is a relatively stable measure over time,5 it is advisable to initially set MCL levels below ESRT threshold in children. The MCL values can be gradually increased over time to a level corresponding to the ESRT threshold. THR levels can be set at roughly 10 percent of the MCL value on each electrode. This fitting method should provide speech perception scores equal to or better than those possible with behavioral fitting procedures. Thus, even very young children and children with special needs can be fit reliably with the COMBI 40+.
In addition to implementing effective techniques for fitting the speech processor, it is important to monitor a patient's progress over time with the implant. Speech perception testing can demonstrate the overall benefit of implant use, as well as the impact of programming changes. It is important to note that the impact of a change to the patient's map is often not immediately apparent. For many children, it takes time to adjust to a programming change. Parents should be counseled to observe the child's behavior and/or speech production following each fitting session. While significant changes in the child's responses to sound or speech perception may indicate the need for further follow-up, not all changes in speech production and performance are directly related to mapping characteristics. Children may show regression in certain phonemes or skills while developing new phonemes and skill. Most children can be expected to show progress over time based solely on their stage and rate of development.
Finally, it is beneficial to understand and utilize the services offered by each cochlear implant manufacturer. Manufacturers provide audiological support via telephone and on-site visits with the cochlear implant center. MED-EL Corporation, for example, employs a Clinical Manager for each region of the United States. Additionally, with MED-EL's audiology on-call service, telephone assistance is available 24 hours a day for clinical emergencies. Cochlear implant manufacturers can assist with difficult cases or fitting dilemmas and are a great resource for clinical questions or concerns. MED-EL also has teams to assist with surgical support and patient support needs.
Acknowledgements
The author greatly appreciates the professional review and valuable comments provided during the development of this manuscript by Ingeborg Hochmair, Chris Bertrand, Tim Campos, Patrick D'Haese, Darla Franz, Kim Jackson, Christy Kruger, Jennifer Lingvai, Marcus Schmidt, and Martin Zimmerling.
References
1Stephan K, Welzl-Müller K. Post-operative stapedius reflex tests with simultaneous loudness scaling in patients supplied with cochlear implants. Audiology. 2000;39:13-18.
2Smoorenburg GF, Willeboer C, van Dijk, JE. Speech perception in Nucleus CI24M cochlear implant users with processor settings based on electrically evoked compound action potential thresholds. Audiol Neurootol. 2002;7:335-47.
3Dawson PW, Skok M, Clark GM. The effect of loudness imbalance between electrodes in cochlear implant users. Ear Hear. 1997;18:156-65
4Langman AW, Quigley SM, Souliere CR. Cochlear implants in children. Pediatric Otolaryngology. 1996;43:1217-31.
5Spivak LG, Chute PM, Popp AL, Parisier SC. Programming the cochlear implant based on electrical acoustic reflex thresholds: patient performance. Laryngoscope. 1994 Oct;104(10):1225-30.
6Waltzman SB, Cohen NL. Cochlear implantation in children younger than 2 years old. American Journal of Otology. 1998;19(2):158-62.
7Balkany, TJ, Hodges A, Miyamoto RT, Gibbin K, Odabassi O. Cochlear implants in children. Otolaryngol Clin North Am. 2001 Apr;34(2):455-67.
8Seyle K, Brown CJ. Speech perception using maps based on neural response telemetry measures. Ear Hear. 2002 Feb;23(1 Suppl):72S-79S.
9Robbins AM, Iler-Kirk K. Speech perception assessment and performance in pediatric cochlear implant users. Seminars in Hearing. 1996;17(4):353-69.
10Hodges AV, Balkany TJ, Ruth RA, Lambert PR, Dolan-Ash S, Schloffman JJ. Electrical middle ear muscle reflex: use in cochlear implant programming. Otolaryngol Head Neck Surg. 1997 Sep;117(3 Pt 1):255-61.
11Brown CJ, Hughes ML, Luk B, Abbas PJ, Wolaver A, Gervais J. The relationship between EAP and EABR thresholds and levels used to program the Nucleus 24 speech processor: data from adults. Ear Hear. 2000 Apr;21(2):151-63.
12Hughes ML, Vander Werff KR, Brown CJ, Abbas PJ, Kelsay DM, Teagle HF, Lowder MW. A longitudinal study of electrode impedance, the electrically evoked compound action potential, and behavioral measures in Nucleus 24 cochlear implant users. Ear Hear. 2001 Dec;22(6):471-86.
13Hughes ML, Brown CJ, Abbas PJ, Wolaver AA, Gervais JP. Comparison of EAP thresholds with MAP levels in the Nucleus 24 cochlear implant: data from children. Ear Hear. 2000 Apr;21(2):164-74.
14Balkany TJ, Hodges AV, Eshragni AA, Butts S, Bricker K, Lingvai J, Polak M, King J. Cochlear implants in children--a review. Acta Otolaryngol. 2002 Jun;122(4):356-62.
For more information on MED-EL click here.
Click here to visit the MED-EL website.
Fitting Techniques for the Pediatric Cochlear Implant Patient
May 12, 2003
Related Courses
1
https://www.audiologyonline.com/audiology-ceus/course/singing-in-rain-using-music-22785
Singing in the Rain: Using Music to Reinforce Listening (Professionals)
Presented by Christine Barton, this course provides participants with additional approaches to using music for spoken language development in young deaf and hard of hearing children. A special emphasis on the use of songs that focus on listening and language development while a young child is playing in the water will be provided.
textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Singing in the Rain: Using Music to Reinforce Listening (Professionals)
Presented by Christine Barton, this course provides participants with additional approaches to using music for spoken language development in young deaf and hard of hearing children. A special emphasis on the use of songs that focus on listening and language development while a young child is playing in the water will be provided.
22785
Online
PT60M
Singing in the Rain: Using Music to Reinforce Listening (Professionals)
Presented by Chris Barton, MM, MT-BC
Course: #22785Level: Intermediate1 Hour
No CEUs/Hours Offered
Presented by Christine Barton, this course provides participants with additional approaches to using music for spoken language development in young deaf and hard of hearing children. A special emphasis on the use of songs that focus on listening and language development while a young child is playing in the water will be provided.
2
https://www.audiologyonline.com/audiology-ceus/course/implementation-cochlear-implants-enhanced-candidacy-37377
Implementation of Cochlear Implants: Enhanced Candidacy Criteria and Technology Advances
The participant in this course will understand the extended candidacy criteria with cochlear implantation and expectations. The course will cover implanting under age one, hybrid hearing with cochlear implantation, CI under local anesthesia, single-sided deafness, cochlear implantation, and auditory brainstem implantation.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Implementation of Cochlear Implants: Enhanced Candidacy Criteria and Technology Advances
The participant in this course will understand the extended candidacy criteria with cochlear implantation and expectations. The course will cover implanting under age one, hybrid hearing with cochlear implantation, CI under local anesthesia, single-sided deafness, cochlear implantation, and auditory brainstem implantation.
37377
Online
PT60M
Implementation of Cochlear Implants: Enhanced Candidacy Criteria and Technology Advances
Presented by J. Thomas Roland, MD Jr.
Course: #37377Level: Intermediate1 Hour
AAA/0.1 Intermediate; ACAud inc HAASA/1.0; AHIP/1.0; ASHA/0.1 Intermediate, Professional; BAA/1.0; CAA/1.0; Calif. SLPAB/1.0; IACET/0.1; IHS/1.0; Kansas, LTS-S0035/1.0; NZAS/1.0; SAC/1.0
The participant in this course will understand the extended candidacy criteria with cochlear implantation and expectations. The course will cover implanting under age one, hybrid hearing with cochlear implantation, CI under local anesthesia, single-sided deafness, cochlear implantation, and auditory brainstem implantation.
3
https://www.audiologyonline.com/audiology-ceus/course/using-gsi-for-cochlear-implant-39682
Using GSI for Cochlear Implant Evaluations
This course is designed to educate audiologists on the practical workflow for patients who require cochlear implants. From Food and Drug Administration (FDA) approved indications and Medicare requirements to pre-op and post-op evaluations, audiologists will gain a clear understanding of the cochlear implant process.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Using GSI for Cochlear Implant Evaluations
This course is designed to educate audiologists on the practical workflow for patients who require cochlear implants. From Food and Drug Administration (FDA) approved indications and Medicare requirements to pre-op and post-op evaluations, audiologists will gain a clear understanding of the cochlear implant process.
39682
Online
PT60M
Using GSI for Cochlear Implant Evaluations
Presented by Joseph Dansie, AuD
Course: #39682Level: Introductory1 Hour
AAA/0.1 Introductory; ACAud inc HAASA/1.0; AHIP/1.0; BAA/1.0; CAA/1.0; Calif. SLPAB/1.0; IACET/0.1; IHS/1.0; Kansas, LTS-S0035/1.0; NZAS/1.0; SAC/1.0
This course is designed to educate audiologists on the practical workflow for patients who require cochlear implants. From Food and Drug Administration (FDA) approved indications and Medicare requirements to pre-op and post-op evaluations, audiologists will gain a clear understanding of the cochlear implant process.
4
https://www.audiologyonline.com/audiology-ceus/course/adult-assessments-in-hearing-healthcare-38660
Adult Assessments in Hearing Healthcare: Working Across the Continuum
This five-course series on adult assessments in hearing health is intended to stimulate collaborative approaches for hearing health professionals, regardless of what hearing technologies they typically provide. Ideally, professionals will support patients in their long-term hearing loss journey, facilitating transitions when appropriate and a comfortable sense of the range of ways hearing loss can be addressed throughout one’s hearing journey.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Adult Assessments in Hearing Healthcare: Working Across the Continuum
This five-course series on adult assessments in hearing health is intended to stimulate collaborative approaches for hearing health professionals, regardless of what hearing technologies they typically provide. Ideally, professionals will support patients in their long-term hearing loss journey, facilitating transitions when appropriate and a comfortable sense of the range of ways hearing loss can be addressed throughout one’s hearing journey.
38660
Online
PT300M
Adult Assessments in Hearing Healthcare: Working Across the Continuum
Presented by Camille Dunn, PhD, Susan Good, AuD, MBA, Alejandra Ullauri, AuD, MPH, Ted McRackan, MD, MSCR, Donna L. Sorkin, MA, Rene Gifford, PhD
Course: #38660Level: Intermediate5 Hours
AAA/0.5 Intermediate; ACAud inc HAASA/5.0; ASHA/0.5 Intermediate, Professional; BAA/5.0; CAA/5.0; Calif. SLPAB/5.0; IACET/0.5; IHS/5.0; Kansas, LTS-S0035/5.0; NZAS/3.0; SAC/5.0; Tier 1 (ABA Certificants)/0.5
This five-course series on adult assessments in hearing health is intended to stimulate collaborative approaches for hearing health professionals, regardless of what hearing technologies they typically provide. Ideally, professionals will support patients in their long-term hearing loss journey, facilitating transitions when appropriate and a comfortable sense of the range of ways hearing loss can be addressed throughout one’s hearing journey.
5
https://www.audiologyonline.com/audiology-ceus/course/adult-perceptions-hearing-status-and-38407
Adult Perceptions of Hearing Status and Options: Professionals Facilitating a Life-long Hearing Journey, in partnership with American Cochlear Implant Alliance
Adults with severe-to-profound hearing loss utilizing hearing aids are typically not benefitting sufficiently from traditional amplification and often would benefit from hearing implants. This course will review ways hearing care professionals can support adult patients who may benefit from implants (either now or in the future) by presenting information on the cochlear implant (CI) option early in an individual’s hearing journey. A CI is not a last resort, but rather a means for those who fall within the guidelines to experience hearing improvement, rather than continued decline.
auditory, textual, visual
129
USD
Subscription
Unlimited COURSE Access for $129/year
OnlineOnly
AudiologyOnline
www.audiologyonline.com
Adult Perceptions of Hearing Status and Options: Professionals Facilitating a Life-long Hearing Journey, in partnership with American Cochlear Implant Alliance
Adults with severe-to-profound hearing loss utilizing hearing aids are typically not benefitting sufficiently from traditional amplification and often would benefit from hearing implants. This course will review ways hearing care professionals can support adult patients who may benefit from implants (either now or in the future) by presenting information on the cochlear implant (CI) option early in an individual’s hearing journey. A CI is not a last resort, but rather a means for those who fall within the guidelines to experience hearing improvement, rather than continued decline.
38407
Online
PT60M
Adult Perceptions of Hearing Status and Options: Professionals Facilitating a Life-long Hearing Journey, in partnership with American Cochlear Implant Alliance
Presented by Donna L. Sorkin, MA
Course: #38407Level: Intermediate1 Hour
AAA/0.1 Intermediate; ACAud inc HAASA/1.0; AHIP/1.0; ASHA/0.1 Intermediate, Professional; BAA/1.0; CAA/1.0; Calif. SLPAB/1.0; IACET/0.1; IHS/1.0; Kansas, LTS-S0035/1.0; NZAS/1.0; SAC/1.0
Adults with severe-to-profound hearing loss utilizing hearing aids are typically not benefitting sufficiently from traditional amplification and often would benefit from hearing implants. This course will review ways hearing care professionals can support adult patients who may benefit from implants (either now or in the future) by presenting information on the cochlear implant (CI) option early in an individual’s hearing journey. A CI is not a last resort, but rather a means for those who fall within the guidelines to experience hearing improvement, rather than continued decline.

